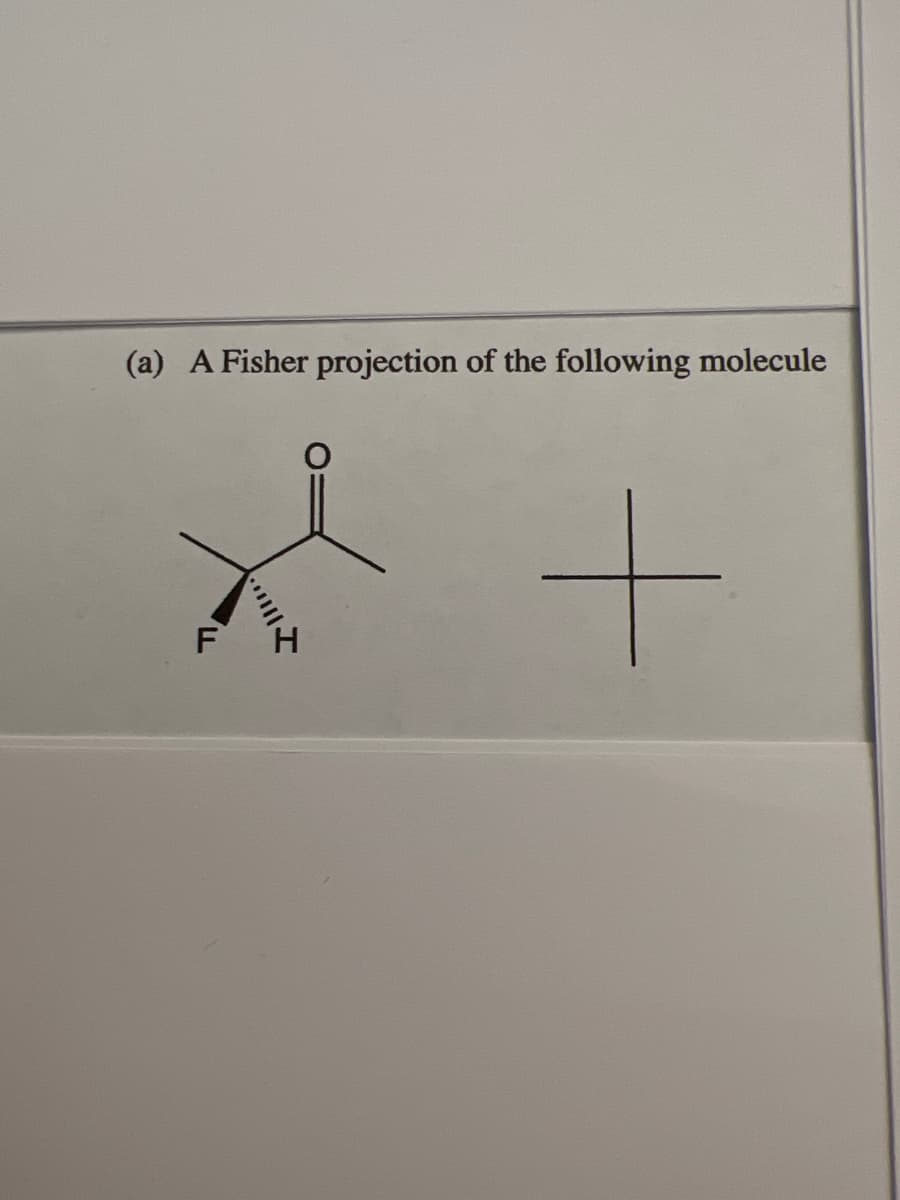
Q: Organic Chemistry presented by Macmillan Learning Draw the unique stereoisomers for…
A: Draw the structure of the given name ?
Q: [References] Identify the molecules or lons below as Lewis acids, Lewis bases, or neither. If there…
A: Here, we have to identify the given molecules as Lewis acids, Lewis bases, or neither.
Q: The molecular geometry of the CH2F2 molecule is tetrahedral, non polar seesaw, polar seesaw, non…
A:
Q: e the following redox reaction. What are the coeffici HNO 3(aq) → Zn 2+ (aq) + NO(g) O=4, NO = 2…
A: Reaction, Zn + HNO3 ----> Zn2+ + NO USing oxidation number method 3Zn + 2HNO3 +6H+ ---->…
Q: Calculate for the normality of H₂SO4 for the reaction H₂SO4 + 2 NaOH → Na2SO4 + 2 H₂O if the acid…
A: Initial volume of H2SO4 = 7.5 ml Initial Molarity of H2SO4 = 1.0 M After dilution total volume =…
Q: When 0.900 g of Mg was burned in oxygen in a constant volume calorimeter, 22.3 kJ of energy as heat…
A: Given: Mass of Mg = 0.900 g Heat evolved = 22.3 kJ Mass of water in container = 880. gm Initial…
Q: The phase diagram of a substance is given below. The region marked "x" represents: super critical…
A: Phase diagram gives the information regarding phase transition of any substance.
Q: What mass of KI is required to make 500 mL of a 2.80 M KI solution? Hint 2 901
A:
Q: What are the coefficients?
A: Recall the given reaction, HBr + SO42- → SO2 + Br2Here we have to…
Q: answers clearly. 1.1) From the concept of reversed SN2 Disconnection, draw the best set of positive…
A:
Q: The molecule below contains H H H O 7pi & 9 sigma 4 pi & 6 sigma O 7 pi & 6 sigma O 4 pi & 9 sigma…
A:
Q: Et3N ** Et₂O H Find out the product A, B and C. i. i. Me NaNH, NH3 NaNH, B ii. ii. Br H₂O (Weak…
A: Note - The given question is a multiple questions , hence I solved first question according to rules…
Q: H5106+ Cr→103 + Cr³+ Match the following for the above reaction. Drag and drop options on the…
A: H5IO6 + Cr -----> IO3- + Cr3+ -> The species for which oxidation number increases is oxidized.…
Q: Question 11 Two gas mixtures, A and B, are compared for their carbon dioxide content. Mixture A has…
A: Given : Composition of mixture A (on mole basis) Nitrogen = 50 % Oxygen = 25 % Carbon dioxide =…
Q: Provide the correct IUPAC name for the compound shown here. O
A: In the question, an organic compound is given and its nomenclature has been asked according to IUPAC…
Q: 1. CH3 H3C-C. CH3 :Br r-Br: CH3 H3C-C-Br: I CH3 Br:
A:
Q: 13. Determine the shape of the following molecules and identify what intermolecular forces exist in…
A: All the compounds are covalent compounds as they are formed of only nonmetals.
Q: Which of the following primary standards is best used for redox titrations? O Na2CO3 EDTA O AgNO3…
A: A good primary standard should contain, high level of purity, low reactivity, high equivalent…
Q: The empirical formula for trichloroisocyanuric acid, the active ingredient in many household…
A: Empirical formula : Simplest whole number ratio of a compound is called empirical formula…
Q: Based on the following information, which compound has the strongest intermolecular forces?…
A: The intermolecular force present in the molecule is directly proportional to the bond strength of…
Q: How would this chemical react with (CH2=CH)2CuLi?
A: Organic reactions are those in which organic reactant react to form organic products.
Q: a) CH3 -CH-CH-COOH + Ca → CH, NH, b) CH₂ - CH-CH-COOH + CH₂OH- CH, NH, c) CH3 -CH-CH-COOH + Ba(OH)₂…
A:
Q: Which of (a)-(d) shows the increasing order of basicity of compounds 1-4? 1 NH₂ O a. 4 <3 <2<1 O b.…
A: Basicity is tendency to accept H+ or we can say availability of lone pair for H+ . Basicity is…
Q: ब) 44307 DBH3 • THF 27 4202, NaoH t Br, H2O compuede the following rrection
A:
Q: HO ·||| I OH a. 2S,4S O b. 2R,4S C. 2S,4R O d. 2R, 4R
A:
Q: 2.1) Draw the structure of "major organic final product" of the following Addition of H₂O to alkyne…
A: -> Alkyne give first of all syn addition of (Sia)2BH to it then H2O2/OH- reacts and form enol…
Q: Classify the following substituents according to whether they are electron donors or electron…
A: Mesomeric effect=> acceptor is the one when it contains electron withdrawing group. Donor when…
Q: Use the information provided to determine AH°rxn for the following reaction: AH°f (kJ/mol) CH4(g) +4…
A:
Q: Unshared, or lone, electron pairs play an important role in determining the chemical and physical…
A: Elements have electrons in valence shell called valence electrons . In formation of compounds…
Q: (a) What is the main difference between a UV-Vis spectrophotometer and a spectrofluorometer? (b)…
A:
Q: When heated, KClO3KClO3 decomposes into KClKCl and O2.O2. 2KClO3⟶2KCl+3O22KClO3⟶2KCl+3O2 If…
A:
Q: A 0.04321 g sample of gas occupies 10.0 mL. at 294.5 K and 1.10 atm. Upon further analysis, the…
A: First of all we need to calculate the molar mass of the compound by using gas equation. And then we…
Q: Assuming 100% dissociation, calculate the freezing Point (TF) and boiling point (16) of 1.98m cact…
A: Given : Molality of CaCl2 solution = 1.96 M Kb of water = 0.52 K-kg/mol Kf of water = 1.86…
Q: Chlorine gas can be prepared in the laboratory by the reaction of hydrochloric acid with…
A:
Q: What is the major organic product of the following reaction? Problem viewing the image. Click Here O…
A: The addition of hydrogen halide to the alkene in presence of peroxide follows anti-markovnikov's…
Q: αcarboxyl and α-amine are 2.2 and 9.1 perspectively. Calculate isoelectric point of tyrosine. i want…
A:
Q: Use this graph to answer the following questions: What is the half life of the reaction? Round your…
A: Given the decomposition reaction of H2CO3(aq) is first order with respect to H2CO3(aq). Your…
Q: A 100ml sample of 0.20M HF is titrated with 0.10M NaOH. Determine the pH of the solution after the…
A:
Q: e. C. What is the standard cell potential (E°) for this reaction? d. What is the standard free…
A:
Q: The parent compound of phospholipids: O A. glycerol B. phosphatidic acid O c. ethanolamine OD. none…
A: B. Phosphatidic acid is the parent compound of phospholipids.
Q: Glutamine is a OA. basic OB. acid O c. neutral O D. racemin amino acid:
A:
Q: ew Walue Unit A Review 65 2 Complete the following written-response questions in the spaces…
A: Protons are positively charged and electrons are negatively charged.Neutrons have no charge.
Q: By the fast reaction, the iodine forms tetrathi t -12AA A A aly-A-E-EE ~ Styles 1- New Comment Align…
A:
Q: For the standardization procedure, 0.2023 g of anhydrous sodium carbonate was dissolved in 100 mt…
A: mass of anhydrous sodium carbonate, Na2CO3(s) taken for standardization = 0.2023 g Molar mass of…
Q: The F-C-F bond angle in the CH2F2 molecule is approximately. 180° 109.5° 90° O 120° O 60°
A: Given -> CH2F2 -> central atom is C . -> Side atoms are H and F.
Q: Write a chemical equation that shows the response of a HC2H3O2/C2H3O2- buffer to the addition of OH-…
A:
Q: GABA, gamma-aminobutyric acid, also known as "the calming chemical", is an inhibitory…
A: Here we have to determine the precursor amino acid for GABA.
Q: Determine the number of protons, neutrons and electrons in the following: 65 29 X O pt = 29 O pt =…
A:
Q: Which of (a)-(d) shows the same compound as the following? CH3 I HIC CI CH₂CH3
A:
Q: How would the reactant react with (CH2=CH)2CuLi ?
A: How would the reactant react with (CH2=CH)2CuLi ?
Step by step
Solved in 2 steps with 1 images

- Draw the molecular dipole or demonstrate the symmetrical cancellation of vectors in the following molecule, and describe it as polar or nonpolar. HCNDraw the molecular dipole or demonstrate the symmetrical cancellation of vectors in the following molecule, and describe it as polar or nonpolar. BF3Find the next term of the sequence. a, ar, ar2, ar3, . .
- Use appropriate translation vector, starting from the corner atom (0,0,0), to get to an atomon the face of the cubeDraw a Morse potential for a diatomic representing a bonding MO and one representing an antibonding MO.What is the point group for molecule C? I already found the point group for molecule A (C2h) and B (C2v), but I'm not sure which one is C

