A certain ideal gas (unknown) inside a close piston-cylinder assembly undergoes a set of processes that composed of Isothermal expansion from State point 1 to state point 2, Isometric heat rejection from state point 2 to state point 3, and Isentropic compression from state point 3 back to the initial condition. If the maximum pressure is 3000kPa and maximum and minimum volume is 200cm³ and 50cm³, analyze the problem and perform the following: a. Draw and label the graph of this set of processes in the P-V and T-S diagram showing the state point numbers and energy directions. b. From the known ideal gases as shown in Table 1.0, select the best suited ideal gas to attain the processes stated if the lowest pressure is limited to 629.8kPa. Assume a constant specific heat. c. For the selected ideal gas from b, compute the work of compression (kJ/kg), work of expansion (kJ/kg), and heat rejected (kJ/kg) Table 1.0: Ideal gas specific heats of various common gases Gas constant, R k/kg-K Kikg-K kkg-K Gas Formula Air 0.2870 0.2081 0.1433 0.1889 0.2968 0.2765 0.2964 2.0769 4.1240 0.5182 0.4119 0.2968 1.005 0.5203 1.7164 0.846 1.040 1.7662 1.5482 5.1926 14.307 2.2537 1.0299 1.039 0.718 0.3122 1.5734 0.657 Argon Butane Carbon diaxide Carbon monoxide Ethane Ethylene Helium Hydrogen Methane Neon 1.400 1.667 1.091 1.289 1.400 1.186 1.237 1.667 1.405 1.299 1.667 1.400 Ar C,H0 co 0.744 1.4897 1.2518 3.1156 C,H, Не H2 CH. Ne 10.183 1.7354 0.6179 0.743 1.6385 0.658 1.4909 1.4108 Nitrogen Octane Oxygen Propane 0.0729 0.2598 0.1885 2.4615 1.7113 0.918 1.6794 1.8723 1.044 1.395 1.126 1.327 C,H,
A certain ideal gas (unknown) inside a close piston-cylinder assembly undergoes a set of processes that composed of Isothermal expansion from State point 1 to state point 2, Isometric heat rejection from state point 2 to state point 3, and Isentropic compression from state point 3 back to the initial condition. If the maximum pressure is 3000kPa and maximum and minimum volume is 200cm³ and 50cm³, analyze the problem and perform the following: a. Draw and label the graph of this set of processes in the P-V and T-S diagram showing the state point numbers and energy directions. b. From the known ideal gases as shown in Table 1.0, select the best suited ideal gas to attain the processes stated if the lowest pressure is limited to 629.8kPa. Assume a constant specific heat. c. For the selected ideal gas from b, compute the work of compression (kJ/kg), work of expansion (kJ/kg), and heat rejected (kJ/kg) Table 1.0: Ideal gas specific heats of various common gases Gas constant, R k/kg-K Kikg-K kkg-K Gas Formula Air 0.2870 0.2081 0.1433 0.1889 0.2968 0.2765 0.2964 2.0769 4.1240 0.5182 0.4119 0.2968 1.005 0.5203 1.7164 0.846 1.040 1.7662 1.5482 5.1926 14.307 2.2537 1.0299 1.039 0.718 0.3122 1.5734 0.657 Argon Butane Carbon diaxide Carbon monoxide Ethane Ethylene Helium Hydrogen Methane Neon 1.400 1.667 1.091 1.289 1.400 1.186 1.237 1.667 1.405 1.299 1.667 1.400 Ar C,H0 co 0.744 1.4897 1.2518 3.1156 C,H, Не H2 CH. Ne 10.183 1.7354 0.6179 0.743 1.6385 0.658 1.4909 1.4108 Nitrogen Octane Oxygen Propane 0.0729 0.2598 0.1885 2.4615 1.7113 0.918 1.6794 1.8723 1.044 1.395 1.126 1.327 C,H,
Elements Of Electromagnetics
7th Edition
ISBN:9780190698614
Author:Sadiku, Matthew N. O.
Publisher:Sadiku, Matthew N. O.
ChapterMA: Math Assessment
Section: Chapter Questions
Problem 1.1MA
Related questions
Question
Find the Given, Figure and the Unknown
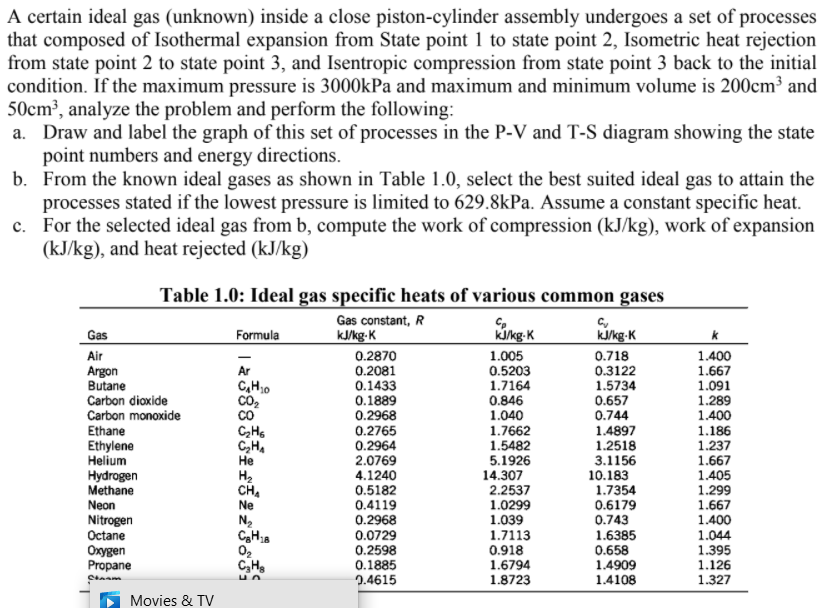
Transcribed Image Text:A certain ideal gas (unknown) inside a close piston-cylinder assembly undergoes a set of processes
that composed of Isothermal expansion from State point 1 to state point 2, Isometric heat rejection
from state point 2 to state point 3, and Isentropic compression from state point 3 back to the initial
condition. If the maximum pressure is 3000kPa and maximum and minimum volume is 200cm³ and
50cm³, analyze the problem and perform the following:
a. Draw and label the graph of this set of processes in the P-V and T-S diagram showing the state
point numbers and energy directions.
b. From the known ideal gases as shown in Table 1.0, select the best suited ideal gas to attain the
processes stated if the lowest pressure is limited to 629.8kPa. Assume a constant specific heat.
c. For the selected ideal gas from b, compute the work of compression (kJ/kg), work of expansion
(kJ/kg), and heat rejected (kJ/kg)
Table 1.0: Ideal gas specific heats of various common gases
Gas constant, R
kJ/kg-K
kikg-K
k/kg-K
Gas
Formula
1.005
0.5203
1.7164
0.846
1.040
Air
0.2870
0.2081
0.1433
0.1889
0.2968
0.2765
0.2964
2.0769
4.1240
0.5182
0.4119
0.2968
0.0729
0.2598
0.1885
2.4615
Argon
Butane
Carbon dioxide
Carbon monoxide
Ethane
0.718
0.3122
1.5734
0.657
0.744
1.400
1.667
1.091
1.289
1.400
C,H10
co
CHs
CH,
Не
H2
CH,
Ne
1.7662
1.5482
5.1926
14.307
2.2537
1.0299
1.039
1.7113
0.918
1.6794
1.8723
1.4897
1.2518
3.1156
10.183
1.7354
0.6179
0.743
1.6385
0.658
1.4909
1.4108
1.186
1.237
1.667
1.405
1.299
1.667
1.400
1.044
1.395
1.126
1.327
Ethylene
Helium
Hydrogen
Methane
Neon
Nitrogen
Octane
Oxygen
Propane
N2
CHa
02
Movies & TV
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Elements Of Electromagnetics
Mechanical Engineering
ISBN:
9780190698614
Author:
Sadiku, Matthew N. O.
Publisher:
Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:
9780134319650
Author:
Russell C. Hibbeler
Publisher:
PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:
9781259822674
Author:
Yunus A. Cengel Dr., Michael A. Boles
Publisher:
McGraw-Hill Education

Elements Of Electromagnetics
Mechanical Engineering
ISBN:
9780190698614
Author:
Sadiku, Matthew N. O.
Publisher:
Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:
9780134319650
Author:
Russell C. Hibbeler
Publisher:
PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:
9781259822674
Author:
Yunus A. Cengel Dr., Michael A. Boles
Publisher:
McGraw-Hill Education

Control Systems Engineering
Mechanical Engineering
ISBN:
9781118170519
Author:
Norman S. Nise
Publisher:
WILEY

Mechanics of Materials (MindTap Course List)
Mechanical Engineering
ISBN:
9781337093347
Author:
Barry J. Goodno, James M. Gere
Publisher:
Cengage Learning

Engineering Mechanics: Statics
Mechanical Engineering
ISBN:
9781118807330
Author:
James L. Meriam, L. G. Kraige, J. N. Bolton
Publisher:
WILEY