A chemist measures the energy change AH during the following reaction: 2 H,O(1) 2 H2(9)+O2(9) AH=572. kJ Use the information to answer the following questions. endothermic. This reaction is... x10 exothermic. Suppose 42.1 g of H,O react. Yes, absorbed. O Yes, released. Will any heat be released or absorbed? No. you said heat will be released or absorbed in the second part of this question, calculate how much heat will be released or absorbed. If Round your answer to 3 significant digits.
A chemist measures the energy change AH during the following reaction: 2 H,O(1) 2 H2(9)+O2(9) AH=572. kJ Use the information to answer the following questions. endothermic. This reaction is... x10 exothermic. Suppose 42.1 g of H,O react. Yes, absorbed. O Yes, released. Will any heat be released or absorbed? No. you said heat will be released or absorbed in the second part of this question, calculate how much heat will be released or absorbed. If Round your answer to 3 significant digits.
Chapter10: Energy
Section: Chapter Questions
Problem 61A
Related questions
Question
100%
Use the photo attached please
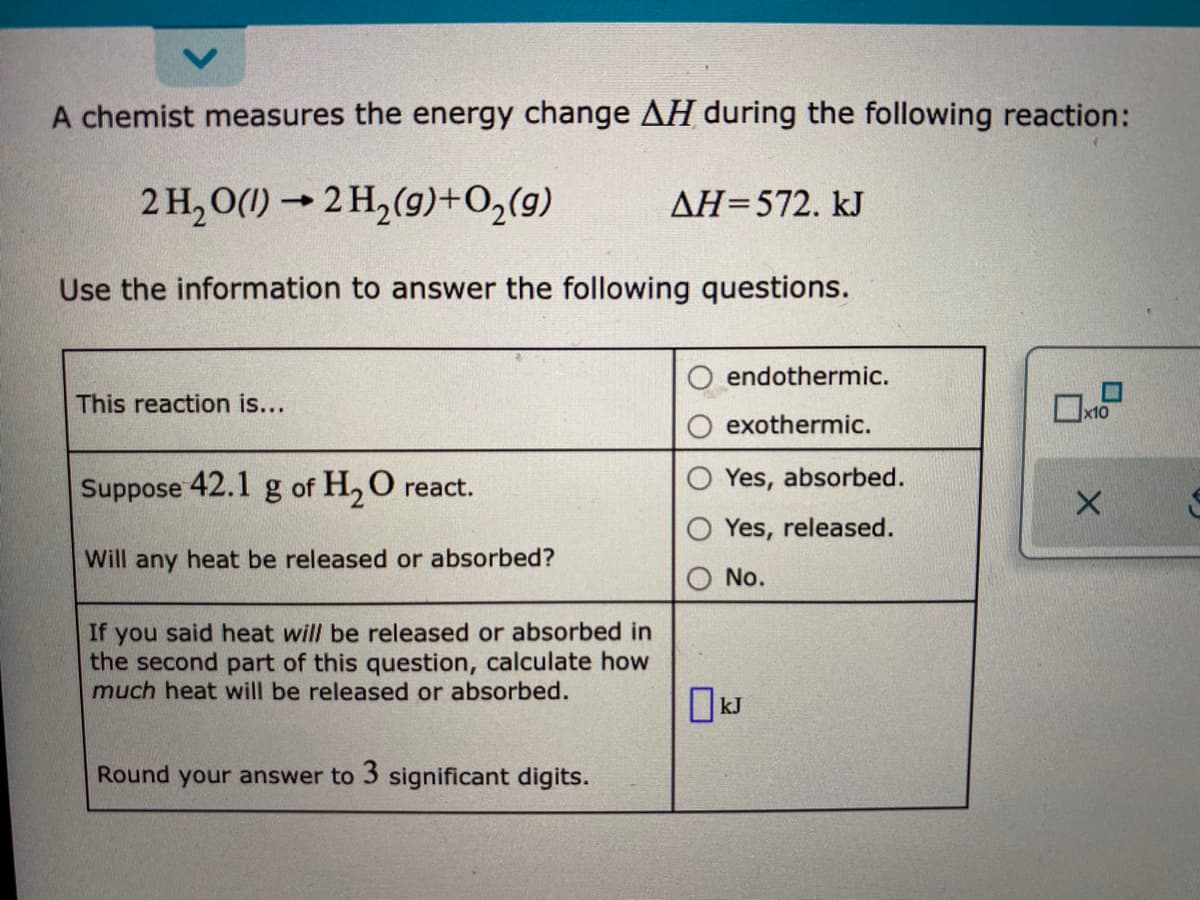
Transcribed Image Text:A chemist measures the energy change AH during the following reaction:
2 H, O(1) → 2 H,(9)+O,(9)
AH=572. kJ
Use the information to answer the following questions.
endothermic.
This reaction is...
x10
exothermic.
Suppose 42.1 g of H, O react.
Yes, absorbed.
Yes, released.
Will any heat be released or absorbed?
No.
If you said heat will be released or absorbed in
the second part of this question, calculate how
much heat will be released or absorbed.
OkJ
Round your answer to 3 significant digits.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning