Q: 3. Match the following molecular ions with their corresponding compound: 1001 50 92 100 50 92 100 92…
A: The objective of this question is to match the molecular ions with their corresponding compound.
Q: What neutral atom would have a photoelectron spectrum as the one shown below? × Cl B Ca C Ar DP D Р…
A:
Q: The reaction A - B + C is known to be first order in A Below are data showing the concentration of A…
A: Rate of reaction is defined as the change in concentration of reactant or product per unit time.…
Q: Try this Problem: Draw the lowest energy conformation and the highest energy conformation along the…
A: Conformations are molecules that can be interconverted to each other by rotation across…
Q: The Henry's law constant at 25 \deg C for CO₂ in water is 0.035 M/atm. What is the solubility of…
A:
Q: 2) For each compound below, identify the atom most likely to be attacked first by a nucleophile and…
A: When a atom is attracted by the nucleophile means the atom is electronically defficient.
Q: Ligand L binds to a binding protein with a K of 400 nM. Calculate the ligand concentration based on…
A:
Q: An ore sample was analyzed for its Fe content. Student A analyzed the sample a total of six times…
A: Analyzing ore samples is a crucial step in the mining and mineral processing industry to determine…
Q: Draw the products of the four step reaction sequence shown below. Ignore inorganic byproducts. If…
A: Aromatic compounds show electrophilic substitution reactions. Further substitution for mono…
Q: The glass electrode used in commercial pH meters gives an electrical response proportional to the…
A: A buffer is a solution which resists any change in pH on adding a small amount of acid or base.It is…
Q: CH3 HỌC=C-CH, CH, CH Spell out the IUPAC name of the compound.
A: Organic compounds can be defined as compounds that contain carbon and hydrogen atoms. The carbon…
Q: 1) Sketch out a structure showing the geometry about the metal center as accurately as possil…
A: The electrons in the valence shell are counted by the electron-counting method. A complex with 18…
Q: Indicate the substance indicated would correspond to this proton NMR spectrum. (The numbers…
A: The objective of the question is to identify the substance that corresponds to the given proton NMR…
Q: Oxygen can be generated by the thermal decomposition of potassium chlorate 2KCIO3(s) → 2KCI(s) +…
A: The volume of oxygen gas (O2) given is .The pressure is .The temperature is .The given balanced…
Q: Identify the Major and ALL Minor product(s) that are expected for each of the following reactions.…
A:
Q: Draw structural formulas for the alkoxide ion and the alkyl(aryl)bromide that may be used in a…
A: Organic compounds can be defined as compounds that contain carbon and hydrogen atoms. The carbon…
Q: Calculation of lonic Radii The edge length of the unit cell of LiCl (NaCl-like structure, FCC) is…
A: The unit cell is shown as follows:
Q: [ReferencEIJ How many monochloro substitution products are produced when the alkane below is…
A: The hydrogen in alkane undergoes radical reaction in presence of UV light to form chlorinated…
Q: EXPERIMENT 3 FREEZING POINT DEPRESSION AND VAN'T HOFF FACTOR OF SALINE SOLUTIONS
A: The colligative properties are the properties that undergo a change when a solute is introduced to a…
Q: D. Compounds containing deuterium are useful for metabolic studies. One way to introduce deuterium…
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: What would be the major product(s) of the following reaction? i IV and V I and II Ill only OIII and…
A:
Q: How many moles of sodium are in 1.73 x 10^24 Na atoms?
A: We have to find how many moles of sodium are in 1.73 x 10^24 Na atoms.
Q: Three samples of three different gases are listed in the table below. All the samples contain…
A: The ideal gas equation gives the relation between pressure, volume, temperature, and the number of…
Q: For the following reaction, 34.4 grams of sulfuric acid are allowed to react with 23.0 grams of…
A:
Q: 2. Determine whether cis-trans isomerism is possible for each of the following cycloalkanes. If so,…
A: The objective of the question is to predict cis - trans isomerism for the following cycloalkanes…
Q: glycoside formation (to form disaccharides and polysaccharides): CH₂OH OH ОН CH2OH OH HO OH OH…
A: Given two reactions of carbohydrates -Glycoside formation is a reaction in which hemiacetal converts…
Q: Three samples of three different gases are listed in the table below. All the samples contain…
A: The ideal gas equation gives the relation between pressure, volume, temperature, and the number of…
Q: 11) Bromine has two naturally-occurring isotopes. 79Br has a mass of 78.9 amu and accounts for 50.3%…
A:
Q: nswer the questions in the table below about this molecule: What is this molecule's chemical…
A: Here, we need to give the chemical formula of a given chemical structure. The chemical formula of a…
Q: When 21.0 g of calcium metal is reacted with water, 21.00 g of calcium hydroxide is produced. Using…
A: The objective of this question is to calculate the percent yield of the reaction of calcium with…
Q: Curved arrows are used to illustrate the flow of electrons. Follow the curved arrows and draw the…
A: Sodamide is a strong base, which abstracts beta-hydrogen to form corresponding alkene. The given…
Q: Ionization is the process of removing an electron from an atom. For an atom, X, the first and second…
A: Given that, the equation is:
Q: Acetylation: tro S 요 :0: CH3CO₂H HH 11 OH H 12 HH CH3
A: Given reaction is an example of the substitution reaction of amine to form amide as a product. i.e.…
Q: It bris 7) Write the expanded structural formula for this molecule. Make sure that you have the…
A: Answer:In the structural formula each bond is represented using simple straight lines with standard…
Q: Sample Exercise 4: Calculation of molality and Molarity A solution is made by dissolving 4.35 g…
A: The objective is to find out the molality and molarity of glucose given glucose. water. density…
Q: From the following reaction, please select the major endo product. CH3 A. H3C. ко-право А. B. O C. O…
A: Since you have posted a question with multiple questions, we will provide the solution only to the…
Q: □ A compound with molecular formula Cg H₁7 N 2 cannot be a: THD= 848.5 40.5 0.5 +6.5 +1=2 IHD-…
A: This problem is based on Basic Organic chemistry.Here it is asked that which functional group may be…
Q: Calculate the DGxn for the following reaction at 25°C under the given partial pressure conditions:…
A:
Q: A solution is made by mixing 41.0 mL of ethanol, C₂H6O, and 59.0 mL of water. Assuming ideal…
A:
Q: Use an orbital diagram to describe the electron configuration of the valence shell of each of the…
A: Given some elements from the periodic table to write the orbital electronic configuration.The…
Q: What is the correct name of this hydrocarbon compound by the IUPAC style?…
A:
Q: The transformations above can be performed with some combination of the reagents listed below. Give…
A:
Q: A + B → C [A](M) [B](M) Initial Rate (M/h) 0.139 0.228 132 0.621 0.228 362 0.139 0.779 862 Find the…
A: In this reaction, reactants are A and B.Rate of the reaction depends on the concentration of…
Q: A 2.45 g mixture of BaCl2 and Ba(NO3)2 is dissolved in water. Excess AgNO3 is added and all of the…
A: The objective of this question is to determine the mass of Ba(NO3)2 in the original mixture. This…
Q: Naproxen has the chemical formula, C14H1403. How many moles of carbon are in 72.41grams of Naproxen?…
A:
Q: An analytical chemist is titrating 160.9mL of a 0.6300M solution of ethylamine C2H5NH2 with a…
A:
Q: What is the change in boiling point for a solution containing 0.251 mol of naphthalene (a…
A: The moles of naphthalene = 0.251 molThe mass of the solvent (benzene) = 250 g =0.250 kgThe change in…
Q: Draw the reactants that would react to form this cyclohexene derivative in a Diels-Alder reaction.…
A: Given that, the structure of the Diels-Alder product is:Diels-Alder reaction is a type of…
Q: The quality assurance lab just received a shipment of three solutions. The lab manager asks you to…
A: We have to calculate the hydrogen ion concentration of each solution.
Q: Greenriverose is a fake disaccharide composed of \alpha D-tagatose and \beta -D-allose (in that…
A: The aim of the question is to draw the Haworth structure of an imaginary disaccharide. Disaccharides…
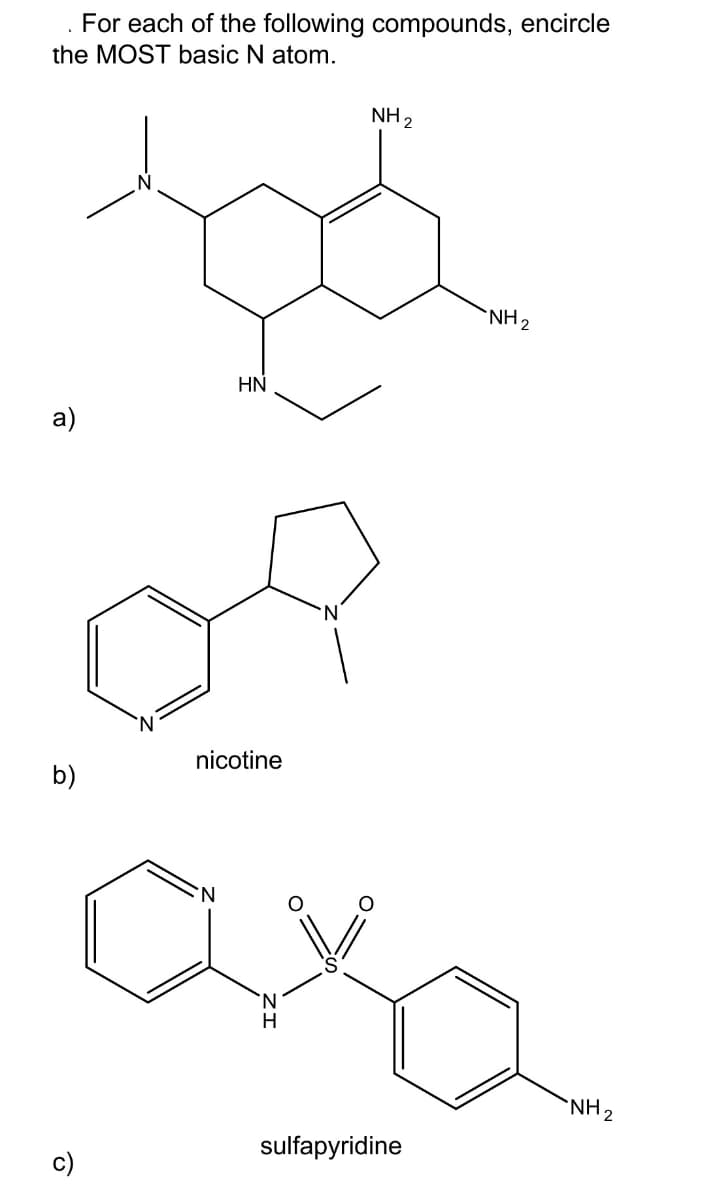
Step by step
Solved in 3 steps with 1 images

- Compound A + Compound B = ? Please draw the compoundThe structure of a potent analgesic agent (pain reliever) is provided below. Circle and identify each functional group in this compoundIn HF , neither H nor F holds a full formal charge of +1 or 1 . Organic chemists represent apartial charge using the Greek letter delta () . On the electron density map of the molecule HF above, add a + to one atom and a to the other to indicate which way the bond is polarized.
- Answer the following questions by referring to the ball-and-stick model of fentanyl, a potent narcotic analgesic used in surgical procedures. a. Identify the functional groups. b. Label the most acidic proton. c. Label the most basic atom. d. What types of intermolecular forces are present between two molecules of fentanyl? e. Draw an isomer predicted to have a higher boiling point. f. Which sites in the molecule can hydrogen bond to water? g. Label all electrophilic carbons.Answer the following questions by referring to the ball-and-stick model of fentanyl, a potent narcotic analgesic used in surgical procedures.a.Identify the functional groups. b. Label the most acidic proton. c.Label the most basic atom. d.What types of intermolecular forces are present between two molecules of fentanyl? e.Draw an isomer predicted to have a higher boiling point. f.Which sites in the molecule can hydrogen bond to water? g.Label all electrophilic carbons.Which are aromatic species? Please circle correct species. (assume planarity unless otherwise noted)

