Q: Determine the oxidation state for Sodium Iodide, NaI
A: The oxidation state (OS) of an element corresponds to the number of electrons, e-, that an atom…
Q: Gnraphite and iodine are non-metals but they shine. Explain it withlsuitable yeasons.
A: Periodic table mainly consists of metals, non metals and metalloids. Metals are present on the left…
Q: Choose one of the questions you circled above and explain how element could be used as a substitute…
A:
Q: Identify compound as: Ionic, molecular, acid. Dissociate them into ions but leave molecule…
A: An ionic solid is a solid in which the constituent particles are ions. The positively and negatively…
Q: In what category is the substance germanium?
A: There are four statements about substances of germanium ..... 1) Covalent network solid. 2)…
Q: Write a balanced chemical equation for thereaction of aluminum with oxygen to produce aluminumoxide.
A: A balanced chemical equation is defined as the equation where equal numbers of total individual…
Q: Oxygen oxidizing Sulfide [ Choose ] Superoxide [ Choose ]
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Which choice incorrectly identifies the oxidation number (O.N.) for the given species?
A: Oxidation number is the formal charge on an atom in a compound or an ion.
Q: Why is there only a double bond between the oxygen and carbon but not the chlorine?
A: We have to answer why only a double bond is formed between the carbon and oxygen atoms but not…
Q: Hydrogenic ions have..
A: 1) Hydrogenic ions are ions which contains only one electron and it is similar to hydrogen.…
Q: Define milliequivalent
A: An equivalent is the quantity of a compound or substance which reacts with a given amount of another…
Q: 2) Does sodium hypochlorite react with either acetic acid and/or sodium hydroxide? If so, write out…
A: Sodium hypochlorite is sodium salt of hypochlorous acid. It is used as bleach.
Q: Aluminum containers are frequently used to transport trioxonitrate(v) acid because Aluminum are…
A: Aluminum containers are frequently used to transport trioxonitrate(v) acid because Aluminum are…
Q: WGener | and the US. what is the formula ionic compound for the formed by ma and iodine? agresiom
A: When two or more different atoms Combine, they give different type of compounds with different…
Q: List Oxy Compounds of Phosphorus?
A: The elements of group 13 to 18 in the long form periodic table form the p-block elements. They have…
Q: Supply
A: Ionic bond is formed between most electropositive element and most electronegative element Most…
Q: In aluminum fluoride (AlF) it is the aluminum that will ______ electrons
A: Note - The formula for aluminium fluoride is incorrect. Thus, we will take into consideration the…
Q: Determine the oxidation state of oxygen in Al2O3 : Include the sign (+ or -) as needed.
A: Explanation to the correct answer is given below
Q: Give me electron count and oxidation state
A: Oxidation state is charge on central metal ion.
Q: lonic compounds are usually in water.
A: Ionic compound are usually Soluble in water.
Q: Explain why tin has a sn
A: Answer - first ionization energy - the first ionization energy of an element is the energy needed to…
Q: how do you seperate startch and water. someone poure iodine on a sheet and the sheet got black…
A: Separation procedures are methods for separating two distinct phases of matter, such as liquid and…
Q: B) Enantionmers
A:
Q: Provide the balanced net ionic chemical equation between IO3- and I-
A: The net Ionic reaction for the given chemical equation can be given as: (1) The balanced reaction of…
Q: Part A What is the oxidation state of an individual sulfur atom in BaSO4? Express the oxidation…
A: Given :- Part A : BaSO4 Part B : HNO3 Part C : PO33- To.be determined :- Oxidation state of S…
Q: Oxygen and neon are both gases at room temperature. Why does oxygen combine readily with other…
A: Noble gas, any of the six chemical elements that make up Group 18 of the periodic table. The…
Q: a) b) o.
A:
Q: Oxonium ion is considered
A: The correct option has to be chosen regarding oxonium ion,
Q: What did the conducting solutions have in common?
A: Conducting solution is basically a solution that has the ability to conduct heat or electricity, or…
Q: Calculate the oxidation state of each element in:
A: FOR ASSIGN OXIDATION STATE :IN NEUTRAL COMPOUNDS ALL OXDATION NUMBERS MUST ADD UP TO ZERO HYDROGEN…
Q: Why is HF a weak acid in water while HCl, HBr, and HI are strong acids?
A: There are various factors that affect the acidic strength of an acid, e.g., Bond strength Stability…
Q: A piece of magnesium metal is placed in a solution of hydrochloric acid, resulting in the formation…
A:
Q: Borane (BH3) is a basic compound, but doesn't conduct electricity when you dissolve it in water.…
A: Arrhenius concept of acids and bases tells us that acids dissociate in water to give H+ ions and…
Q: What is tin pest, also known as tin disease?
A: Tin pest occurs when the element tin changes allotropes from its silvery metallic β form to the…
Q: oxidizing agent an
A:
Q: Using VSEPR method identify SiCl3F and Cl2O
A: Bond angle is decreased by presence of lone pairs. Both molecule has sp3 hybridised and tetrahedral…
Q: Oxygen belongs to family number—— and forms a(n)——-
A: Find family number of oxygen .
Q: below
A: Na2S dissociates in water to it's ions
Q: What must be present in an aqueous solution for it to conductan electric current? What general…
A: The aqueous solution conducts the electric current.
Q: oxidizing agent is:
A: Since you have asked multiparts, we will solve the first three subparts for you. If you want any…
Q: How should I combine two equations when iodine and iodide are involved?
A: In this question, we have to find out the correct answer of given problem by the help of the…
Q: formula of the oxidizing agent
A:
Q: Whats the name of phosphorus and sodium both burnt in excess and limited oxygen
A: Oxygen reacts rapidly with Group 1 elements. All alkali metal oxides form basic solutions when…
Q: c. Describe your conclusions regarding the identities of any evolved gases. d. Write an equation for…
A: Since the gases evolved are not given, thus a general description has been provided. c) If CO2 gas…
Step by step
Solved in 2 steps with 1 images

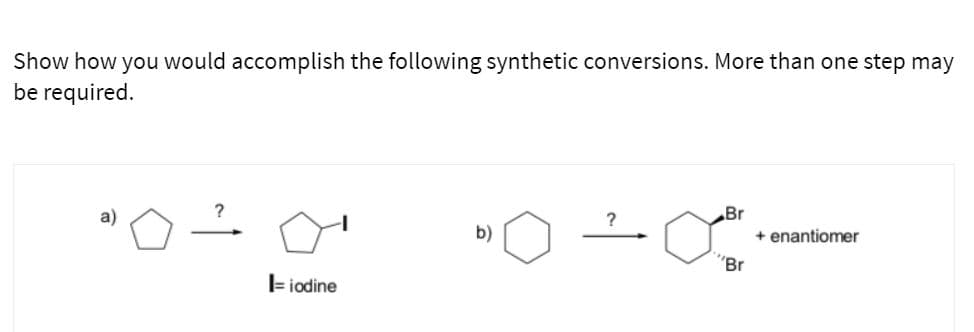
- Please help me name each alkene and specify its configuration using the E,Z system.show an arrow pushing mechanism for the reaction between α-phellandrene and maleic anhydride. Explicitly identify which reactant is the diene and which is the dienophile. Mark the chiral carbon on the α-phellandrene with a *. Draw the 4 possible products from the reaction between α-phellandrene and maleic anhydride. These should include an (R)-endo, (R)-exo, (S)-endo, (S)-exo. Which set of products would be predicted as the major product, endo- or exo-? Explain your reasoning.Show how to carry out the following transformation. Show all reagents and all molecules synthesized along the way. You do not have to use curved arrows. You are not allowed to use NBS/light or Br2/light. You MUST use SOCl2/pyr. for this drawing
- What reagents are necessary for the following conversions? show the intermediate(s) formed. When stereochemistry is specified in the product, be sure to use reagents that form the desired enantiomer.Please help! Provide the reagent needed to complete each transformationFill in the box with the reagents that could be used to accomplish the transformation shown
- Provide a reagent that will complete the transformations.Draw the structure of the major product. If the answer for the product is a mixture of enantiomers draw one of the enantiomers and write "+ enantiomer." If the answer is a mixture of diaseteromers, draw all of them. Show arrow pushing mechanism and provide reasoning for each.Identity reagents that can be used to accomplish each of the transformations show below
- I know that there is a SN1 and E1 reaction that would give different products but I have only seen it with cyclohexane, will it differ with pentane?If dehydration of 2-methylcyclohexanol were to occur under an E2 mechanism, draw the chair conformations of 2-methylcyclohexanol that would be necessary for the reaction to occur. 2-methylcyclohexanol is sold as a mixture of cis and trans isomersDevise a synthetic strategy to complete the following transformation

