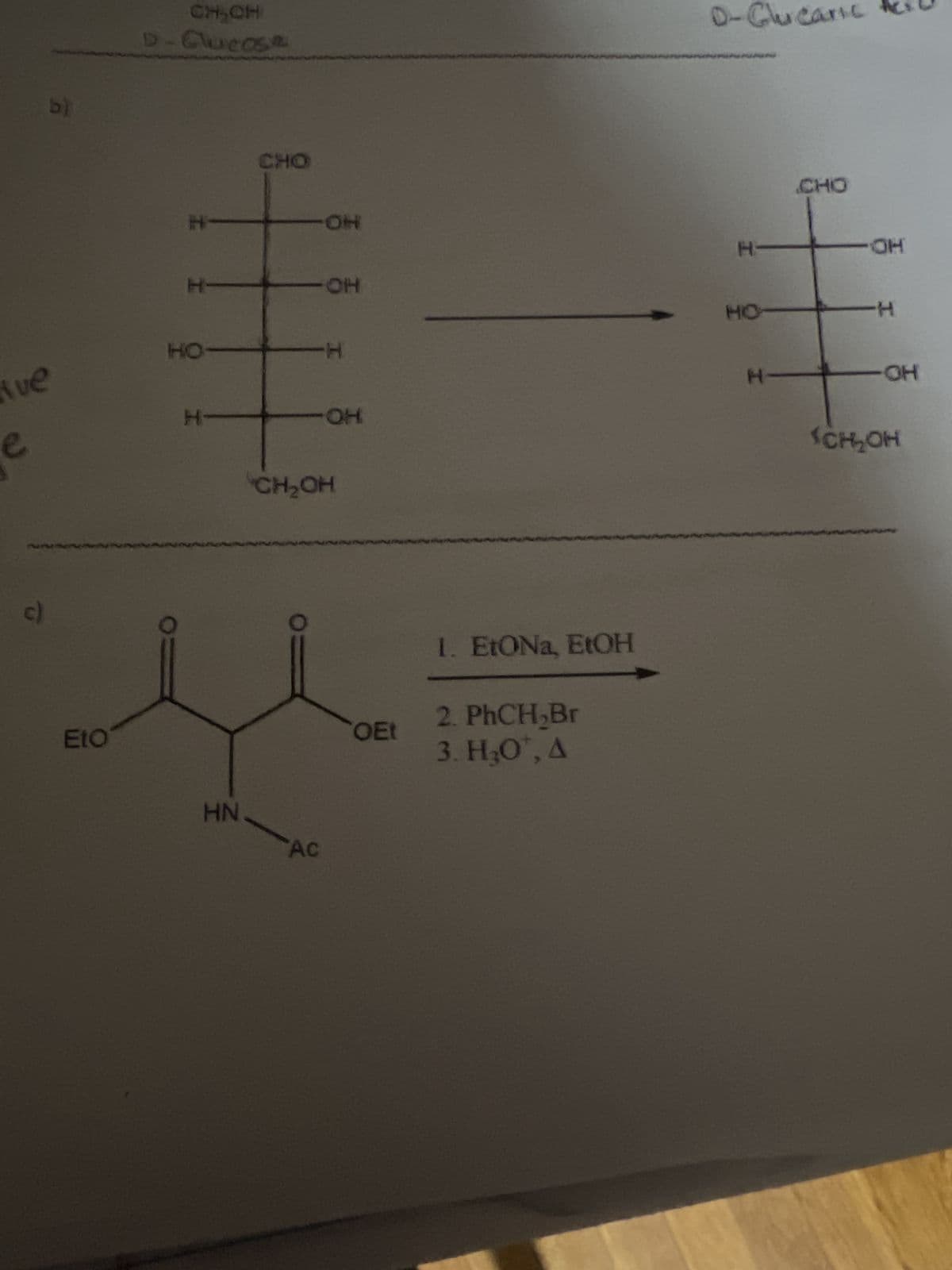
b) CHON D-Clucose e Ave c) -H CHO -OH D-Glucaric H- H- -OH HO- HO- -H H- -OH EtO HN CH₂OH AC OEt 1. EtONa, EtOH 2. PhCH₂Br 3. H₂OT, A H- CHO -OH -H -OH SCH₂OH
Q: A buffer is a mixture of HCN and KCN. When a small amount of HCI is added to this buffer, which…
A: In the buffer mixture of HCN (hydrogen cyanide) and KCN (potassium cyanide), you have both a weak…
Q: For a molecule, the phosphorescence lifetime without quenching is 1.0 s while with a quencher…
A: Given:Phosphorescence lifetime without quenching (τ0) = 1.0 sPhosphorescence lifetime with quencher…
Q: Draw the product of the reaction shown below at physiological pH (pH = 7.4). Ignore inorganic…
A: Step 1: Step 2: Step 3: Step 4:
Q: Determine the ΔG° in KJ for a voltaic cell made of a Ag strip and an Al strip immersed in 1.0 M…
A: 1. ExplanationOxidation half reaction Pb(s) <-------> Pb2+(aq) + 2e- E°red =…
Q: Show calculations to prepare 100 mL of 0.1 M CH3CO₂H solution from the 1 M CH,CO solution. Table…
A: The objective of the question is to prepare a 0.1 M CH3CO2H solution from a 1 M CH3CO2H solution and…
Q: Problem 11 of 30 Submit Draw the possible 1,2- and 1,4-addition products formed when this diene…
A: Step 1: Diene reacts with Br2 and gives 1, 2- addition product and 1, 4 - addition product. Step 2:…
Q: Vinegar is 5.00 % (by mass) acetic acid (MW = 60.052 g/mol). It has a density of 1.02 g/mL. What is…
A: The objective of this question is to calculate the molarity of acetic acid in vinegar. Molarity is a…
Q: 9. (10 marks) The equilibrium constant (KP) at 25 °C is 3.2 x 10-10 for the reaction: 2 Cr³+ (aq) +…
A: The objective of this question is to find the equilibrium concentrations of Cr3+, Cr2+, and Pb2+ in…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: Step 1: Step 2: Step 3: Step 4:
Q: Ree 1. Using the IUPAC nomenclature, write the name for the following organic compounds. HO- Nam HO…
A: Step 1: Step 2: Step 3: Step 4:
Q: Polymers may be composed of thousands of monomers. Draw three repeat units (trimer) of the polymer…
A: Thank you.
Q: An analytical chemist is titrating 173.3 mL of a 0.1600M solution of diethylamine ((C₂H)2NH) with a…
A: The objective of this question is to calculate the pH of the base solution after a certain volume of…
Q: Draw the x anomer product of this reaction. Ignore inorganic byproducts. HO HOH OH HO H H H3O+…
A: In cyclic monosaccharides or glycosides, anomers are epimers which means that they differ only in…
Q: Calculate the volume percent of solute in each of the solutions. A solution made by adding 20.1 mL…
A: The objective of the question is to calculate the volume percent of solute in the second solution.…
Q: A 100.0 g piece of metal at 100°C is placed in contact with 50.0 g of water (s = 4.184 J g–1 °C–1)…
A:
Q: 2. Use an empirical fit of the Clausius-Clapeyron equation es (T) = 6.1094 exp 17.625T T + 243.04…
A: The formula for saturation vapor pressure is given as:es(T)=6.1094⋅e(T+243.0417.625T) Tabulating…
Q: Show the steps necessary to transform the acid on the left into the compound on the right. Be sure…
A: Answer:
Q: 1. Propose reagents for the following reaction. (Hint: Only one of the two ways of adding a carbon…
A: Here's the step-by-step mechanism for the conversion of a 1,4-halohydrin to a lactone. In your case,…
Q: Give the major organic product(s) of the following reaction. NaOCH2CH3 NaOH, H₂O H+ ? CH₂CH₂OH heat…
A:
Q: None
A:
Q: Of the following, which represents the intermediate that is most likely formed in the first step of…
A: The question is not clear whether the addition to the alkene is using Br2 or HBr. If it is…
Q: ■ Vodacom 4G 20:13 21% := ACA470S Assignment Done Û ACA470S Assignment Due: 12 April 2024 Total: 30…
A: The objective of the question is to understand the reasons behind certain practices in voltammetry…
Q: For the reaction 4HCl(g) + O2(g) → 2H2O(g) + 2Cl2 (9) AH° -114 kJ and AS° = -129 J/K The equilibrium…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Solid copper(Il) acetate is slowly added to 50.0 mL of a 0.0631 M ammonium sulfide solution. The…
A: The Ksp expression for this reaction is:Ksp = [Cu^2+] [S^2-]We know that Ksp for CuS is 7.9 ×…
Q: Conjugate acid base pKa 2) Consider the reaction B: () + H3O+ →>> BH(+) + H2O. For the following…
A: Note: The pKa values are not exactly correct there may be minor differences.
Q: None
A: The correct IUPAC name for the compound shown in the image is…
Q: None
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: None
A:
Q: i need help filling out the following SN2 reactions with appopiate reactants, products, or reagents,
A: Step 1:(a) SN2 (Bimolecular substitution reaction):In this type of reaction mechanism, the…
Q: For the reaction2C2H6(g) + 7O2(g)4CO2(g) + 6H2O(g)H° = -2.86×103 kJ and S° = 92.7 J/KAt standard…
A: The objective of the question is to determine at what temperature range the given reaction would be…
Q: When the Pb2+ concentration is 5.00\times 10-4 M, the observed cell potential at 298K for an…
A: Step 1:Introduction to data :Given ,Concentration of Pb2+= 5.00 X 10-4Objective :Determine the…
Q: For the reaction CaCO3(s) CaO(s) + CO2(g) AG° = 136 kJ and AH = 178 kJ at 259 K and 1 atm. This…
A: To find the entropy change (Delta S) for the reaction, we can use the relationship between the Gibbs…
Q: Lead consists of four naturally occurring isotopes. Calculate the atomic mass for lead using the…
A: IsotopeMassAbundance (%)204.01.40206.024.10207.022.10208.052.40
Q: Organic Chemistry Problem. Please help with figuring out what steps to take to obtain the product of…
A: Primary alcohols as well as aldehydes such as the given in the problem can undergo oxidation to form…
Q: Please answer question below, be very deatiled and draw out the mechanism reaction with arrows
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A: Answer well explained above
Q: For the reaction N2(g) + O2(g) → 2NO(g) AG° = 173 kJ and AS° = 24.9 J/K at 323 K and 1 atm. favored…
A: The objective of the question is to determine whether the given reaction is favored under standard…
Q: Which of the following determines the direction of spontaneity? A Go Only option (B) Both (A) and…
A:
Q: Conjugate base acid Iso-Butyronitrile pka_ → 1) Consider the reaction AH(+) + H₂O A:+H3O+. For the…
A: Iso-ButyronitrileStructure of acid: CH3−CH(CH3)−CN.pKa of conjugate acid: 25Name of conjugate…
Q: Please correct answer this question
A:
Q: The table below contains partial information about several reactions. Fill in the missing…
A:
Q: I need answer expert solutions Need answer step by step
A: Step 1: Answer is as follows: In this reaction carbonyl double bond breaks and form the negative…
Q: Problem 23 of 30 Submit Draw the major addition product for this reaction. Do not include any…
A: The clue here is that Bromine attaches to a much stable carbocation…
Q: y" 3y 18y = sin(8) = y(0) 3, y'(0) = -1 Using Y for the Laplace transform of y(t), i.e., Y =…
A: Step 1: Step 2:
Q: Question 5 Consider the equilibrium reaction: N2(g) + t 3 H2(g) 2 NH3(g) AH = -91.8 kJ (exothermic)…
A: The objective of this question is to identify the methods that can shift the equilibrium of the…
Q: Use the activities to calculate the molar solubility of Zn(OH)2 in 0.0167M K2SO4. What is the…
A: Step 1: Step 2: Step 3: Step 4:
Q: For the reaction 2 H2(g) + O2(g) → 2 H₂O(g) which equation would be used to determine the entropy…
A: Step 1: Step 2: entropy change can be determined by calculating the change in number of gaseous…
Q: Need answer step by step I need answer expert solution
A: Step 1: Step 2: Step 3: Step 4:
Q: Which of the following will not undergo catalytic reduction in the presence of H, and metal…
A: The objective of the question is to identify which of the given groups, OH or CEN, will not undergo…
Q: 6. Starting with benzyl bromide, show how you would synthesize each of the following: (a) (b) OH (c)…
A: a: To synthesize phenyl-2-propanol from benzyl bromide, the following steps can be…
B)
I need expert solutions and step by step answer
Unlock instant AI solutions
Tap the button
to generate a solution
Click the button to generate
a solution
- Draw the products formed when CH3COCH2CH2CH = CH2 is treated with each reagent:(a) LiAlH4, then H2O;(b) NaBH4 in CH3OH;(c) H2 (1 equiv), Pd-C;(d) H2 (excess), Pd-C;(e) NaBH4 (excess) in CH3OH;(f) NaBD4 in CH3OH.Write down the reactions: D-Glucose + [Ag(NH3)2]+ →1. Carbohydrates classification. 2. Write down the reactions: a) α,D-Glucopyranose + C2H5OH → b) D-Glucose + [Ag(NH3)2]+ → c) D-Glucopyranose + (CH3CO)2 O → d) D-Glucopyranose + CH3I → e) D-Glucose + HNO3 → f) D-Glucose + H2 → g) Lactose formation h) Sucrose hydrolysis 3. Write down the formula of β,D-galactopyranose
- Draw the products formed when β-D-galactose is treated with each reagent. a. Ag2O + CH3I b. NaH + C6H5CH2Cl c. The product in (b), then H3O+ d. Ac2O + pyridine e. C6H5COCl + pyridine f. The product in (c), then C6H5COCl + pyridineO2CCH,CH,-C-CO2 + CH2 NH3 NH3 O2CCH,CH,-C-CO2 + CH2 CH2-C-CO2 + H. phenylalanine -CO2 0,CCH,CH2—CO2 CH2-C-CO2Predict the products obtained when d-galactose reacts with each reagent. (h) NaBH4(i) Br2, H2O, then H2O2 and Fe2(SO4)3
- 1) Phenyl propyl ether can be obtained by reacting? 2) Raffinose is A trisaccharide which is made up of two sugar molecules of aldoses and one sugar molecule of ketoses? True or false!Compound A boils at 25 C Compound B boils at 80 C Which has the strong IMFs? OB O APredict the products obtained when d-galactose reacts with each reagent. (f) excess Ac2O and pyridine (g) excess CH3 I, Ag2O
- If anisole is allowed to sit in D2O that contains a small amount of D2SO4, what products are formed?Draw the products formed when ß-D-galactose is treated with each reagent.a. Ag2O + CH3Ib. NaH + C6H5CH2Clc. The product in (b), then H3O+d. Ac2O + pyridinee. C6H5COCl + pyridinef. The product in (c), then C6H5COCl + pyridineDraw the products formed (including stereoisomers) when attached compound is reduced with NaBH4 in CH3OH.

