Q: Examine the cell below and answer the questions that follow: a) What type of cell is this?…
A: Galvanic cell Electrolytic cell Converts chemical energy to electrical energy. Converts electrical…
Q: Starting with 3,5-hexadiyn-1-ol, select reagents from the table below that should be used to…
A: NaNH2/NH3 ====> Acidic hydrogen abstraction H2/Pt ===> Reduction
Q: What is the advantage of using automated peptide synthesis? A) No side products are formed. B) Ease…
A: Automated peptide synthesis is a fast and easy to synthesize many peptides simultaneuosly. It…
Q: Upon treatment with Tollen's reagent, you would expect one of these compounds to be unreactive. OH…
A:
Q: B) Write the most stable enol forms of the following compound, and choose which of them is more stal…
A: In this question, we will write down the most stable enol form for the given compound and also we…
Q: How to draw product C? Please give detailed structure for the product C
A: here we are required to predict the product of the reaction
Q: Ksp = 1.50 x 10-16 @ 25°C X = 1.22 x 10-8 What is the concentration of l- ions in the saturated…
A:
Q: A sealed container contains 4.0 mols of O2 (g), 6.0 mols of NH3(g) and 8.0 mols of Ne(g). The total…
A:
Q: The equilibrium constant for the reaction below is 5.90 x 10-5. 2 NO(g) + O₂(g) 2 NO₂(g) What can be…
A:
Q: The following reaction is a redox reaction: CuSO4 (aq) + Ni(s) --> NiSO4 (aq) + Cu(s) a) Write the…
A:
Q: The important steps in solid phase peptide synthesis are? A) Coupling and hydrolysis B) Hydrolysis…
A: Solid-phase peptide synthesis (SPPS) involves the successive addition of protected amino acid and…
Q: For the following reaction, 3.56 grams of nitrogen gas are mixed with excess hydrogen gas. The…
A:
Q: which involve large number of small molecules callec 9. monomers to from molecule high molecule…
A: Here from the given reactions we have to choose the correct one, which involves to formation of…
Q: Calculate the quantity of heat in KJ, associated with a 33.5 celcius decrease in temperature in a…
A: 5.85 Kg of aluminum bar is associated with a 33.5 °C decrease in temperature. The specific heat of…
Q: Balance the following reaction in BASIC conditions. Use any method of your choice. * Show all your…
A: Redox reaction is the reaction in which both oxidation and reduction occur together.
Q: What is the product of the reaction below? Xo ค้ำชู LOH + назад DCC
A:
Q: QUESTION 11 Using the following table to calculate the energy difference between the axial and…
A: The general chair conformation of cyclohexane is given by When all the substituents are hydrogen…
Q: what compound has the highest ∆fusionH AlCl3, NaCl , FeCl2
A:
Q: Q14- A 0.100 M solution of the weak acid HA has a pH of 2.36. Calculate pK, for HA. 2.36 C) 1.66 B)…
A: Given: The pH of the weak acid, Ha = 2.36 The concentration of HA = 0.100 M We have to calculate the…
Q: 44 - CC14 is. A - Polar Not polar 45-CH3CI is OA -Polar OB - Not polar k
A: Non- Polar molecule has net dipole moment zero while polar molecules has net dipole moment non zero.…
Q: Compound HO HO 00 OH A is an example of reducing sugar. НО HO он ""OH HƠ B HO" "ОН OH C НО HƠ D -OH…
A: ->Compound which contains Hemiacetal group that is Known as reducing sugar . ->Hemiacetal…
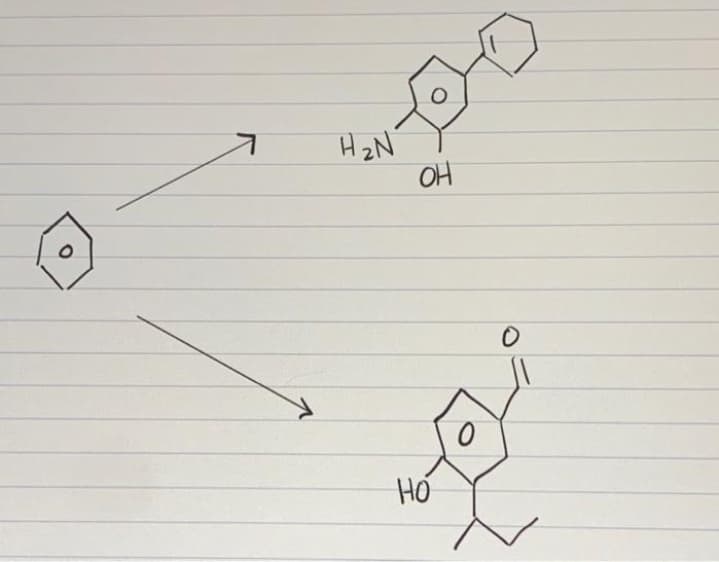
Q: Describe how you would prepare the following. Name reactants and products and rx conditions. b) 1,…
A:
Q: When 66.875 is added to 18.16, the result should be reported with When 88.94 is subtracted from…
A:
Q: Which pair of reactants is most likely to undergo an E1 reaction? O CH3SNa + (CH3)2CHCH₂CH₂CH₂Br at…
A: E1 (unimolecular elimination) is a reaction where there is a loss of the leaving group followed by…
Q: نقطة واحدة نقطة واحدة نقطة واحدة १:.१ Most monomers are organic .12 materials, atoms are joined in…
A: Given in following question some polymer releated subjective question choose a correct answer
Q: Method is preparing compounds with double and triple bonds is. A. Elimination reaction. B.…
A:
Q: 23 - General formula of alkene compounds are. OA - CnH2n+2. O B - CnH2n-2. O C - CnH2n.
A: Alkenes are those compounds which has a double bond in it.
Q: Mekdon-540 is a new radioactive substance discovered by scientists. They would like to find the rate…
A:
Q: In the Inorganic laboratory, you made a dark red complex ion when you mixed potassium thiocyanate…
A:
Q: Consider the following reaction: 3Cu + 2HNO3 + 6HCl → 3CuCl₂ + 2NO+ 4H₂O Which of the following is…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: At 50°C, the solubility of KCL in water is 42.6 g in 100 g water. What is the molality of KCL in…
A: Here we are required to find the molality of KCl solution
Q: Which is the correct formation reaction for KCI? OK(s) + 12 Cl₂(s) ----> KCI(s) OK(aq) + 2Cl₂(g)…
A: A question based on stoichiometry that is to be accomplished.
Q: ............33 .reshaped is A. Thermoset. O B. Thermoplastic. O Cannot be…
A:
Q: 1. 5% (w/v) Sodium hydroxide solution a. molality: _______________ b. molarity: _______________…
A: Here density of the solution is not mentioned in both the cases so assume the density to be same as…
Q: 22 General formula of aromatic compounds are. O A - CnH2n. O B - CnHn+2. O C - CnH2n-2.
A:
Q: 5. Identify the molecule that corresponds to the IR spectra provided below. Indicate the significant…
A:
Q: Write the formula and Identify the factors of the following: 1. Aluminum hydroxide Formula:…
A: Salt is made from acid and base. Acid is defined as substance which release hydrogen ions. Acid is…
Q: 2. Classify the nitrogens in the following compound as primary, secondary or tertiary. Explain your…
A: Explanation :
Q: 15. Network polymer structure is. O A. Branching. O B. Liner. C. Cross-link.
A: Network polymers are highly complex type of polymers with three dimensional linkage. This type of…
Q: Arrange the following in compounds in INCREASING reactivity to hydrolysis. Drag the structures to…
A: The molecules given in the options are acid derivatives with -Cl, -CH2CH3 , and -NH2 groups attached…
Q: Suppose you are measuring the mass of a solid sample on a balance using a weigh boat. You record the…
A: given, The mass of weigh boat = 3.451 g. Mass of weigh boat and sample =…
Q: A 267−mL benzene solution containing 2.27 g of an organic polymer has an osmotic pressure of 7.37 mm…
A: 267 mL of benzene solution contains 2.27 g of an organic polymer has an osmotic pressureof 7.37 mm…
Q: For the following reaction, 5.77 grams oxygen gas are mixed with excess carbon (graphite). The…
A:
Q: There are 12 inches in 1 foot and 3 feet in one yard. There are 1760 yards in 1 mile. a. How many…
A: To solve the given questions, use the dimensional analysis and conversion factor to calculate…
Q: Predict the products of the reaction below? CH₂OH =0 NaOH H -H H₂O H-OH CH₂OH HO HO
A:
Q: The correct answer seems to be 51 torr
A: Given that, a mixture of an equal mass of benzene (C6H6) and toluene (C7H8). The vapour pressure of…
Q: What would be the product if D-mannose is reacted with a.. 1.) NH2OH then 2.) (CH3CO)2O, NaOCOCH3…
A: D-mannose reaction are as follow.
Q: Calculate the pCa for the titration of 100 ml of 0.050 M Ca²+ at pH = 8 with 0.1 M EDTA After…
A: The balanced equation for the reaction of Ca2+(aq) and EDTA, Y4-(aq) is: Ca2+(aq) + Y4-(aq) ⇌…
Q: Give the order of reactivity of carboxylic acid and its derivatives. What is the significance of…
A: The reactivity of carboxylic acid derivatives is as follows: amide < carboxylic…
Q: Consider the structure of the TAG below and answer the questions that follow. Select your answer…
A: #1: The given molecule is a triacylglycerol (TAG) that is made by the dehydration reaction between…
H8.
reactants and name compounds
Step by step
Solved in 3 steps with 3 images


