Q: The amount of isotope decreases in a first-order process. If 13.5% of the original amount of 14C…
A: For a first order reaction : t1/2 = 0.693/k k = rate constant t1/2 = Half life Initial…
Q: 3. Attached is the IR for one of the compounds you crystallized in this lab. (a) Is this molecule 2-…
A:
Q: Explain how radiolabelling has been used as a tool for elucidation of the mechanism of alkene…
A: Radiolabelling is a technique used to label molecules with radioactive isotopes, allowing…
Q: For the following compound draw (R) and (S) absolute configurations using Fischer projections (2…
A:
Q: 2. Which reaction would synthesize Br-C-C-H ? Be sure to explain clearly. Br H 1. CH3-CH3 + 2 Br2 2.…
A: Alkenes reacts with halogens through a halonium ion intermediate to form vicinal dihalides. Alkanes…
Q: mass
A: The mass can be calculated with the formula = Density × Volume As given, phase 1 contains 8% of…
Q: In the reaction shown below, methane is being H H-CH- 02 0=C=0 H reduced and is therefore the…
A: Since, In oxidation reaction, Oxidation number increases or number of oxygen increases or number of…
Q: Directions: Show all work (all conversions and final answer) for each problem and balance each…
A:
Q: N₂(g) + 3H₂(g) = 2 NHz (g) 1. In the Haber if •process, what happens. Hydrogen gas? you add more
A: Since, According to Le-Chateliar principle--Changing concentration affect the equilibrium. When…
Q: A particular allotrope of nitrogen, N4, was first detected in 2002. (Allotrope just means a chemical…
A: Lewis structure is a method of representing bonding pattern in a molecule. it tells how different…
Q: ہے نے
A:
Q: Solid CO₂ (0.43 mol) is reacted with CH3MgBr (0.22 mol) in ether solvent. The product of the…
A: Moles of CO2 = 0.43 mol. Moles of CH3MgBr = 0.22 mol. Moles of HCl = 0.13 mol. pH of the resulting…
Q: Examine the geometry of the molecule in the window. ball & stick + labels The molecule has a…
A: Aldehyde and ketone both are considered as carbonyl compounds. The functional group of aldehyde is…
Q: Write the molecular formula for the following compound. XX. He N 8.
A: The molecular formula is an expression that represents the number of atoms of each element in one…
Q: но но A OMe OMe OH Ph B и
A: In the given question 1 3 diol which is substrate (A) is reacted with the 1,1 dimethoxybenzyle in…
Q: CN CN ⇒?
A:
Q: 10) How would you use a Grignard reaction on an aldehyde or ketone to synthesize the following…
A: Grignard reagent RMgX can reacts with aldehydes and ketones and gives corresponding alcohol as…
Q: OsO4 (cat) NMO OH OH Select to Draw > mCPBA O J Select to Edit
A: Reaction of alkene with OsO4 gives syn-1,2-diol. Reaction of alkene with mCPBA gives epoxide.
Q: Identify the products formed in this Brønsted-Lowry reaction. HSO4 + NO₂ acid + base acid: base:
A: According to Bronsted and Lowry concept an Acid is a substance that has the tendency to donate a…
Q: in acidic solution, sodium oxalate, Na₂C₂O4, is oxidized to CO₂ by MnO4. In the process, MnO, is…
A: Given that In acidic solution, sodium oxalate is oxidized to CO2 by MnO4- In the processMnO4- is…
Q: Predict the major product(s) of the following reaction: Br Br Di Br₂ hv ? (only this enantiomer) Br.…
A: This is an example of benzylic bromination involving radical mechanism
Q: (b) Using the equation below, explain what happens to maintain the pH of the solution: HA (aq) (i)…
A: A buffer is a system in which there is not much change in pH of system on adding slight amount of…
Q: The rate of decay of a radioactive substance is proportional to the amount of the substance present.…
A: Given that The rate of decay of a radioactive substance is proportional to the amount of the…
Q: In the reaction shown below, the reactant molecule is O O OH oxidized and is therefore the oxidizing…
A: •Oxidising agent which is reduced itself and oxidise other compounds.
Q: 8 (a) Define in words the following: (i) Ka (ii) p.Ka
A: Ka and Pka both are constants. Ka is called as a dissociation constant for acids. Pka is simply the…
Q: how can I make an aldehyde & the location circled after disconnecting? And prepare this for an…
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first question out…
Q: How many of the following processes are nonspontaneous? climbing stairs dissolving salt in water…
A: How many given processes are non-spontaneous? Non-spontaneous process is the one which doesn't…
Q: Plot a Graph and Determine the Rate: Make a graphical plot of reaction time (x-axis) and…
A: We have to plot the graph of absorbance vs time and determine the rate of the reaction.
Q: Choose the best reagents to complete the reaction shown below. 00 A B C D H3O+ NaOH NaHCO3 NaNHz
A: This reaction is an example of acidic hydrogen abstraction using appropriate base.
Q: Complete these Brønsted-Lowry reactions. reaction 1: HSO3 + H+ = reaction 2: HSO3 + OH =
A: According to Bronsted-Lowry concept, acid can be defined as the substance which can donate a proton…
Q: a) b) Rate of Reaction Concentration of Acid (mol dm³) Explain, using collision theory, why the…
A: The collision theory was developed to explain the rate constant of a reaction. It consider reaction…
Q: 100 80 60 40 20 0 m/tpt/pt/profe 25 50 75 m/z 100 125 150
A: The question is based on the concept of mass spectroscopy. we need to analyse the mass spectra and…
Q: H3CO Но NHCH3
A:
Q: Phosphoric acid, H3PO4 (aq), is used to make fertilizers and is present in cola drinks. Phosphoric…
A: Mass of phosphorus = 272 g Yield of 1st reaction =89.5% Yield of 2nd reaction= 96.8% 1.…
Q: Reaction mechanics and diagram 1. Consider these three reactions as the elementary steps in the…
A: Given the reaction proceeds via 3 elementary steps. So there will be 3 peaks in the potential…
Q: Calculate the equilibrium constant, K, for the reaction shown at 25 °C. Fe³+(aq) + B(s) + 6H₂O(1)→…
A:
Q: n-a transition can be found in these compounds: Alcohols and Aromatic Hydrocarbons. Alcohols and…
A:
Q: To study the following reaction at 20 °C, NO(g) + NO₂(g) + H₂O(g) a mixture of NO(g), NO2(g), and…
A:
Q: Off Pd (ally 1)₂ Cl₂ (S)-BINAP лос. .OTIPS сами le AI Licl H₂O, DMSD 4 1) B₂-C1 2) ивицNF 3) MSCI 4)…
A: As per the rules, only the first question can be answered. Please resubmit for the solution of the…
Q: 2. You are given an electrochemical cell where the chemical reaction involves a silver complex, and…
A: First calculate the silver electrode potential using electrochemical potential. Then use same…
Q: For how many minutes must a current of 4.97 amp be provided to deliver 326 coulombs? Give the time…
A:
Q: For the reaction N₂(g) + 3 H₂(g)⇌ 2 NH₃(g) Kc = 0.0600 at a certain temperature. In an equilibrium…
A: Kc is the ratio of the equilibrium concentrations of product over equilibrium concentrations of…
Q: Which of the following increases moving down a column on the periodic table? ionic radius,…
A: Given properties, Ionic radius, electronegativity, atomic radius, ionization energy Which of the…
Q: Write the products formed in the following acid-base reaction. NaOH + HCl →> Then, describe the…
A: The hydrogen ion of acid reacts with the hydroxide ion of a base to form salt and water, the…
Q: You look at the solubility of Ca(OH)2 in water (Ksp = 4 x 10–10). You wait until the system reaches…
A: The solubility product of calcium hydroxide Ca(OH)2 is given to be 4 × 10-10. To a solution of…
Q: A sample occupies of oxygen, at 24°C, a volume of 4.18 x 102 milliliters (mL) at 1.32 atm pressure.…
A: Answer: According to Boyle's law, for any gaseous system, at constant temperature product of…
Q: Carbon-14 dating can be used to estimate the age of formerly living materials because the uptake of…
A: The half-life of a reaction is the time taken for the reactant to reduce to half its initial value…
Q: The voltage generated by the zinc concentration cell described by the line notation Zn(s) | Zn²+(aq,…
A: The question is based on the concept of electrochemistry. we need to calculate concentration of zinc…
Q: 10. For the following absorbance data, determine the concentration range that corresponds to the…
A: A question based on tools in analytical chemistry. A set of data is given that is to be converted…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

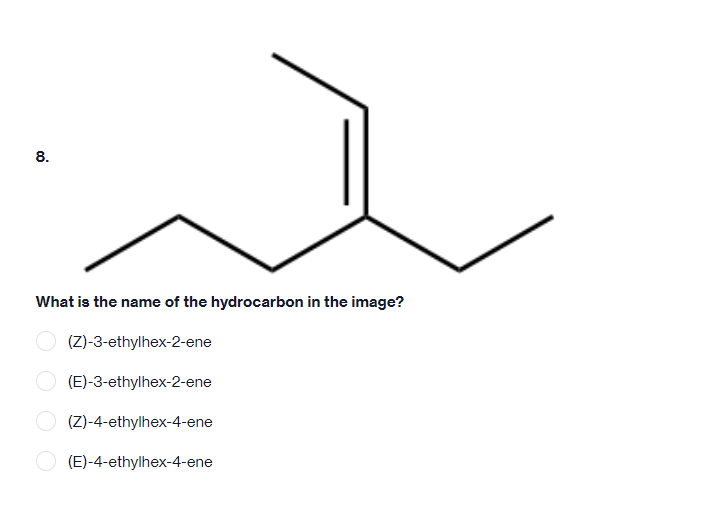
- Answer the following questions about compound A, which contains a CH3 group and OH group bonded to the carbon skeleton that consists of three six-membered rings in the conformation shown a.) Are the CH3 and OH groups oriented cis or trans to each other?b.) Is a substituent on Ca that is cis to the CH3 group located in the axial or equatorial position? c.) Is an equatorial Br at Cb oriented cis or trans to the OH group?d.) Is the H atom on Cc located cis or trans to the OH group?e.) Is a substituent on Cd that is trans to the OH group located in the axial or equatorial position?Answer the following questions about compound A, which contains a CH3 group and OH group bonded to the carbon skeleton that consists of three six-membered rings in the conformation shown.a.Are the CH3 and OH groups oriented cis or trans to each other? b. Is a substituent on Ca that is cis to the CH3 group located in the axial or equatorial position? c. Is an equatorial Br at Cb oriented cis or trans to the OH group? d. Is the H atom on Cc located cis or trans to the OH group? e.Is a substituent on Cd that is trans to the OH group located in the axial or equatorial position?6. Draw the correct structures for the following: a. what is the correct structure of 2-heptene? b. what is the correct structure of 4-octanone? c. what is the correct structure of dipropyl ether? d. what is the correct structure of 3-ethyl hexane? e. what is the correct structure of 2,2 dimethyl 1-hexanol?
- Sight along the C2-Cl bond of 2-methylpropane (isobutane). a. Draw a Newman projection of the most stable conformation. b. Draw a Newman projection of the least stable conformation. c. Make a graph of energy versus angle of rotation around the C2-Cl bond. d. Assign relative values to the maxima and minima in your graph, given that an H↔H eclipsing interaction costs 0 kJ/mol and an H↔CH3 eclipsing interaction costs 6.0 kJ/mol.A.) On any clean sheet of paper, draw both the condensed and line-bond structure of the following compounds: 7) 4-ethylcycloheptene 8) 4,4-dimethylpent-2-ene 9) 3-chloro-2,7-dimethylnon-4-yne (Cl- Chloro branch) 10) 1-cyclohexylbut-2-ynePlease name the following compounds. Make sure you spell each name correctly, leave no spaces since any variations in spelling will result in the loss of points. Example1: (1Z,3E,6Z)-1,6,8,8-tetramethylcyclonona-1,3,6-triene

