(a) Iz(s) + 5 Cu²* (aq) + 6 H2O(1) (b) Hg²*(aq) + 21 (aq) (c) H2SO3(aq) + 2 Mn(s) + 4 H*(aq) → S(s) + 2 Mn²* (aq) + 3 H,O(I) 2 10;"(aq) + 5 Cu(s) + 12 H*(aq) - Hg(1) + I2(s) - -
(a) Iz(s) + 5 Cu²* (aq) + 6 H2O(1) (b) Hg²*(aq) + 21 (aq) (c) H2SO3(aq) + 2 Mn(s) + 4 H*(aq) → S(s) + 2 Mn²* (aq) + 3 H,O(I) 2 10;"(aq) + 5 Cu(s) + 12 H*(aq) - Hg(1) + I2(s) - -
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter4: Stoichiometry
Section: Chapter Questions
Problem 4.98PAE
Related questions
Question
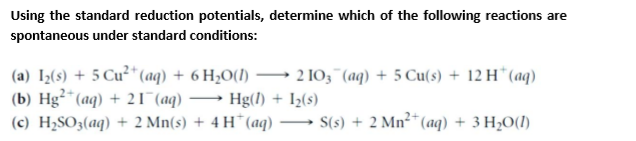
Transcribed Image Text:(a) Iz(s) + 5 Cu²* (aq) + 6 H2O(1)
(b) Hg²*(aq) + 21 (aq)
(c) H2SO3(aq) + 2 Mn(s) + 4 H*(aq) → S(s) + 2 Mn²* (aq) + 3 H,O(I)
2 10;"(aq) + 5 Cu(s) + 12 H*(aq)
-
Hg(1) + I2(s)
-
-
Expert Solution
Step 1
- I2(s) + 5Cu+2 (aq) + 6H2O → 2IO3-(aq) + 5 Cu (s) + 12 H+
E cell of the reaction = Ecathode – Eanode …..(1)
Gibbs energy is obtained by ∆G = -nFEcell. ….(2)
I2(s) → 2IO3-(aq) oxidation, Eₒ = -1.20 V
5Cu+2 (aq) → 5 Cu (s) , Reduction Eₒ = 0.34 V
E cell = Ecathode – E anode
= 0.34 – (- 1.20) = 1.54 V, which is the positive potential , hence ∆G becomes negative and reaction will be spontaneous.
Step 2
- Hg+2(aq) + 2I- (aq) → Hg(l) + I2 (s)
Hg+2(aq) → Hg (l) Reduction Eₒ = 0.789 V
2I- (aq) → I2 (s) , oxidation, Eₒ = -0.54 V
E cell = Ecathode – E anode
= 0.789 – (- 0.54) V = 1.329 V , positive E cell means negative ∆G and spontaneous reaction.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning