(a) Determine whether the value of K of an equilibrium system will be affected or unaffected by the following changes: Changes Value of K (affected or unaffected) (Decreasing the temperature (u) Increasing the pressure of the system (uD Decreasing the concentration of reactant (b) The following graph represents the amount of CO formed at two different temperatures, and Ts. The reaction as shown below: Cts) + H,O(g) = Co(g) + H:(g) T 385 K T = 335 K time Based on the graph, determine whether the forward reaction is endothermic or exothermic. Predict if the concentration of H.O gas increases, decreases or remains the same if the temperature is decreased to 298 K. Explain. A mixture of 0.350 M of H:0, 0.125 M of CO and 0.125 M of H: was prepared and allowed to reach equilibrium in a 1.00 L flask. The equilibrium concentration of H:0 is 0.182 M. Calculate the Ke of the reaction at constant: temperature.
(a) Determine whether the value of K of an equilibrium system will be affected or unaffected by the following changes: Changes Value of K (affected or unaffected) (Decreasing the temperature (u) Increasing the pressure of the system (uD Decreasing the concentration of reactant (b) The following graph represents the amount of CO formed at two different temperatures, and Ts. The reaction as shown below: Cts) + H,O(g) = Co(g) + H:(g) T 385 K T = 335 K time Based on the graph, determine whether the forward reaction is endothermic or exothermic. Predict if the concentration of H.O gas increases, decreases or remains the same if the temperature is decreased to 298 K. Explain. A mixture of 0.350 M of H:0, 0.125 M of CO and 0.125 M of H: was prepared and allowed to reach equilibrium in a 1.00 L flask. The equilibrium concentration of H:0 is 0.182 M. Calculate the Ke of the reaction at constant: temperature.
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter13: Chemical Equilibrium
Section: Chapter Questions
Problem 123CP: A sample of gaseous nitrosyl bromide (NOBr) was placed in a container tiued with a frictionless,...
Related questions
Question
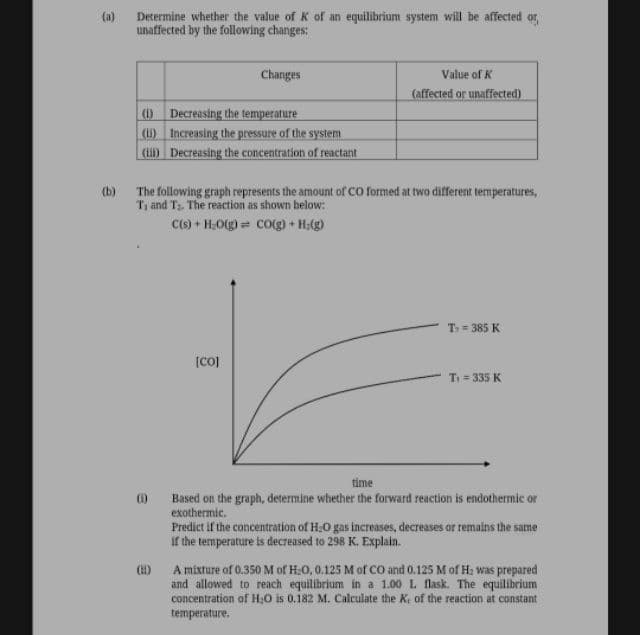
Transcribed Image Text:(a) Determine whether the value of K of an equilibrium system will be affected or,
unaffected by the following changes:
Changes
Value of K
(affected or unaffected)
(O Decreasing the temperature
(1) Increasing the pressure of the system
() Decreasing the concentration of reactant
(b) The following graph represents the amount of CO formed at two different temperatures,
T; and T. The reaction as shown below:
C() + H,O(g) = co(g) + H(g)
T = 385 K
[co)
T = 335 K
time
Based on the graph, determine whether the forward reaction is endothermic or
exothermic.
Predict if the concentration of H:0 gas increases, decreases or remains the same
if the temperature is decreased to 298 K. Explain.
A mixture of 0.350 M of H:0, 0.125 M of CO and 0.125 M of H: was prepared
and allowed to reach equilibrium in a 1.00 L flask. The equilibrium
concentration of H;0 is 0.182 M. Calculate the Ke of the reaction at constant
temperature.
(H)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning