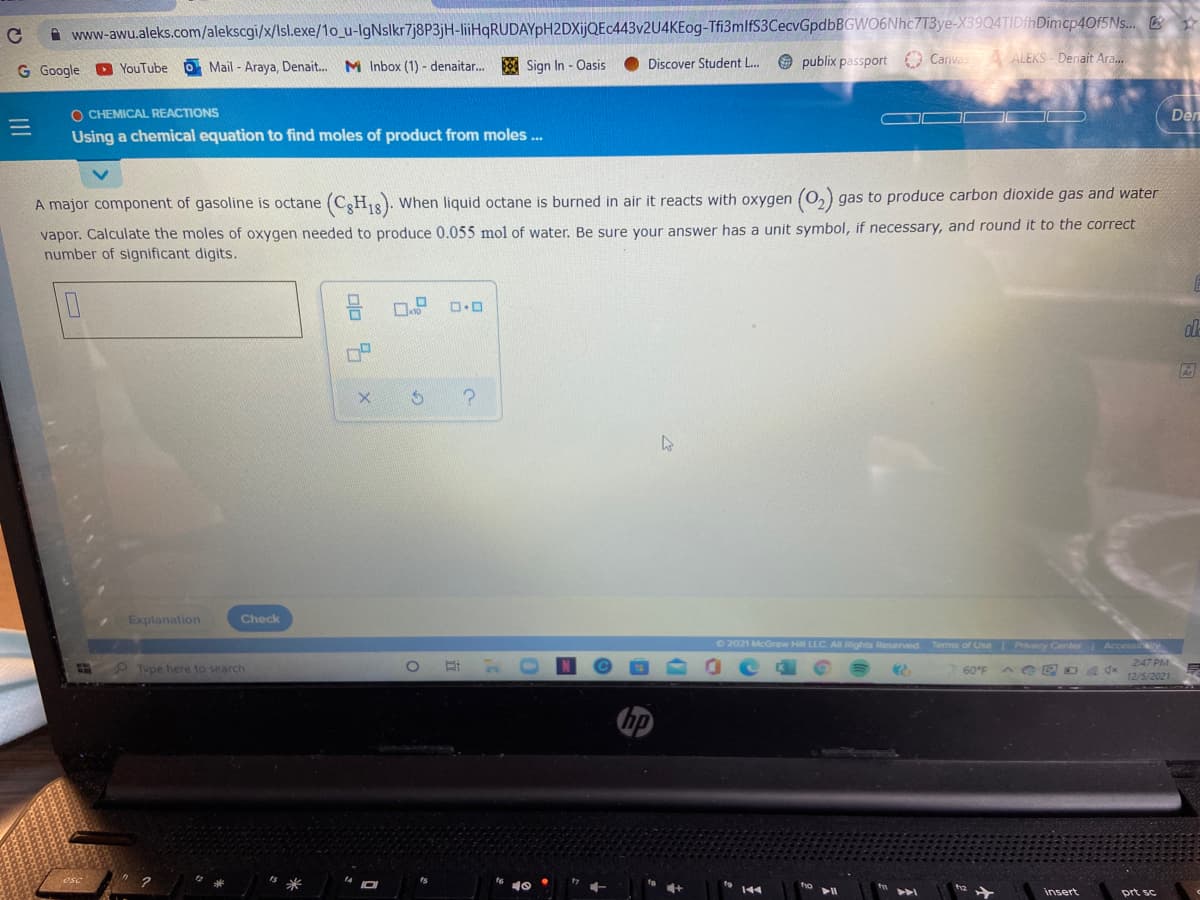
A major component of gasoline is octane (CgH18). When liquid octane is burned in air it reacts with oxygen (0,) gas to produce carbon dioxide gas and water vapor. Calculate the moles of oxygen needed to produce 0.055 mol of water. Be sure your answer has a unit symbol, if necessary, and round it to the correct number of significant digits.
A major component of gasoline is octane (CgH18). When liquid octane is burned in air it reacts with oxygen (0,) gas to produce carbon dioxide gas and water vapor. Calculate the moles of oxygen needed to produce 0.055 mol of water. Be sure your answer has a unit symbol, if necessary, and round it to the correct number of significant digits.
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter34: Particle Size Determination
Section: Chapter Questions
Problem 34.10QAP
Related questions
Question
Transcribed Image Text:A www-awu.aleks.com/alekscgi/x/lsl.exe/1o_u-lgNslkr7j8P3jH-liiHqRUDAYpH2DXijQEc443v2U4KEog-Tfi3mlfS3CecvGpdbBGWO6Nhc7T3ye-X39Q4TIDfhDimcp40f5Ns... E
Mail - Araya, Denait. M Inbox (1) - denaitar. Sign In - Oasis
O Discover Student L..
publix passport Canvas ALEKS - Denait Ara.
G Google O YouTube
O CHEMICAL REACTIONS
Den
三
Using a chemical equation to find moles of product from moles..
A major component of gasoline is octane
(C3H18). When liquid octane is burned in air it reacts with oxygen (0,) gas to produce carbon dioxide gas and water
vapor. Calculate the moles of oxygen needed to produce 0.055 mol of water. Be sure your answer has a unit symbol, if necessary, and round it to the correct
number of significant digits.
dl-
Explanation
Check
O 2021 McGraw Hi LLC All Rights Reserved Terms of Use Prvacy Center AccessiY
247 PM
Type here to search
60°F
12/5/2021
hp
esc
14
4+
144
insert
prt sc
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning