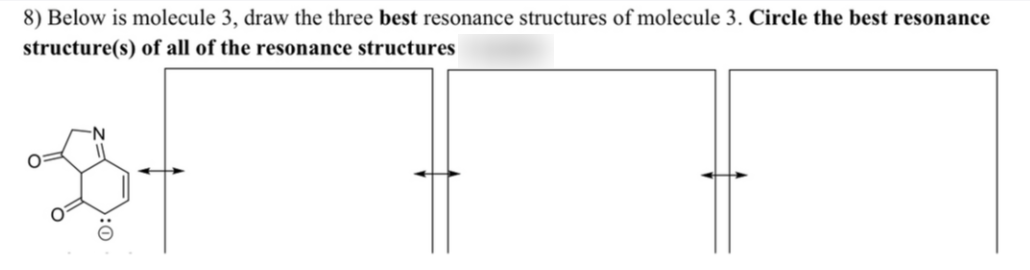
8) Below is molecule 3, draw the three best resonance structures of molecule 3. Circle the best resonance structure(s) of all of the resonance structures
Q: How do I find how many resonance forms does a compound have beside drawing
A: How to find how many resonance forms does a compound have beside drawing?
Q: RESONANCE THEORY 1. Explain why and when there is a need for using resonance theory ?
A: Resonance is the ability of system to move its pi electrons in the system. 2. The delocalized…
Q: Follow the curved arrows to draw a second resonance structure for eachspecies.
A: Given species,
Q: 9. Draw all resonance structures of the following compounds, showing electron movement with curved…
A:
Q: a) Draw the structure below and the two other important resonance forms. b) Circle the most…
A: A question based on introduction to organic chemistry that is to be accomplished.
Q: O Draw all important resonance structures for the compound shown below.
A:
Q: Use resonance structures to explain why the compound shown below is considered to be antiaromatic.
A:
Q: ОН d) b)
A: Resonance structures are formed by the movement of electrons between the conjugated atoms in a…
Q: 6. Draw four alternative, reasonable resonance structures to represent the anion show below. Also,…
A: It is the path to describe the bonding in a certain molecule by combination of certain contributing…
Q: 5. Draw as many resonance structures as possible for p-nitrophenol (see below). Make sure to include…
A: Resonance structures are sets of Lewis structures that describe the delocalization of electrons in a…
Q: HN ÓH
A: The two resonating structures will be formed as follows:
Q: 2.Use curved arrows to draw all the significant resonance structures for the following molecule and…
A: Interpretation - To draw all the significant resonance structure for the following molecule which…
Q: 2) Draw two more resonance structures for the following:
A: Resonating structure
Q: 7. a) Draw the resonance arrows to form the structure on the right, b) Circle the major resonance…
A: The transformation given is,
Q: Click to see additional instructions The following questions are about the best Lewis structure of…
A: See the lewis structure of O3 +1 Single bond, double bond, and lone pairs of electrons.
Q: Draw a second resonance form for the structure shown below. :o:
A:
Q: 7. Draw two possible resonance structures for the following molecule.
A: Since you have posted more than one question, we will solve only first question for you. If you want…
Q: Which of the following species (B, C,D) is a valid resonance of A? Use curved arrows to show how A…
A: Valid resonating structure
Q: `NH
A: Possible resonating structure are:
Q: Draw a second resonance form for the structure shown below.
A: Resonance is a method of representation of bonds in various molecules or ions. Resonance structure…
Q: Draw all reasonable resonance structures for each compound.
A: Rsonance structure : Resonance means distribution of electron density & forms nearly same energy…
Q: Which resonance form contributes most to the overall structure of the molecule? Click on a letter A…
A: The most stable resonating structure providing the major contribution should have : Octets of all…
Q: Shown below is the major resonance structure for a molecule. Draw the second best resonance…
A:
Q: Decide wnether each row is a set of resonance structures for a single molecule and select "yes" or…
A:
Q: 4. Draw three possible resonance structures of the following molecule.
A: Resonance structure:If the Lewis structure of a molecule or ion cannot explain by a single structure…
Q: Which of these species are free radicals? Check all that apply. NO2 NO O2 O3
A: Free radical is neutral species which is having an unpaired electron.
Q: (a) Write three more resonance structures for each of compounds 1 and 2. (b) In each of compounds 1…
A: Resonance: The capability of a system to delocalize the π electrons in it.
Q: ▾ Part A Draw the resonance structure of the following substance. Interactive 3D display mode H3C…
A: Resonance structures represents a set of Lewis structures which shows the delocalization of…
Q: All resonance structures with arrow. Major resonance?
A:
Q: 1. Draw all resonance structures for the following molecules. CF3 a. b.
A: Resonance is defined as transfer of charge from one atom to another atom
Q: ) Draw all reasonable resonance structures for the following molecules. b) Label the major and minor…
A: Resonance structures are the charge delocalized structures and the more stable ones are the major…
Q: Can someone draw and explain this resonance?
A: In resonance structures, the bonded pair of electrons convert into the lone pair electrons and lone…
Q: I struggle with resonance. Can you help show me how to do these?
A: Resonance is delocalisation of pi-electrons and unshared pair of electrons to attain stability.The…
Q: For this molecule draw the 2 best resonance structures in the boxes. Circle the best.
A: The lone pair of electrons of oxygen makes a double bond with the carbon atom. It eliminates the…
Q: For each of the following pairs of resonance structures, circle the one that would make greater…
A: IN PART A 1st structure contributes to the actual resonance hybrid because the + ve sign is on less…
Q: Draw all reasonable resonance structures for each species.
A: Please find the attached image below for the solution-
Q: Determining the Relative Energy of Resonance Structures and the Hybrid Draw a second resonance…
A: The electrons' delocalization over atoms keeping their positions fixed is referred as "resonance".…
Q: Provide the two resonance structures of the molecules shown. Circle the best resonance structure out…
A: Stability of carbocations: primary < secondary < tertiary
Q: Decide whether each row is a set of resonance structures for a single molecule and select "yes" or…
A: In this question ,we are going to compare different sets of resonance structure.
Q: A A with partial charges B B with partial charges
A:
Q: Decide whether each row is a set of resonance structures for a single molecule and select "yes" or…
A: Resonance structure:If the Lewis structure of a molecule or ion cannot explain by a single structure…
Q: 4. Draw all resonance structures for each compound below (including arrow pushing) and indicate…
A:
Q: C. Provide reasonable resonance structures for the molecules below that show the movement of…
A: In this question, we will write the all resonance structures for the given compounds. You can see…
Q: Considering the structure on the far left as the original resonance structure, which of the…
A: The original structure and the resonance structures given are,
Q: Draw all possible resonance structures for the species below
A:
Q: Problem attached
A: The best three resonance structure of the molecule 3 has to be drawn below.
Q: :: :0: О.
A:
Q: Write the resonance structure of the following anions.
A: Resonating structures are the structure of the same species which are sets of Lewis structures that…
Q: Draw a second resonance form for the structure shown below. ö: ö:
A: Resonance structure are sets of Lewis structures that describe the de-localization of electrons in a…
Step by step
Solved in 3 steps with 1 images

- 1. Add nonbonding electron pairs to each atom where it is implied by the line-angle drawings shown below. 2. Use the curved arrow formalism to generate four additional, valid resonance structures for each compound. 3. Rank your resonance structures in order of increasing contribution to the overall nature of the molecule.All resonance structures with arrow. Major resonance?I need to indicate how the first resonance structure (the one with #4 above it) turns into the second one with arrows, but I cannot figure it out.
- I got this marked wrong on an assignment, but I'm not sure why. Please help me find the correct resonance structure.a) Draw all reasonable resonance structures for the following molecules. b) Label the major and minor resonance structures. If they are all equal write equal.For the structure shown in the box below identify two corresponding correct resonance structures.

