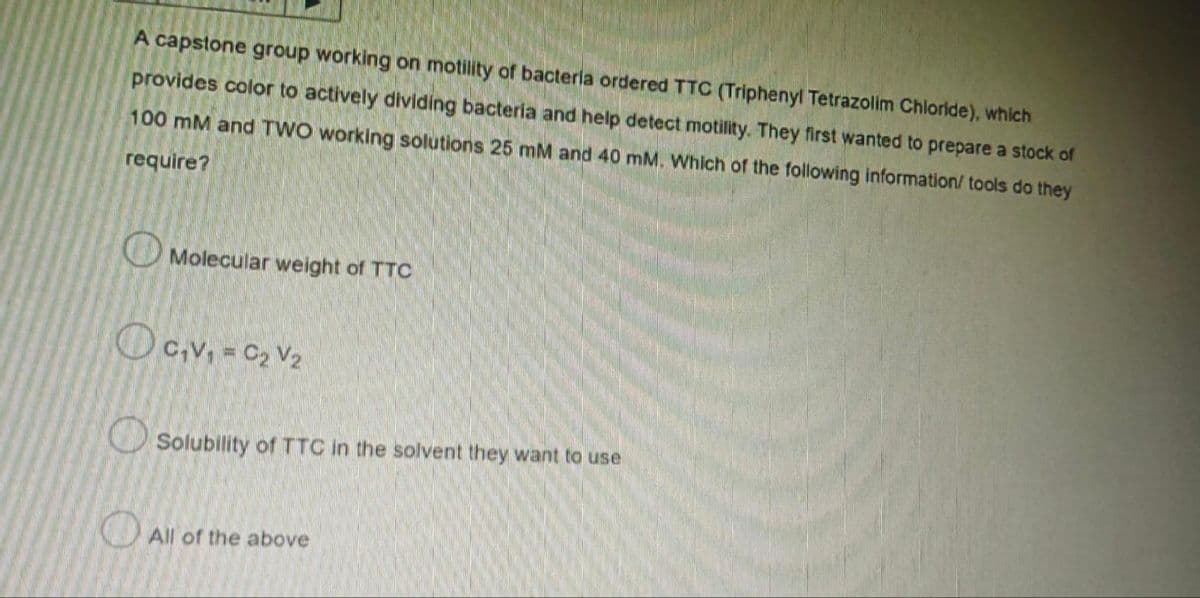
A capstone group working on motility of bacteria ordered TTC (Triphenyl Tetrazolim Chloride), which provides color to actively dividing bacteria and help detect motility. They first wanted to prepare a stock of 100 mM and TWO working solutions 25 mM and 40 mM. Which of the following information/ tools do they require? Molecular weight of TTC C₁V₁ = C₂ V₂ Solubility of TTC in the solvent they want to use All of the above
A capstone group working on motility of bacteria ordered TTC (Triphenyl Tetrazolim Chloride), which provides color to actively dividing bacteria and help detect motility. They first wanted to prepare a stock of 100 mM and TWO working solutions 25 mM and 40 mM. Which of the following information/ tools do they require? Molecular weight of TTC C₁V₁ = C₂ V₂ Solubility of TTC in the solvent they want to use All of the above
Chapter31: Introduction To Analytical Separations
Section: Chapter Questions
Problem 31.19QAP
Related questions
Question
100%
Transcribed Image Text:A capstone group working on motility of bacteria ordered TTC (Triphenyl Tetrazolim Chloride), which
provides color to actively dividing bacteria and help detect motility. They first wanted to prepare a stock of
100 mM and TWO working solutions 25 mM and 40 mM. Which of the following information/ tools do they
require?
Molecular weight of TTC
C₁V
C₁V₁ = C₂ V₂
Solubility of TTC in the solvent they want to use
A
All of the above
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning