A 5.00 L tank at 15.3°C is filled with 6.86 g of dinitrogen monoxide gas, 12.5 g of sulfur hexafluoride gas, and 19.4 g chlorine pentafluoride gas. You can assume both gases behave as ideal gases under these conditions. Which of the following tables CORRECTLY presents the mole fraction of each gas in the tank? GAS MOLE FRACTION CIFS N20 SF6 0.149 0.156 0.0856 TABLE A GAS MOLE FRACTION CIF5 N20 SF6 0.381 0.399 0.219 TABLE B GAS MOLE FRACTION CIF5 N20 SF6 0.543 0.0648 0.393 TABLE C GAS MOLE FRACTION CIF5 N20 SF6 2530 302 1830 TABLE D
A 5.00 L tank at 15.3°C is filled with 6.86 g of dinitrogen monoxide gas, 12.5 g of sulfur hexafluoride gas, and 19.4 g chlorine pentafluoride gas. You can assume both gases behave as ideal gases under these conditions. Which of the following tables CORRECTLY presents the mole fraction of each gas in the tank? GAS MOLE FRACTION CIFS N20 SF6 0.149 0.156 0.0856 TABLE A GAS MOLE FRACTION CIF5 N20 SF6 0.381 0.399 0.219 TABLE B GAS MOLE FRACTION CIF5 N20 SF6 0.543 0.0648 0.393 TABLE C GAS MOLE FRACTION CIF5 N20 SF6 2530 302 1830 TABLE D
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter10: Gases And Their Properties
Section: Chapter Questions
Problem 75GQ: The density of air 20 km above Earths surface is 92 g/m3. The pressure of the atmosphere is 42 mm...
Related questions
Question
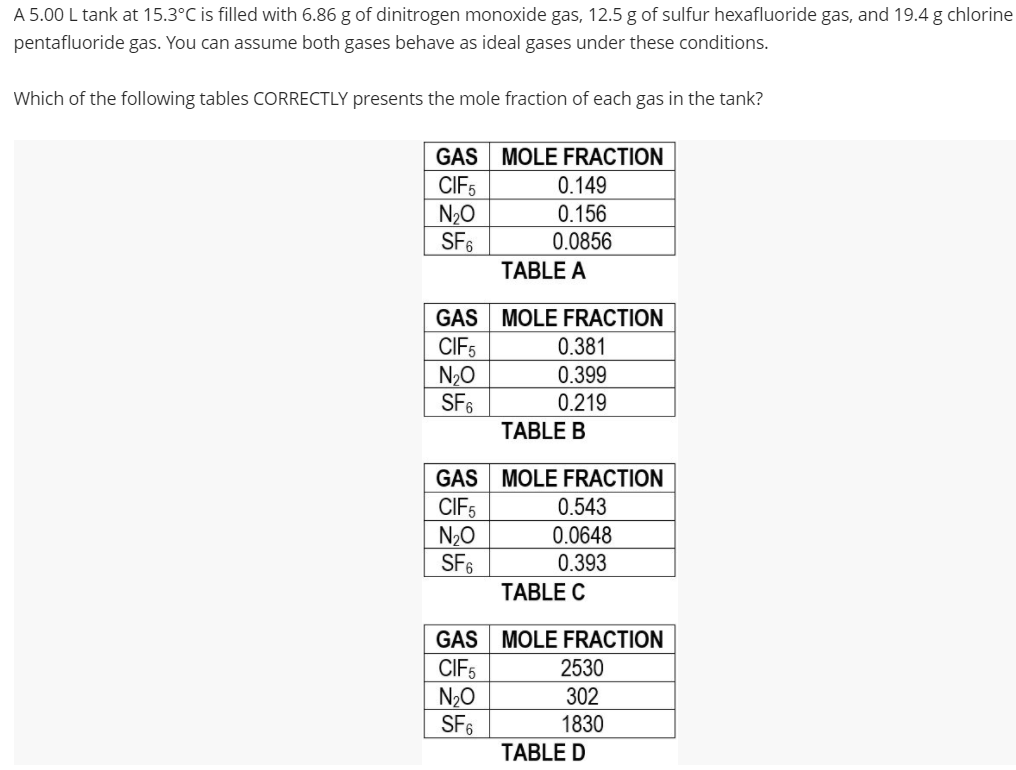
Transcribed Image Text:A 5.00 L tank at 15.3°C is filled with 6.86 g of dinitrogen monoxide gas, 12.5 g of sulfur hexafluoride gas, and 19.4 g chlorine
pentafluoride gas. You can assume both gases behave as ideal gases under these conditions.
Which of the following tables CORRECTLY presents the mole fraction of each gas in the tank?
GAS MOLE FRACTION
CIFS
N20
SF6
0.149
0.156
0.0856
TABLE A
GAS MOLE FRACTION
CIF5
N20
SF6
0.381
0.399
0.219
TABLE B
GAS MOLE FRACTION
CIF5
N20
SF6
0.543
0.0648
0.393
TABLE C
GAS MOLE FRACTION
CIF5
N20
SF6
2530
302
1830
TABLE D
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning