Q: 3. СНО + _(3a) + NH.OH → _(3b) +Agl+ NH&NOs + H2O
A: When benzaldehyde is treated with silver nitrate in ammonium hydroxide solution , which is also…
Q: By Volume, % By Mass, and Parts Per Million 6 By Volume 100% par (CH COOH HUUSolUion with a volume…
A: The concentration in terms of v/v is calculated by the formula: Concentration=Volume of soluteVolume…
Q: nčentratio a. 1.
A: To determine the molar concentration of OH- in water Given the molar concentration of H3O+in water…
Q: A. [FeBr(NH3)2(H2O)][Cu(CN)4] = %3D B. [PtBrCI(SCN)(H20)][AgCl2] = C. [Zn(H20)4]a[ZnCl(OH)2(S2O3)]2…
A:
Q: O Mg O Na O Pb O Al O Ag
A: Metals which have higher standard reduction potential than Hydrogen doesn't produce H2 gas when…
Q: 43. H Br "X :S: || 1. HNCNH, 2. NaOH, H₂O
A: Thiourea contains nucleophilic centre S which reacts with electrophilic bromine containing carbon…
Q: 4. HCHO + KMNO4 _(4a)_ → _(4b)_+ K2SO4 +| MnO2t + H2O
A: Solved in step 2. This is a redox reaction.
Q: 9. You may have noticed that water sometimes drips from the exhaust of a car as it is running. Is…
A:
Q: 8. Fe203 + CO -----> Fe + CO2
A:
Q: g) H + CH3OH
A: synthesis of ethers from haloalkanes
Q: What is the mass of sodium nitrate (NANO3) that will dissolve in 60 g of water at 12°C? O a. 55g O…
A: The solublity graph for NaNO3 is as below: This graph can be found from books.
Q: O موم H₂N 애 سلامة
A:
Q: I equiv. HBr +45°C
A:
Q: If ln k is plotted vs 1/T, what does the slope of the resulting straight line represent?
A: Hi,As it is not clearly mentioned which equation has to be considered, I am taking the basic…
Q: The Coाmroपकd एoiवssTप्ा तिप्रवेठनाव्ह, RoH TS Soपणह क Wव्नंेशर Tोनह कhट कल्े ठकाट Cqपवकांका णठ नैश…
A: ->KOH is strong base it completely dissociate into their ions when dissolved in water .Hence in…
Q: 1. SOCI2 2. Et,CuLi 3. (a) LİAIH4 (b) H2O OH
A:
Q: Major Entities present in Cu(s) a) Cu(s), H2O(l) b) Cu2+(aq), H2O(l)
A: To answer: Major Entities present in Cu(s) a) Cu (s), H2O (l) b) Cu2+ (aq), H2O (l)
Q: In the reaction Conc.H,SO, OH → P (Major) Pis
A: In reactions, rearrangement takes place to form more stable cation or to expand the ring.
Q: Substance O2(g) PC1(e) POC13(g) AH (KJ/mol) S (J/mol.K) 205.5 273.1 325.5 0 -319.7 -558.5 25 =:…
A:
Q: Br HBr OH
A:
Q: is Silver Nitrate ( AgNO3) a mixture or a compound?? why??
A: All matter can be classified as solid, liquid and gases mainly. In these physical states of matter,…
Q: 2- Calculate the AH of NaCl when it is heated from 0.0°C to 100.0°C.
A: Given that : Initial temperature = 0.0°C Initial temperature = 100.0°C The heat capacity of NaCl is…
Q: Br. "CEN H
A: Organic reactions are those in which organic reactant react to form organic products.
Q: How do I convert 3485.5 K cal to Joules?
A: The conversion of 3485.5kcal to calories is as follows:
Q: Determine el nombre IUPAC para los siguientes compuestos: Br HO NH2 CH3 CI Br OH
A: The IUPAC name of the following compounds are:
Q: complete the Na OEt HO 13
A: Saytzeff rule states that if the halogen is present on any carbon atom within the chain, the alkyl…
Q: pl•K) AGO
A:
Q: Br H2O lo prodect mayor HO Br or OH AO OH
A: Br2 and H2O add -Br and -OH group respectively to the double bond. The addition follows the…
Q: g) CIF,+ j) BF,- enoi no oluelon ad to dosS Tul clul to wb a AV ot ensoy beoler a h) SO2 k) i) SiO,…
A: Lewis structure represent electronic combination or electronic bonding.
Q: 1) Fuming H,SO, 2) HNO, H,SO, 3) Dilute H,S0. AICI, 1) HNO,, H,SO, Fe, H,O 3) NaOH 1) Fe. H,0 „NH2…
A: Nitrating mixture : In organic chemistry, the mixture of nitric acid (Nitrating agent) and sulfuric…
Q: B) to 1. Hg(OAc), CH,CH,OH 2. NaBH. OH O Bu,N'F H20 TIPSCI EtN: D) S. Br
A: The reaction are as follows:
Q: H,0 NH „NH co, H,SO, (cat)
A: ->H2O can acts as nucleophile because it has loan pair.
Q: HO HO, + 4 HI Но. OH |4 CH,ОН, Н*
A:
Q: Br Br AICh CI
A:
Q: Br OH OH Br OH OH Brlm
A: Chiral organic molecules are those that have no plane and axis of symmetry and the molecule must…
Q: 7 whichtof the porlowing eompounds pusbegse a goi omiao (circle all possibie anowers) ?
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 3.06 kPa Ov OP On OT
A: The correct answer is explained below.
Q: H20+Agl → - Туре: HNO3 + Fe(OH)3 → Туре: LiBr + Co(SO3)2 > Туре:
A:
Q: In a pure sample of blue vitriol the Copper content is 25.45%. Calculate the new percentage of…
A: The number of moles of a substance is given by the mass of the substance upon its molar mass. The…
Q: Br. Br Br2 H2O CoH,OBr
A:
Q: 1. LİAIH,éter 2. H20 OCH3 d но HO. b OCH3 HO HO. C H a но H.
A:
Q: 9) H,C. 1. LIAIH 2. H3O* h) 1) CHsMgBrléter anh. 2) H3O* i) H3O /calor
A:
Q: 8. Nickelic Silicate + Stannic Borate ---- ---> Stannic Silciate + Nickelic Borate Coeff. + + 9.…
A: The Chemical reaction follows the law of conservation of mass. According to the law of conservation…
Q: nple of eatifor prerution of aLn de ag
A: When the carbonate salts of metals are reacted with HCl, then corresponding chlorides are formed…
Q: Number these in erder Of ease of hude |= eosiest to huylrokze,4= handst to hujdrolye. SH iV GHs C=N
A:
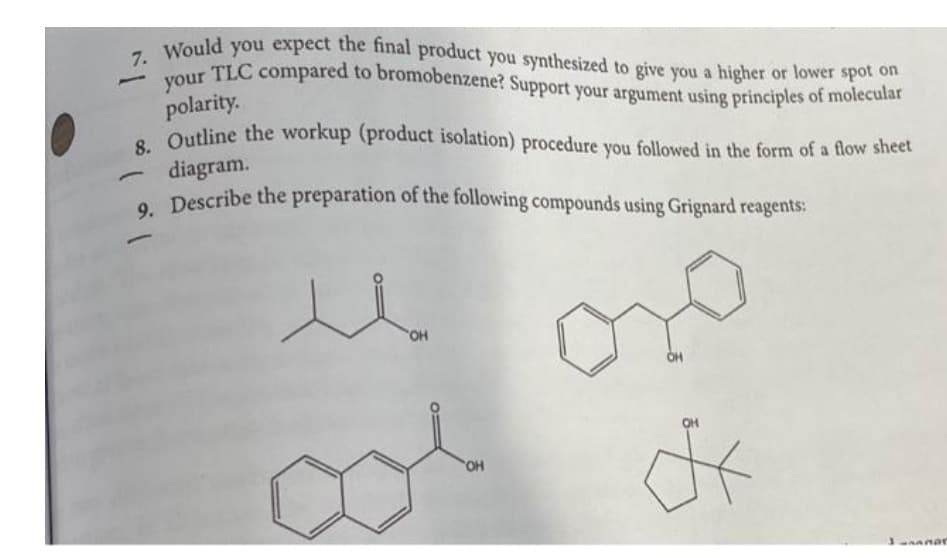
Q: Prepare: No2 @ prepare: prepare:
A: In these questions we will prepared all the products from benzene as a starting material. You can…
Q: What is the mass of sodium nitrate (NaNO3) that will dissolve in 120 g of water at 12°C? O a. 96g O…
A: The amount of solute that can be dissolved in 100 g of water is known as the solubility of a solute.…
Q: A. H,SO.lae) + H,0 O, → HSOiaq) + H,O (ag) 4(aq) B. CH,CO0 en) + H,0, CH,COOH, + OHe (aq) (aq) C.…
A: A Bronsted- Lowry base is that species in the reaction, that accepts a proton from the donor…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

- After the reduction of camphor with NaBH4 experiment, you took a 1H-NMR of your product sample. The sample ave the following 1H-NMR singlas with the given integrations (2.314 and 9.497). Determine the product ratio of isoboreneol to borneol in this particular mixture.In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…
- In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…1. Discuss three methods that can be utilised to synthesise n – heptane in the laboratory 2. Outline the processes you would adopt to purity and test the purity of sodium benzoate, an active ingredient of most cosmetic products in the laboratory 3. The reaction of (Z)-3-Methyl-3-hexene with H2O/H2SO4 produces 3 – Methyl-3-hexanol (75 % yield)(a) Write a balanced chemical equation for the reaction (b) Propose a mechanistic explanation for the reaction1. Compare the IR spectra of the cyclohexanone and cyclohexanol – what are thedifferences? Provide an interpretation.2. Why is NaCl added to the aqueous phase when the product is extracted withether during the reduction of cyclohexanone with sodium borohydride?3. How would you modify the reaction conditions to effect the reduction of cyclohexanone with sodium borohydrideusing lithium aluminium hydride as the reducing agent?
- A Grignard reaction of phenylmagnesium bromide with 3-pentanone gives 3-phenylpentan-3-ol as the major product. The crude product contains the 3-phenylpentan-3-ol product, unreacted 3-pentanone, and biphenyl (a side product). A developing solvent is found that separates the mixture into three spots on a silica gel TLC plate. Considering the functional groups present, predict which compounds would have the smallest and largest Rf values. Briefly justify your answer.Electrophilic bromination of an equimolar mixture of methylbenzene (toluene) and (trifluoromethyl)benzene with one equivalent of bromine (in the presence of FeBr3) gives exclusively 1-bromo-2-methylbenzene and 1-bromo-4-methylbenzene. 1. Explain why none of the (trifluoromethyl)benzene reacts 2. Explain the regiochemical outcome of the reaction In other words, why are 1-bromo-2-methylbenzene and 1-bromo-4-methylbenzene formed from the toluene?Is it possible to distinguish between the following compounds using the spectroscopic techniques covered in CHE331? If so, discuss how. If not, discuss why.Benzonitrile vs Benzaldehyde(a) UV spectroscopy(b) IR spectroscopy(c) Mass spectrometry(d) 13C NMR spectroscopy(e) 1H NMR spectroscopy
- What is the purpose for all of the following ? Carry out an electrophilic aromatic substitution reaction Prepare methyl m-nitrobenzoate by the nitration of methyl benzoate Verify the identity of a product through infrared spectroscopyI need help to find the mechansim for Preparation the derivative of Cyclohexanone when 2,4-DNPH reacted with cyclohexanone and explain the mechanisms step by step so I can understand it pleaseAn organic lab student carried out the reaction of methylmagnesium iodide with acetone(CH3COCH3), followed by hydrolysis. During the distillation to isolate the product, she forgot to mark the vials she usedto collect the fractions. She turned in a product of formula C4H10O that boiled at 35 °C. The IR spectrum showed onlya weak O¬H stretch around 3300 cm-1, and the mass spectrum showed a base peak at m>z 59. The NMR spectrumshowed a quartet (J = 7 Hz) of area 2 at d 3.5 and a triplet (J = 7 Hz) of area 3 at d 1.3. Propose a structure for thisproduct, explain how it corresponds to the observed spectra, and suggest how the student isolated this compound.

