A CH3CHCH2CH2NH2, TSOH CH3CH2CH2NH2, H Q B TSOH C NH2OH, TSOH D E CH3CH2CH2CH2NH2, NaOH 1. CH3CH2CH2CH2NH2, Na(CN)BH3, pH 4-5 2. Neutralizing work- Done
A CH3CHCH2CH2NH2, TSOH CH3CH2CH2NH2, H Q B TSOH C NH2OH, TSOH D E CH3CH2CH2CH2NH2, NaOH 1. CH3CH2CH2CH2NH2, Na(CN)BH3, pH 4-5 2. Neutralizing work- Done
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter6: Solutions And Colloids
Section: Chapter Questions
Problem 6.83P: 6-83 (Chemical Connections 6C) The concentration of Ca2+ ion present in a blood sample is found to...
Related questions
Question
Choose the best reagents for the following reaction.
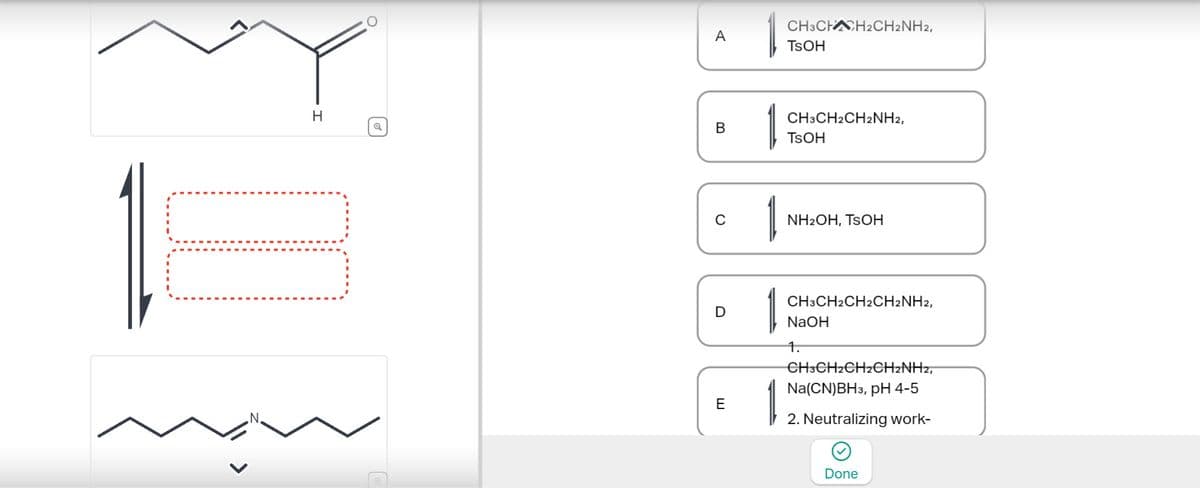
Transcribed Image Text:A
CH3CHCH2CH2NH2,
TSOH
CH3CH2CH2NH2,
H
Q
B
TSOH
C
NH2OH, TSOH
D
E
CH3CH2CH2CH2NH2,
NaOH
1.
CH3CH2CH2CH2NH2,
Na(CN)BH3, pH 4-5
2. Neutralizing work-
Done
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning