A 12.49 g sample of NaBr contains 22.34% Na by mass. Considering the law of constant composition (definite proportions), how many grams of sodium does a 8.61 g sample of sodium bromide contain? On another planet, the isotopes of titanium have the given natural abundances. Abundance Mass (u) 74.900% 45.95263 18.800% 47.94795 6.300% 49.94479 Isotope 4611 g Na 4871 50Ti What is the average atomic mass of titanium on that planet? average atomic mass
A 12.49 g sample of NaBr contains 22.34% Na by mass. Considering the law of constant composition (definite proportions), how many grams of sodium does a 8.61 g sample of sodium bromide contain? On another planet, the isotopes of titanium have the given natural abundances. Abundance Mass (u) 74.900% 45.95263 18.800% 47.94795 6.300% 49.94479 Isotope 4611 g Na 4871 50Ti What is the average atomic mass of titanium on that planet? average atomic mass
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter4: Reactions In Aqueous Solution
Section: Chapter Questions
Problem 73QAP: Copper metal can reduce silver ions to metallic silver. The copper is oxidized to copper ions...
Related questions
Question
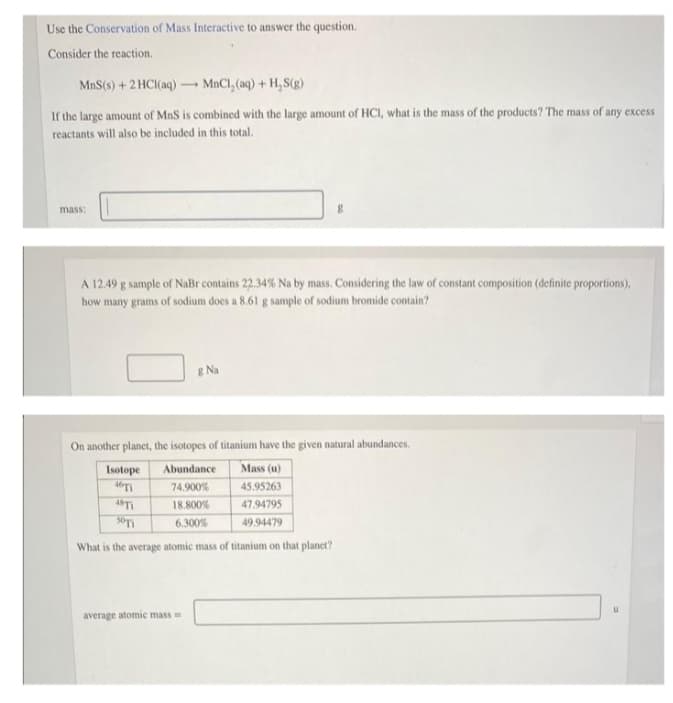
Transcribed Image Text:Use the Conservation of Mass Interactive to answer the question.
Consider the reaction.
MnS (s) + 2HCl(aq) →→→MnCl₂ (aq) + H₂S(g)
If the large amount of MnS is combined with the large amount of HCI, what is the mass of the products? The mass of any excess
reactants will also be included in this total.
mass:
A 12.49 g sample of NaBr contains 22.34% Na by mass. Considering the law of constant composition (definite proportions),
how many grams of sodium does a 8.61 g sample of sodium bromide contain?
g
& Na
average atomic mass=
On another planet, the isotopes of titanium have the given natural abundances.
Isotope
Abundance Mass (u)
46Ti
74.900%
45.95263
4871
18.800%
47.94795
50Ti
6.300%
49.94479
What is the average atomic mass of titanium on that planet?
U
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning