Q: Provide the correct IUPAC name for the compound shown here. 2- 1- 3- 4- 5-)6- tert- iso tri) di cycl...
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly repos...
Q: What is the most probable speed in m/s for molecules of a certain gas with a molar mass of 27.2 u at...
A: The most probable speed is the speed that acquired by most of the molecules of a gas. Here given, ...
Q: Which equation represents a redox equation?
A: Redox reaction: The reaction in which oxidation and reduction take place simultaneously. Oxidation: ...
Q: The reaction between copper and sulfur is represented by the chemical equation shown below. What is ...
A:
Q: 5) Discuss the sources of error in your experiment. 6) List three practical applications of MgO and ...
A: 1,2,3 and 4 is correct. 5. Sources of error a. At the time of measuring weight , fault in digital d...
Q: The test samples are water, cyclohexane, cyclohexene and toluene. Drops of a dye was added to each t...
A: In the test samples water,cyclohexane,Cyclohexene,toluene, drop of a dye is added to each sample.
Q: In the reaction HzCоз+ H20 - нсоз + Нзо", the Brensted acids are Н20 and Hзo* H30* and H2CO3 О нсоз ...
A:
Q: What is the proper cell notation for the reaction: Cu + 2Fe^3+ = Cu^2+ + 2Fe^2+
A:
Q: How many signals would you observe in the 1H NMR spectra of these molecules? Label all the signals w...
A: The number of signals of a molecule in the proton NMR spectrum is equal to the number of different t...
Q: What is the pH of the buffer that is composed of 0.10 M Na2HPO4 and 0.15 M KH2PO4? (Ka1=7.5 x 10-3, ...
A:
Q: Androstenone is a constituent of male body odor. Various individuals perceive it as sweet, offensive...
A:
Q: the molarity of cl- in an aquoues solution which was %w/v 5.85% naCl , 11.1% cacl2 and 5.35% nh4cl w...
A: Given, %w/v of NaCl = 5.85% %w/v of CaCl2 = 11.1% %w/v of NH4Cl = 5.35% What is the molarity of Cl...
Q: 3. (a) The structures of [14]-annulene and [18]-annulene are as shown below. H H. [14]-Annulene [18]...
A: Aromatic compounds are those compounds which obey Huckel's rule and has (4n+2) pie electrons.
Q: For each compound shown below, state how many major products will be formed in a hydration rea and H...
A:
Q: 63. The figure below is the structural formula of O cyclohexane O cyclohexene O cyclopentane
A:
Q: assuming
A: According to the question, After 53.0 min, 16.0% of a compound has decomposed. We need to determine ...
Q: Find the solubility of AgCl in pure water, 0.10M KNO3, and 0.10M NaCl
A: AgCl is sparingly soluble salt , its solublity in absence of common ion " S " mol/L , in presence...
Q: How many moles of citric acid is needed react with 0.74 moles of baking soda
A:
Q: 70. Using the figure below: Identify the part of nos. 6 and 7 respectively in the set-up. 16 condens...
A: Option C is correct answer, Nos.6 and nos. 7 water inlet and water outlet respectively.
Q: The reaction of 8.99 g of Cl2 with 2.20 g of P4 produces 4.12 g of PCI5. What is the percent yield? ...
A:
Q: 66. Using the picture below, describe the test sample placed in the watch glass. White crystalline p...
A: The nature of above compound is decided on the basis of its physical appearance.
Q: 62. In the process of recrystallization, what is the compound that undergo in this process?" Benzoic...
A: The correct option is:
Q: Help in this yes or no activity Questions Can Aldehydes can undergo tautomerization reaction...
A: I have attached the solution of Yes/No questions below:
Q: Check my work 10 attempts left Be sure to answer all parts. Santalbic acid, a fatty acid isolated fr...
A: Sigma bonds are formed by head on overlap of two orbitals . With development of ionic character in c...
Q: 35. The stationary phase, as well as mobile phase, are liquid in Adsorption column chromatography Pa...
A: Answer: Chromatography is a separation technique, in which we separate the components of a mixture b...
Q: species oxidation state of highlighted atom H3O+ (aq) Cu2+ (aq) O2 (g) CrO2−4 (aq)
A: The given molecules are either in neutral or ionic form. Hence we can determine the oxidation state ...
Q: What is hydrogenation
A: What is hydrogenation ?
Q: /hat is the approximate pH of a solution X that gives the following responses with the indicators sh...
A: The correct option is:
Q: Steam reforming of methane ( CH, ) produces "synthesis gas," a mixture of carbon monoxide gas and hy...
A:
Q: How much heat is required to convert 40.0 g of ice at -10.0 oC to 100.00 oC? The specific heat of ic...
A:
Q: The system releases 1705 kJ of heat into its surroundings. A work of 3780 kJ is done by the system o...
A: Given: System released heat = 1705 KJ. And work is done by the system on the surrounding = 3780 KJ. ...
Q: A 0.83 mol of PCl5 is placed in a 1.0 L container and allowed to reach equilibrium at a given temper...
A:
Q: In the following reaction, oxygen is the excess reactant. (8 points) SiCl4 + O2 → SiO2 + Cl2 The t...
A: From Trail:1 Case:1 Let SiCl4 is limiting: Therefore, The amount of SiO2 that would be pr...
Q: Write appropriate K expressions (including the value of the constant from Harris Appendix A or B) an...
A: Given:
Q: num.. OD D 1/5 Ammonia will decompose into nitrogen and hydrogen at high temperature. An industrial ...
A: Given, Ammonia decomposes to nitrogen and hydrogen at high pressure, In 5.0L of vessel, the initial ...
Q: Consider the reaction. 2 Fe,0, 4 Fe +302 AHixn +824.2 kJ The formation of 58.0 g of Fe results in
A: As per the balanced reaction- Generation of 4 mol Fe requires 824.2 kJ energy. So, Energy absorbed =...
Q: A sample of water with a mass of 23.31 g and an initial temperature of 367.67 K loses 8615 Joules. W...
A: Given Mass of water ( m ) = 23.31 gram Release heat ( Q ) = 8615 joule Initial Temperature ( T1 )...
Q: Provide an arrow-pushing mechanism for the following reaction to get to the product shown. Ignore st...
A: Answer: This is the combination of nucleophilic substitution reaction of alcohol and electrophilic a...
Q: The specific heat of a certain type of cooking oil is 1.75 J/ (g.°C). How much heat energy is needed...
A: We have to predict the energy needed.
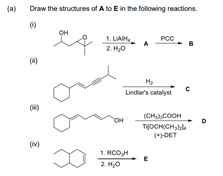
Q: What is the Synthesis for the following reaction! Thank you!
A:
Q: What is the molar mass (in g/mol) of the unknown compound?
A:
Q: Ammonia has been studied as an alternative "clean" fuel for internal combustion engines, since its r...
A: Answer: Equilibrium constant for a reaction is the ratio of the active masses of products and reacta...
Q: In antifreeze, ethylene glycol (dissolved in water) is 44% volume per volume, Antifreeze has a densi...
A: Given: %(v/v) of ethylene glycol = 44 % The density of solution = 1.23 g/mL. The density of ethylene...
Q: The phase diagram for carbon indicates the extreme conditions that are needed to form diamonds from ...
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only. ...
Q: In än atom, what is the maximum number of electrons that can have the following quantum numbers? (a)...
A:
Q: 32. refers to the formation of a chemical bond between two or more atoms, molecules, or ions to give...
A: Please find your solution below : A chemical bond is a force of attraction that holds the atoms toge...
Q: Identify one pro and one con for the agh observation (visual and UV/Vis)
A: A question based on UV-Visible spectroscopy that is to be accomplished.
Q: Determine the cell potential of the following system: a)Pb/PbSO4//Pb2+/Pb b) Fe2+/Fe3+//MnO4-/Mn2+
A:
Q: Subject: Environmental Science Diagram an atom of nitrogen (which consists of 7 protons, 7 neutrons...
A: Atomic number of an element is the number of proton present in nucleus and mass number of an element...
Step by step
Solved in 2 steps with 2 images

- Heptanedial react with KOH (aq). What is the possible reaction products. Some inducate that a cyclic product is formed while other indicate a linieer molecule.???Provide the mechanism of the redica reaction below (b)Which would you think would be a strongerinteraction and why: an interaction between a sodium ion and thepartial negative charge on the oxygen in ethanol (CH3CH2OH), orthe interaction between two ethanol molecules?
- I'm curious, can a decyclization or some chemical reaction to eliminate sulfur take place here, possibly forming SO2? and besides that, I would like you to give me examples of deciclizations of heterocycles, if they are possible, I honestly don't knowOChem reaction scheme help... Please provide the structures for the products obtained in the reaction schemes shown in the attaced image. Discuss the truth of the following statement. Explain why it is true or false Every SN1 reaction produces racemic mixtures in the products

