Q: In CF3Cl the C—Cl bond-di s sociat ion energy i s339 kJ>mol. In CCl4 the C—Cl bond-dissociation…
A: For any type of electromagnetic radiation, the relation between wavelength, wavenumber and energy…
Q: Give chemical reaction in support of the statement that all the bonds inPCl5 molecule are not…
A: PCl5 molecule- In PCl5 molecule dsp3 hybridization is present. Molecular domain geometry is…
Q: Calculate ΔfH∘(SO3) from the following information in kJ/mol . S(s)+O2(g)⟶SO2(g)ΔrH∘= −296.8…
A: Hess's law Hess's law states that if a reaction can take place by more than one route, and the…
Q: i) Discuss the bonding and extra.stability of benzene,
A: The chemical formula of Benzene is C6H6. The hybridization of benzene is Sp2. The bond length of…
Q: Mention all the criteria/ conditions of forming different kinds of chemical bonds along with example…
A: Chemical bonds include covalent, polar covalent, and ionic bonds. Atoms with relatively similar…
Q: An elusive intermediate of atmo spheric reactions of HONO may be nitrosyl O-hydroxide, HOON.…
A:
Q: There is some indication that other hydrogen ring compounds and ions in addition to H3 and D3…
A: The Huckel determinant for each molecule is written as follows:
Q: Xenon, compared to the noble gases of lower molar mass, is the one that forms more compounds, such…
A: Noble gases do not exhibit bonding that easily because they have complete octet. Among all the…
Q: Question attached
A: Cyclopentadienyl is a highly stable anion this can be explained based on the number of pi electrons…
Q: Briefly describe the contribution of Walter nernest,t.w Richard,max plank and g.n Lewis in the…
A: The third law of thermodynamic was first state by Walther Nernst during the years 1906–12 "for…
Q: Both cis and trans isomers of IO2F4- have been observed. Can IR spectra distinguish between these?…
A:
Q: b. Will vo2+ disproportionate or will V3. and VO2 comproportionate to form V02+ under acidic…
A: Under acidic condition V+3 is most stable Under basic condition V2O3 is most stable
Q: On the basis of Molecular Orbital Theory argument, account for the reason why the enthalpy of…
A:
Q: Explain in detail what are dienes and how many types of dienes are there and the difference between…
A: The name of hydrocarbon ends with suffix "ene" termes as alkenes. Alkenes are hydrocarbons having…
Q: The dissociation energy of a carbon–bromine bond is typicallyabout 276 kJ/mol. (a) What is the…
A: a. The amount of energy (E) required to break a single C — Br bond is calculated by the formula,
Q: The wavelength at which the O2 molecule most stronglyabsorbs light is approximately 145 nm. (a) In…
A: INTRODUCTION: Electromagnetic spectrum alse called as EM radiation in which we study about different…
Q: Three biological ly important diatomic species. either because they promote or inh ibit life. are…
A: Electron configuration of given molecules are as follows,
Q: a) LMCT energies of dº oxoanions follow the orders: vo.* Cro,² MnO, MnO, TcO, ReO,
A: In this question, we have to find out the correct answer of given problem by the help of the…
Q: The bond energy for HBr is 363 kJ/mole. What is the longest wavelength (in nm) that can dissociate a…
A: Answr-1 Energy of photon in terms of wavelength is: E=hcλλ=hcE here: C=speed of lighth=planck…
Q: Use the data given below to calculate the bond energies (Do) (in eV) of 1H35Cl and 1H81Br molecules.…
A: HCl Ve (cm^-1): 2991, De(cm^-1): 53194 HBr Ve(cm^-1):2649 , De(cm^-1): 34570
Q: The positions of the Br atoms in the molecule below is designated as Br -Br O meta O ortho O para O…
A: Since you have posted multiple questions, we are entitled to answer the first only.
Q: The bond dissociation energy to break 3 N-H bond(s) in 1 mole of NH, molecules is Single
A: • Ammonia, NH3 has a trigonal pyramidal structure and may be represented as, • Thus, NH3…
Q: 11 of 12 By how much energy is the system stabilized by having the two H atoms 74 pm from each other…
A: In this question, we want to determine the energy in kJmol-1 You can see details Solution below.
Q: he most s ais
A: The valence shell is the last shell of an atom and the electrons that belong to the valence shell is…
Q: =3,ℓ=3, what can be deduced about ??
A: We know about, Azimuthal quantum number ( l ) value depends on principle quantum number ( n ) l = 3…
Q: By using photons of specific wavelengths, chemists can dis-sociate gaseous HI to produce H atoms…
A: The bond energy of HI = 295 kJ/mol. The energy of 1 molecule of HI using the Avogadro's number: E…
Q: has neutral atom of an electrom confienuration ending in 3p while an dnion of with a -2 charge hao°…
A: Neutral atom with 3p6 configuration is Ar. The atom whose -2 anion will have 3p6 configuration is S.
Q: What is the Huckel π–electron energies of benzene?
A:
Q: Draw a Lewis structure of the thiocyanate ion that contains a triple carbon-nitrogen bond. When…
A:
Q: Use the following data (in kJ/mol) to estimate AH for the reaction S (g) +e → s² (g) . Include an…
A:
Q: Cyclopentadienyl anion (shown below) is an interesting organic molecule because it consists only of…
A: Cyclopentadienyl is a highly stable anion. This can be explained based on the number of pi electrons…
Q: In which of these two hydrides:NH3or SbH3,would you expect a greater distortion from the normal…
A: Bond angle increases with the increasing electronegativity of the central atom.
Q: Calculate AS° for C(g) + 2 H2(g) → CH,(g). Substance S° (J/mol·K) С (9) 5.7 H2 (g) 130.6 CH4 (g)…
A: Entropy of a reaction is the randmoness of the system in which it takes place. Entropy is an…
Q: Find the spectral terms of Hez, He' molecules and ions by showing their lowest electronic states.…
A: Solution: Both He2 and H2+ are in less than 14 electron category. So filling the MO to find out…
Q: Determine ASm for H2(g) + I,(g) = 2HI(g) given the following informatic Substance S° (J/mol · K)…
A: Introduction: We have to calculate entropy. Given:
Q: Use the following data (in kJ/mol) to estimate AH for the reac- tion S-(g) + e-→ S²-(g). Include an…
A:
Q: The wavelength at which the O2 molecule most strongly absorbs light is approximately 145 nm. Would a…
A: Given, E=495 KJ/mol For one molecule, E=495×1036.023×1023E=82.184×10-20 J/bond (where…
Q: Using the values provided in the table below, calculate AS for the following reaction: 2SO3(g) O2(g)…
A: Given Reaction2 SO3(g) → O2(g) + 2SO2(g) Species S° (J/mol.K) SO2(g) 248 SO3(g) 114…
Q: What changes in relative energy you would expect as the SN2 conversion from CH3-Br to CH3-Cl takes…
A:
Q: Describe and justify the Pauling and Mulliken electronegativity scales.
A:
Q: Explain when is σ- and π-notation used, and when is a-, e- and t-notation used in MO energy level…
A: MO diagram is a qualitative descriptive tool explaining chemical bonding in molecules in terms of…
Q: Find the spectral terms of H2 and He2* molecules and ions by showing their lowest electronic states.…
A: Spectral term :- It indicates the electron are arranged in different possible…
Q: Consider the bonds labelled A and B below. Which one has a higher bond strength and why? Comment on…
A: A is a C-Cl single covalent bond. B is a C-Br single covalent bond. The strength of a covalent bond…
Q: Louve the mnit tconic A1³+, Co₂²/ Cervation of NaOH and 202 reactal with A13+ Cr3+, Fe3+, mn²+, Ni24…
A:
Q: (d) The elements of Group I and II elements of the Periodic Table are called S-block elements, (i)…
A: The elements of Group 1 and Group 2 of the modern periodic table are called S block elements. The…
Q: Describe the location of the maximum electron density of σ- and π-bonds. How is the difference in…
A: To find: the location of the maximum electron density of σ- and π-bonds? difference in the spatial…
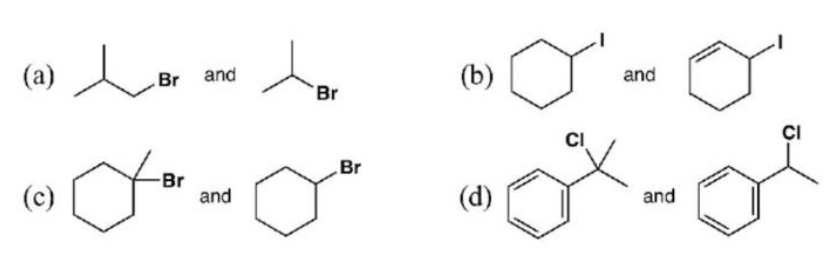
Which of the following substances in each per will be the most reactive in SN1 reactions? justify
Step by step
Solved in 2 steps with 1 images

- The skeleton of benzyldimethylamine is The number of available valence electrons is as follows: from the benzyl group, ___; from each of two methyl groups, ___; and, from the nitrogen atom, ___, for a total of ___. Filling in the skeleton with single bonds gives ____________________________ and adding the appropriate unshared pair gives ____________________________ The number of electrons in the functional group of this structure is ___, and the structure is (correct, incorrect).Sketch the following molecules and label the specific groups or bonds requested with the structure.The branched chain amino acids are _____________,_____________, and _____________.
- The following are the objective To discuss the elements or compounds determined with the help of polarography is different types of samples like food stuff, sea water, fuels, etc. To discuss the polarographic analysis of organic compounds To discuss other industrial important determinations for traces or minor constituents Give me the conclusion and thoughts for the following objectives. If possible notes is a must. Thank you so much.please write the Structure Activity Relationship (SAR) of Bisoprolol and Esmolol? Please write at your own words.How is CCl2F2 polar given that is tetrahedral( highschool level explanation pls)?

