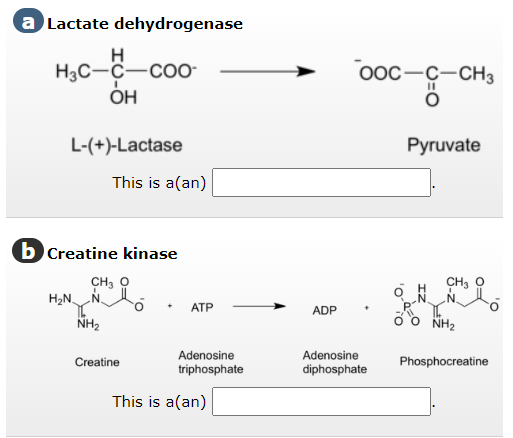
a Lactate dehydrogenase H H3C-C-COO OH L-(+)-Lactase b Creatine kinase CH, O H₂N This is a (an) NH₂ Creatine ATP Adenosine triphosphate This is a (an) ADP OOC-C-CH3 O Adenosine diphosphate Pyruvate CH, O Ỏ Ò NH, Nitt₂ Phosphocreatine
Q: Phosphorolysis is an important part of how our muscle and liver cells break down glycogen. What is…
A: Cellular metabolism refers to the entirety of biochemical reactions that occur within a cell. It…
Q: Based on the image below, select the correct statements. Note: There may be more than 1 correct…
A: Purines are a heterocyclic organic compound that has 5-member rings fused to a 6-member ring.…
Q: DESCRIPTION Carbohydrates Proteins Lipids Nucleic Acids SUBUNIT FUNCTIONAL GROUP EXAMPLES
A: Carbohydrates, lipids and proteins and nucleic acids are all types of macromolecules that are…
Q: The image below shows the ATP cycle. From the statements below, select the correct statement about…
A: ATP stands for adenosine triphosphate. It is very crucial molecule that serves as the primary energy…
Q: analyzing sequencing data, what parameters should be considered?
A: Genomic research has been transformed by improvements in DNA sequencing technology, which have made…
Q: cells of the working tissue as HCO3 - in the blood
A: Transport of Respiratory Gases :
Q: Figure 3 shows the results of an oral glucose tolerance test (OGTT) from a subject with diabetes.…
A: Oral glucose tolerance test is a test to measure the response of body to the glucose it intakes. It…
Q: Using the information in the table below, enter the correct pH that should be used to separate the…
A: Amino acids are organic compounds that contain an amino group (-NH2) and a carboxyl group (-COOH)…
Q: 15.14) The metabolic strategy of oxidative phosphorylation is to convert the chemical potential…
A: Oxidative phosphorylation is a metabolic process that takes place in the mitochondria, specifically…
Q: Chemical potential energy stored in the reduced coenzyme, NADH or FADH2 is converted to chemical…
A: A catabolic reaction refers to a type of metabolic process in which complex molecules are broken…
Q: 1. Describe the difference in the bands for the PCR and RT-PCR from the CD4+ T cells (ie. Make an…
A: PCR is a biochemical technique that is used for target-specific amplification of DNA. dsDNA is…
Q: The active site of an enzyme binds specifically to a substrate. Your text mentions an early idea…
A: Enzymes are high molecular weight proteins molecules that catalyse biochemical reactions. The…
Q: Draw the biosynthetic pathway for tyrosine (anabolism)
A: Tyrosine is an polar amino acid with a hydroxyphenol side group. Tyrosine is a conditional…
Q: Which of the following is most correct about dehydration reactions? a)They often accompanied by…
A: Any reactions that involve the release of elements of water are called dehydration reactions or…
Q: The daily production of a growth-associated byproduct (P) is 250 kg/day. The continuous bioreactor…
A: A bioreactor is basically a controlled environment where the microorganisms or cells grow and hence…
Q: You are working with a 60 kDa protein that you want to visualize on a SDS-PAGE. What % separating…
A: For visualizing a 60 kDa protein in SDS-PAGE, a 12 % separating gel will be used.
Q: Which of the following fatty acids is most likely to be a solid at room temperature? OA) B) C) D) OH…
A: Fatty acids are carboxylic acids with a hydrocarbon chain ranging from 4 carbon to 36 carbons. The…
Q: 2. How are single, double and triple covalent bonds different?
A: Chemical bonds stabilize and form molecules. Covalent chemical bonds are crucial. Atoms share…
Q: how do low levels of NADH regulate the CAC?
A: CAC, also known as the Krebs cycle act as a central metabolic pathway which generates energy in the…
Q: Why is this compound achiral?
A: A plane of symmetry in the molecular framework of 1-chloro-3-methylcyclobutane causes it to be…
Q: The chemical structure of the tripeptide Met-Gin-Tyr is shown below. H₂N NH₂ Met-Gln-Tyr OH OH I
A: The attachment of covalent and non-covalent bonding of polyethylene glycol or PEG to various drugs…
Q: When milk gets sour, it curdles. What are the "curds?" Be sure to describe the "curds and what…
A: Milk can be defined as water in oil type emulsion in which fat molecules disperse in water. Milk…
Q: They want to insert a small sequence into the gene which will be read in the same frame, so that it…
A: The expression of a protein occurs in two steps. Transcription: The gene is transcribed by RNA…
Q: The effectiveness of allosteric effectors in regulating metabolic pathways is based on their ability…
A: Allosteric inhibition is the type of enzyme inhibition. In this, the working of the enzyme is…
Q: 6. Give the name of the disease that is causing the baby's symptoms. Explain why the symptoms are…
A: Glucose dehydrogenase is an enzyme that is involved in the pentose phosphate pathway that catalyzes…
Q: Using proper convention, provide the amino acid sequence for the following protein.…
A: A basic characteristic of proteins is their amino acid composition, which is essential to defining…
Q: Find a method that uses some form of HPLC for the analysis of proteins. What was the stationary…
A: Protein analysis by chromatography is a widely used technique for separating, identifying, and…
Q: Explain the Rf value obtained on the basis of the structure of amino acids use. Explain in 1-3…
A: The Rf value, also known as the retention factor, is a measure of the position of a component in a…
Q: protein purification through hydrophobic column chromatography
A: In hydrophobic interaction chromatography (HIC), molecules are separated based on how hydrophobic…
Q: Draw the structure and titration curve of Cysteine by hand. Make sure to label the x- and y-axes,…
A: Amino acid has a typical structure. It has a central Carbon atom called (Cα). To it 4 groups are…
Q: Nicotiana tabacum cells are cultured to produce a polysaccharide gum. The reactor used is a…
A: The system or any device which provides a controlled environment for the growth of the…
Q: ii) What is the name of the monosaccharide that is produced in the digestion of starch? glucose…
A: Carbohydrate is a macronutrient which is the main nutrient utilized by our body for the purpose of…
Q: For the scenario below relating to Glycogenolysis, please explain how glucose release would be…
A: Glycogen is a macromolecule which acts as a storage of glucose in our body. It is a highly branched…
Q: You are a liver cell and have just been given 1 million C25:0 fatty acid molecules (Note: These…
A: As a liver cell, you have the ability to generate net production of glucose through a process called…
Q: What are 5 regulation of carbohydrate metabolism in the liver
A: The liver controls carbohydrate metabolism and glucose homeostasis. Understanding liver carbohydrate…
Q: Why is this a meso conpound?
A: Meso compounds are are those that are not enantiomers. This means that the mirror image of a meso…
Q: Explain why breathing in a brown bag paper helps with hyperventilation. Explain using Le Chatelier's…
A: Le Chatelier's Principle explains why breathing into a brown paper bag can help manage…
Q: Calculate the pH of the following substances from the [H*]. O Blood plasma, 39.8 nM.
A: Blood is an essential fluid in the body that transports various products from one area to another. A…
Q: Which of the following are more likely to promote the activity of gluconeogenesis rather than…
A: Gluconeogenesis is the process by which glucose is synthesized from non-carbohydrate precursors,…
Q: What is the indication of violet blue color in biuret test?
A: Proteins, essential macromolecules in living organisms, play crucial roles in various biological…
Q: Trend observed in graph and conclusion about the effect of temperature on enzyme activity. In your…
A: Enzymes are biological catalysts. They increase the rate of the reactions they catalyze. Most…
Q: What is the thermodynamic driving force behind the formation of a peptide bond and how does it…
A: A dehydration reaction is a chemical reaction where the formation of a chemical bond involves the…
Q: This technique does not provide atomic-level structures of proteins a)Circular dichroism…
A: The body needs proteins as key macromolecules. They carry out a variety of biological processes. A…
Q: ) In the thin layer chromatography of practical 5 the chromatography tank contained a mixture of…
A: All chromatography experiments have two components: The stationary phase: immobile and binds…
Q: B. Describe in complete sentences how you would identify a target DNA cleavage site for CRISPR-Cas9…
A: A target DNA cleavage site would have PAM sites - 5' NGG flanking both sides of the target.In the…
Q: Shown below is the response of a regulatory motif to a signal S. What does the response show…
A: The signal is provided here in a step-wise manner. Look at the green part of the graph. The signal…
Q: Which of the following pathways will split water and produce oxygen Select one: a. Gluconeogenesis…
A: Photosynthesis is the process by which green plants, algae, and some bacteria convert light energy…
Q: and Carbohydrate Metabolis the B vitamins below, indicate w processes each vitamin is involv…
A: Glycolysis is the metabolic pathway by which glucose is converted into two molecules of Pyruvate.…
Q: 14. What are the functions of nucleic acids? Please explain
A: Nucleic acids store, transmit, and express genetic information. They're essential to life. Nucleic…
Q: 14. In competitive inhibition, an inhibitor— binds reversibly at the active site. binds to both…
A: Enzymes are biological catalysts that increase the rate of chemical reactions in living organisms…
Step by step
Solved in 3 steps

- GTP or ATP is produced during the conversion of isocitrate into ketoglutarate succinyl CoA into succinate fumarate into malate malate into oxaloacetateRadiolabeling with 14C-Glutamate Describe the labeling pattern that would result from the introduction into the TCA cycle of glutamate labeled at Cy with 14C.Consider the synthesis and degradation of tyrosine as shown in Figures 25.37, 25.38, and 25.48 to determine where the carbon atoms in PEP and erythrose-4-P would end up in acetoacetate and fumarate.
- How many ATP may be produced from 1 mole of the following compounds on complete oxidation using the glycerol-3-phosphate shuttle? COMPOUNDSa.) Mannoseb.) Dihydroxyacetone phosphatec.) Citrated.) Malatee.) SuccinateThe malate–aspartate shuttle yields about2.5 moles of ATP for each mole of cytosolic NADH. Why doesnature use the glycerol–phosphate shuttle, which yields only about1.5 moles of ATP?Select the following enzymes that utilize a mechanism where an enediol intermediate is formed. Check all that apply: a)phosphoglucoisomerase b)triose phosphate isomerase c)aldolase d) glyceraldehyde 3-P dehydrogenase e) hexokinase
- How many molecules of ATP are produced from the complete metabolism of 2.12g of triacylglycerol containing two stearic acid chains and one palmitic acid chain? Assume that all electrons of cytosolic NADH are transferred through the dihydroxyacetone phosphate/glycerol 3-phosphate shuttleSelect the following enzymes that utilize a mechanism where ATP serves as the phosphate donor. Select ALL that apply: a) glyceraldehyde 3 phosphate dehydrogenase b) hexokinase c) aldolase class 1 d) PFK e) phosphoglucoisomeraseCopper is a cofactor in several enzymes, including lysyl oxidase and superoxide dismutase.Ceruloplasmin, a deep-blue glycoprotein, is the principal copper-containing protein in blood. It isused to transport Cu2+ and maintain appropriate levels of Cu2+ in the body’s tissues.Ceruloplasmin also catalyzes the oxidation of Fe2+ to Fe3+, an important reaction in ironmetabolism. Because the metal is widely found in foods, copper deficiency is rare in humans.Deficiency symptoms include anemia, leukopenia (reduction in blood levels of white blood cells),bone defects, and weakened arterial walls. The body is partially protected from exposure toexcessive copper (and several other metals) by metallothionein, a small, metal-binding protein thatpossesses a large proportion of cysteine residues. Certain metals (most notably zinc and cadmium)induce the synthesis of metallothionein in the intestine and liver.In Menkes syndrome intestinal absorption of copper is defective. How can affected infants…
- 3-Phosphoglycerate is a metabolite in glycolysis. Which glycolytic enzyme generates this metabolite?Von Gierke’s disease is also known as glycogen storage disease type I. Patients with von Gierke’s disease lackglucose 6-phosphatase activity. Two prominent symptoms of this disorder are fasting hypoglycemia and lactic acidosis (elevated lactate levels in the blood), especially during strenuous exercise. Explain why these symptoms occur. What chemical reaction does this enzyme catalyze? Which pathways involve this enzyme? Lacking thisthe enzyme will cause impairment of which pathways?• Pls consider what pathways are affected by Von Gierke’s disease. Include in your explanation involving Cori’s cycle. can you please do not write by your hand? I mean computer if you can. thank youUsing the glycerol-3-phosphate shuttle, determine how many ATP can be produced from one mole of each of the following compounds on complete oxidation?a.) Mannoseb.) Dihydroxyacetone phosphatec.) Citrated.) Malatee.) Succinate



