Q: Indicate whether the salts below would form solutions that are acidic, basic, or neutral in pH. KCIO...
A:
Q: 702.0mc 3. a) How many mLs of 0.600 M calcium chloride are needed to provide 0.110 moles of chloride...
A: 3.a) We have to calculate the amount of 0.600 M of calcium chloride required to produce 0.110 moles ...
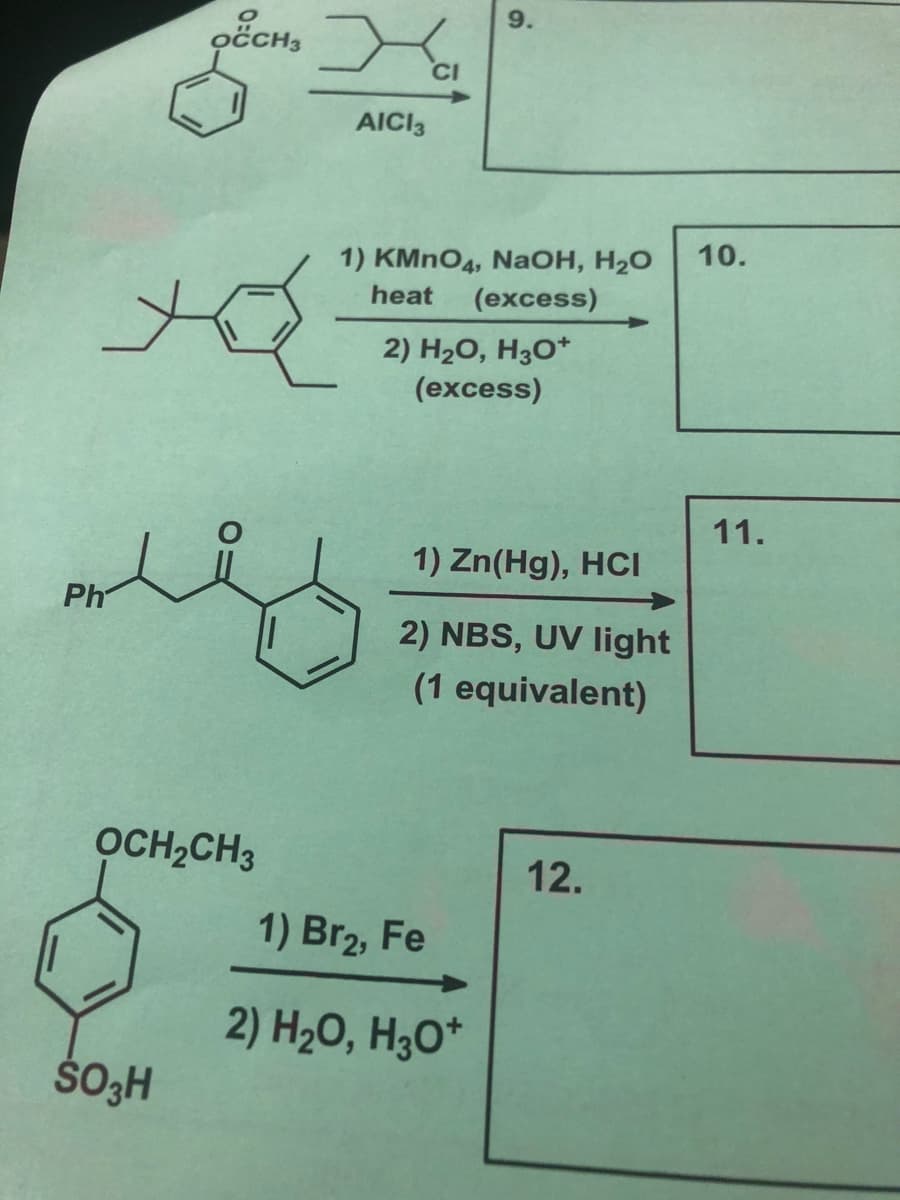
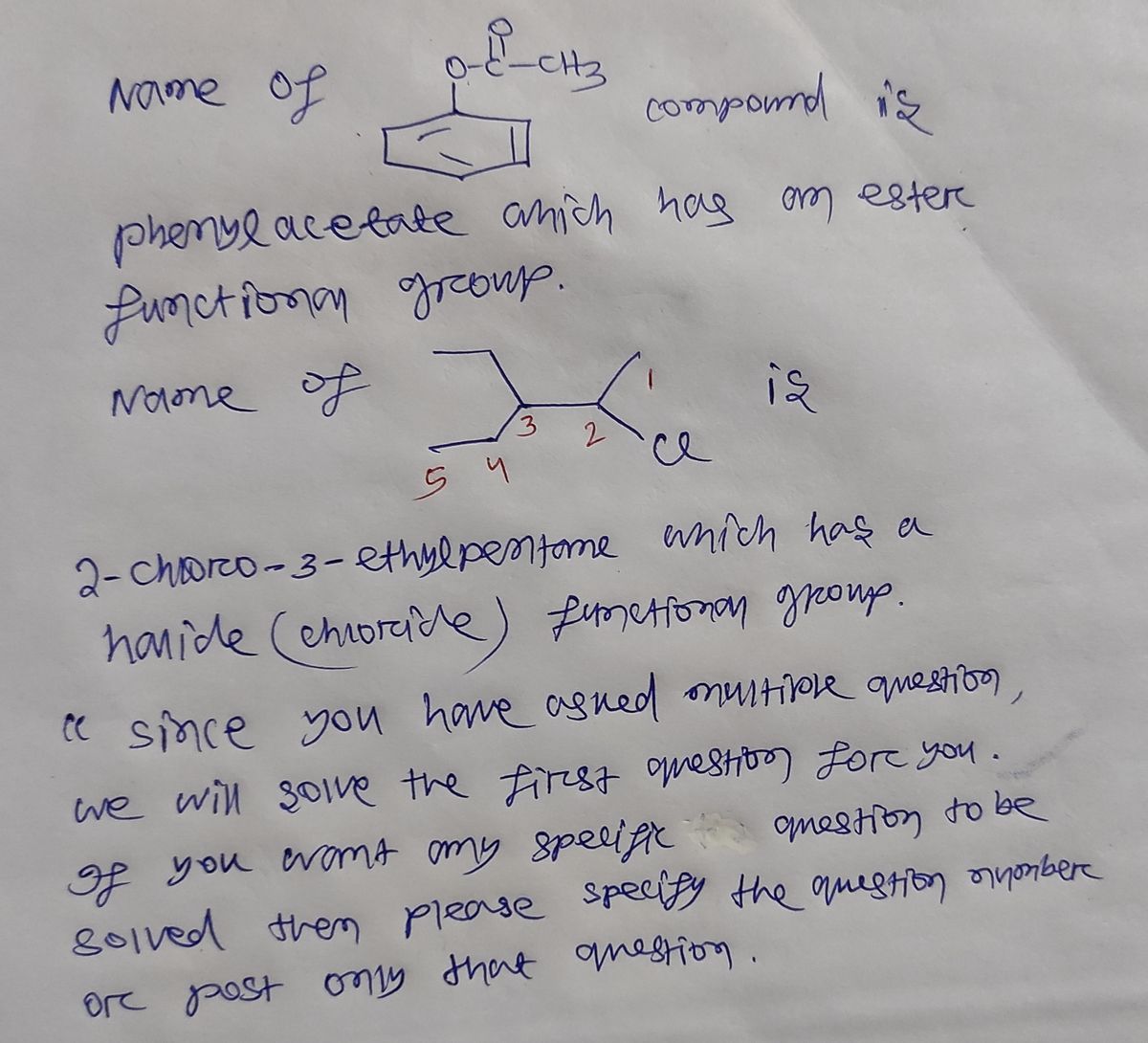
Q: what are the structures for the dollowing written out compltetly b) hexonoate reacts with methyl h...
A:
Q: milliliters
A:
Q: the rate constant for the first-order decomposition of N2O is 2.97 s^-1. what s the half-life of the...
A:
Q: What mass of CO is required to react with 25.13 g of F*e_{2} * O_{3} according to the equation : Fe ...
A:
Q: Part B: NH2
A: A question based on molecules that is to be accomplished.
Q: According to the Evan's pKa table, phenol has a pKa of 9.95. p-Nitro and m-nitrophenol have pKas of ...
A:
Q: large containers of sulfuric acid solution, 9. А chemist has two with different concentrations of ac...
A: Soln
Q: Solving an enthalpy of neutralization problem. (see Chemistry 2e, Example 5.5): when 47.35 g of wate...
A: Given: Mass of water = 47.35 g. Initial temperature of water = 22.6 oC And final temperature of wate...
Q: Calculate the pH of a 2.50x10-4M solution of hydroiodic acid, HI. pH =
A:
Q: A side reaction in the manufacture of rayon from wood pulp is 3CS2 + 6NAOH → 2NA2CS3 + Na2CO3 + 3H2O...
A: The balanced reaction given is, Given: Moles of NaOH taken = 2.78 mol. Volume of CS2 taken = 92.5 m...
Q: 3. What is the pH of a cleaning solution with a [H,O'] = 7.4 x 10 M H,O? ibility Good to go
A: Formula to calculate pH is :- pH = -log[H3O+]
Q: S, For ench renction. A7 write n word erntin B) Write he balmced olecuir equton C) write Hhe lonlmce...
A: 1) The reactants given are AgNO3 (aq) i.e. silver nitrate and NaOH (aq) i.e. sodium hydroxide.
Q: 6. A solution is made by mixing 100.0 mL of 0.200 M calcium chloride with 50.00 mL of 0.200 M sodium...
A: Soln
Q: After the reaction is complete, 6.22 g of nitrogen oxide are obtained. write the balanced equa...
A: Here 7.6 g NH3 is mixed with 10 g of O2 to produce 6.22 g of NO experimentally in Ostwald process. W...
Q: hy is bromination more selective than chlorination?
A: bromination more selective than chlorination because
Q: Determine whether each of the molecules is aromatic, nonaromatic, or antiaromatic.
A: Aromatic compounds are made up of conjugated planar ring systems with delocalized pi-electron clouds...
Q: milliliters
A:
Q: onvert each of the following pH values to a hydroxide ion concentration. y = 1.0E-14. 1.32 i M eText...
A: pH = -log [H+] [H+] x [OH-] = 1 x 10-14 As per our guidelines we can only solve first three sub-part...
Q: The equilibrium constant, Ke, for the following reaction is 1.80x104 at 298 K. NH4HS(s) =NH3(g) + H2...
A: Given, NH4HS(s) ⇌ NH3(g) + H2S(g) Temperature (T) = 298 K Equilibrium constant (KC) = 1.80 × 10-4 In...
Q: Why is the existence of a series of entering groups with different rate constants evidence for an as...
A: Introduction : Ligand are those which give their lone pair of electron to the central metal atom or...
Q: unit 3 Test - Energy in Which statement about the change in bond energy of this reaction is correct?...
A: Reaction :- CH4 + 4Cl2 ------> CCl4 + 4HCl
Q: Which is statement is true about the neutrons of atom?
A: (a) Option (a) is incorrect. Atomic mass equals the sum of neutrons and protons present in the n...
Q: What is the formula fortheedges, expressed as “f”, if the radius of the atom is expressed as “k”: a...
A: In general, edge length is represented by 'a' and radius is represented by 'r' Here , radius is den...
Q: Which species is the conjugate base of HPO4 2-? Write the formula net charge
A: A base is any substance that can accept proton from any other substances or substance that ionize in...
Q: 10. Which of the following describes a crystalline solid? a. Crystalline solids have particles that ...
A: There are three phases of matter. The phase of matter can be interchanged by changing the temperatur...
Q: A mixture of 0.02993 mol of O, 0.02165 mal of NOCI, 0.02691 mol of NO, and 0.01037 mol of C is place...
A: Answer: First of all, with the help of values of initial moles of gases and equilibrium moles of chl...
Q: What is a mixture is a combination of
A: To explain: A mixture is a combination of what?
Q: The chemical reaction shown was performed and the concentration of CCl4CCl4 was measured over time. ...
A:
Q: Determine if the statements are true. The larger the surface area, the higher the dissolution. ...
A: True or false: As per our guideline we have to answer first three questions only:
Q: a. Mg I Mgaa) l Cuag) I Cus) b. Nice I Niaa) | Agiaa) I Ag) 3. For the cell notations in the previou...
A: Here from the given Galvanic cell notation we have to write the cell reaction. For this we have to w...
Q: Fluoride ion is added to drinking water to prevent tooth decay. What is the mass in grams of sodium ...
A: Given Formula units = 1.19 × 1019
Q: c. 20.45 g Al(NOsla 5. A saline solution with a mass of 233 g has 34.5 g NaCl dissolved in it. What ...
A:
Q: (PP-1 and PP-2). PP-1 has Mw of 100,000 g/mol and PP-2 has Mw 500,000 g/mol, If the PDI (Molecular w...
A: The correct option is:
Q: Calculate the pH of a solution that is 0.0400 M in a) NaH,PO. b) NaHC:O. c) NaHS
A: Soln
Q: Why do meltiing, freezing and boiling point varies and depends on a kind of matter? Not less than 6 ...
A: Matter can be defined as substance that possess definite mass and volume. Matter can be solid, liqui...
Q: No of D isomers are possible for hexose Select one: a. 8 b. 4 c. 6 d. 16
A:
Q: 3) Design a reasonable synthesis for the following transformation. = trort + B.
A: Here we have synthesize the given polymer which is a polyester obtained from starting material 1,4-d...
Q: According to the following reaction, how many grams of calcium oxide will be formed upon the complet...
A:
Q: KHP
A:
Q: identify each substance Acid Base conjugate acid Conjugate base
A:
Q: Differentiate intensive to extensive properties. Not less than 6 sentences.
A:
Q: 4. [4] Give a valid set of four quantum numbers for each electron in the valence sh ell of a ground ...
A: There are four set of quantum numbers which describes or gives all the information about an electron...
Q: Element X has two isotopes. If 72.0% of the element has an atomic mass of 84.9 atomic mass units, an...
A: Given : We have to calculate the average atomic mass.
Q: Provide the missing reagents required for the following transformation: OH OH
A: Alkene reacts with peracid, RCO3H (such as metachloroperbenzoic acid, mCPBA) to form an epoxide. The...
Q: . Which graph correctly displays the relationship between wavelength, in meters, and frequency, in h...
A: To explain: The graphical relationship between the wavelength in m and frequency in the hertz of the...
Q: 3. Consider Lewis structure for hydrogen fluoride (HF). a. If hydrogen fluoride were to behave as an...
A: An acid donates proton while a base accepts proton. HA---> H+ + A- HA is acid while A- is con...
Q: a) What is the advantage of very small particle sizes in LSC columns? (b) With reference to the Va...
A: a) Small particle sizes are used in the LSC columns.
Q: Given the reaction: CuSO4.3H2O CuSO4.2H2O + H2O the DELTA H° = 9900 and DELTA G°=4500 at 25°C. Cal...
A: The given reaction is : CuSO4·3H2O(s) ↔ CuSO4.2H2O(s) + H2O(g) ∆H° = 9900 J/mol and ∆G°=4500 J/m...
Step by step
Solved in 3 steps with 3 images

- 6. A sample of an alum was analysed by atomic spectroscopy and gravimetrically to determine its structure. A 0.1000 g of the sample when treated with BaCl₂ produced 0.0984 g of a white solid after drying to constant weight. Another 0.1000 g sample of the alum when heated at 105°C for several hours produced a weight loss of 0.04558 g while the % of K in the sample was determined by atomic spectroscopy to be 8.240%. Use these results to work out a formula for the alum. [M,/ g mole¹: K 39.10; Al 26.98; BaSO, 233.39; SO² 96.06; H₂O 18.0148]Mesityl oxide( (CH3)2C=CHCOCH3) shows two bands at 230 nm (12600) and 327 nm (98). But its isomer, ( CH2=C(CH)3-CH2COCH3) does not show any bands above 210 nm. Explain.) Calculate the ΔG°rxn using the following information. 4 HNO3(g) + 5 N2H4(l) → 7 N2(g) + 12 H2O(l) ΔG°rxn = ? ΔG°f (kJ/mol) -73.5 149.3 -237.1
- Noncontact AFM images for the HBC, or Hexa-peri-hexabenzocoronene molecule. (a) Stick model. (b) Constant-height AFM measurements for an HBC molecule on a Cu (111) surface substrate. (c) A pseudo-three-dimensional representation that highlights the local maxima. (d) Calculated electron density above the molecular plane. From Gross et al. [15 Links to an external site.]. Based on the AFM image shown in (b), what kind of substance is Hexa-peri-hexabenzocoronene? There may be more than one correct answer.As part of an investigation on Pb in biological ecosystems, some grasses that grew along the road and were exposed to emissions from motor gasoline were collected. 6.2501 g of grass were calcined to destroy all organic matter, the inorganic residue was properly treated and finally made up to 100 mL. An aliquot of 50 mL of this solution was read in an atomic absorption spectrophotometer registering an absorbance of 0.1250; then 0.10 mL of a standard solution of 55 µg of Pb / mL were added to the other 50 mL, with this fortified sample, another absorbance measurement was made, which was 0.1798. Calculate the µg of Pb / g of grassThe maximum safe [Ba2+]=1.5x10-5M. Maintaining 1.0x10-3M levels for each of the anions below would ensure that this level is achieved except (Ksp of BaF2=1.7x10-6, BaCO3=5.9x10-9, BaC2O4=7.4x10-14, and BaSO4=1.1x10-10) A SO42- B C2O42+ C CO32- D F-
- Please help me fill the remaining spaces. The spaces marked with NA don't need to be calculate. Name Molecular Weight Amounts used mmol Equivalents M.P or BP (C) Density (g/mL) Indene 116.16 .5mL MP=-2 BP=181-182 .996 Borane-tetrahydrofuran Complex 85.94 2.8mL 1 MP= -108 BP=66 NA 30% Hydrogen Peroxide 34.01 1mL MP= -33 BP=108 NA 3 M NaOH(aq) 40 .7mL MP=318 BP=1390 NA Diethyl Ether 74.12 NA NA NA BP=75-78 .902 Hexanes 86.17 NA NA NA BP=65.5-68.3 .659 Potassium Permanganate 158.03 NA NA NA BP=100 1.0 1-Indanol (Product) 134.13 NA MP=50-54 NA 2-Indanol (Product) Same as Above Same as Above Same as Above NA MP=68-71 NAWeight of crude/pure product: 0.819/0.047g Percent yield: Theoretical Yield: Melting Point crude/pure:195/190 Q: What would the theoretical yield froms the Diels-Alder Adduct with your product. Also, what is the actual yield? How do i calculate those? i used anthracene 200mg, maleic anhydride 110 mg and 6ml of xyleneINORGANIC Chemistry Starting product was [(Me5dien)CoCl2] and reacted with NaSCN to produce [(Me5dien)Co(SCN)2] Using the graphs attached, the IR data tells you that the binding mode is N coordination. How does the IR data agree (or disagree) with the peak shifts (Uv-Vis spect) IR Product 1, [(Me5dien)CoCl2] 29041.04, 2812.76, 2761.62 cm-1 (carbon bond) 1457.35 cm-1 (CH2) 1263.91 cm-1 (possibly C-N stretch) Product 2, [(Me5dien)Co(SCN)2] 2968.54-2870.67 cm-1 (C-C) 2059.41 cm-1 (N=C=S) 1470.28 cm-1 (CH2) Uv-vis [(Me5dien)CoCl2] Peak A: 531.8 nm, 0.722 Abs Peak B: 609.7 nm, 0.666 Abs [(Me5dien)Co(SCN)2] Peak A: 510.20 nm, 0.357 Abs Peak B: 608.20 nm, 0.401 Abs
- The two square-planar isomers of [PtBrCl(PR3)2] (where PR3 is a trialkylphosphene) have different 31P-NMR spectra as shown below. For this exercise, we ignore the coupling to 195Pt (I=1/2 at 33% abundance). One isomer (A) shows a single 31P resonance; the other (B) shows two 31P resonances, each of which is split into a doublet by the second 31P nucleus. Which isomer is cis and which is trans? Draw the structures of the two isomers.Explain in detail Remote Spectroscopy via NIRWhat is the molecular mass observed for Man9(GlcNAc)2 if it has a mass spectrometry ionization that imparts a +3 charge state? .







