A mixture of H2, N2, andNH3 with molar concentrations 3.0 x 10-³ mol/L, 1.0 x 10- 3mol/L and 2.0 x 10-³ mol/L, respectively, was prepared and heated to 500 K, at which temperature Kc = 62 for the reaction : N₂(g) + 3H₂(g) → 2NH3(g). Deduce whether ammonia tends to form or decompose. Equilibrium expression Value of reaction quotient Qc Relationship between Qc and Kc NH3 will under these conditions
A mixture of H2, N2, andNH3 with molar concentrations 3.0 x 10-³ mol/L, 1.0 x 10- 3mol/L and 2.0 x 10-³ mol/L, respectively, was prepared and heated to 500 K, at which temperature Kc = 62 for the reaction : N₂(g) + 3H₂(g) → 2NH3(g). Deduce whether ammonia tends to form or decompose. Equilibrium expression Value of reaction quotient Qc Relationship between Qc and Kc NH3 will under these conditions
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter16: Principles Of Chemical Reactivity: The Chemistry Of Acids And Bases
Section16.7: Calculations With Equilibrium Constants
Problem 16.7CYU: The weak base, CIO (hypochlorite ion), is used in the form of NaCIO as a disinfectant in swimming...
Related questions
Question
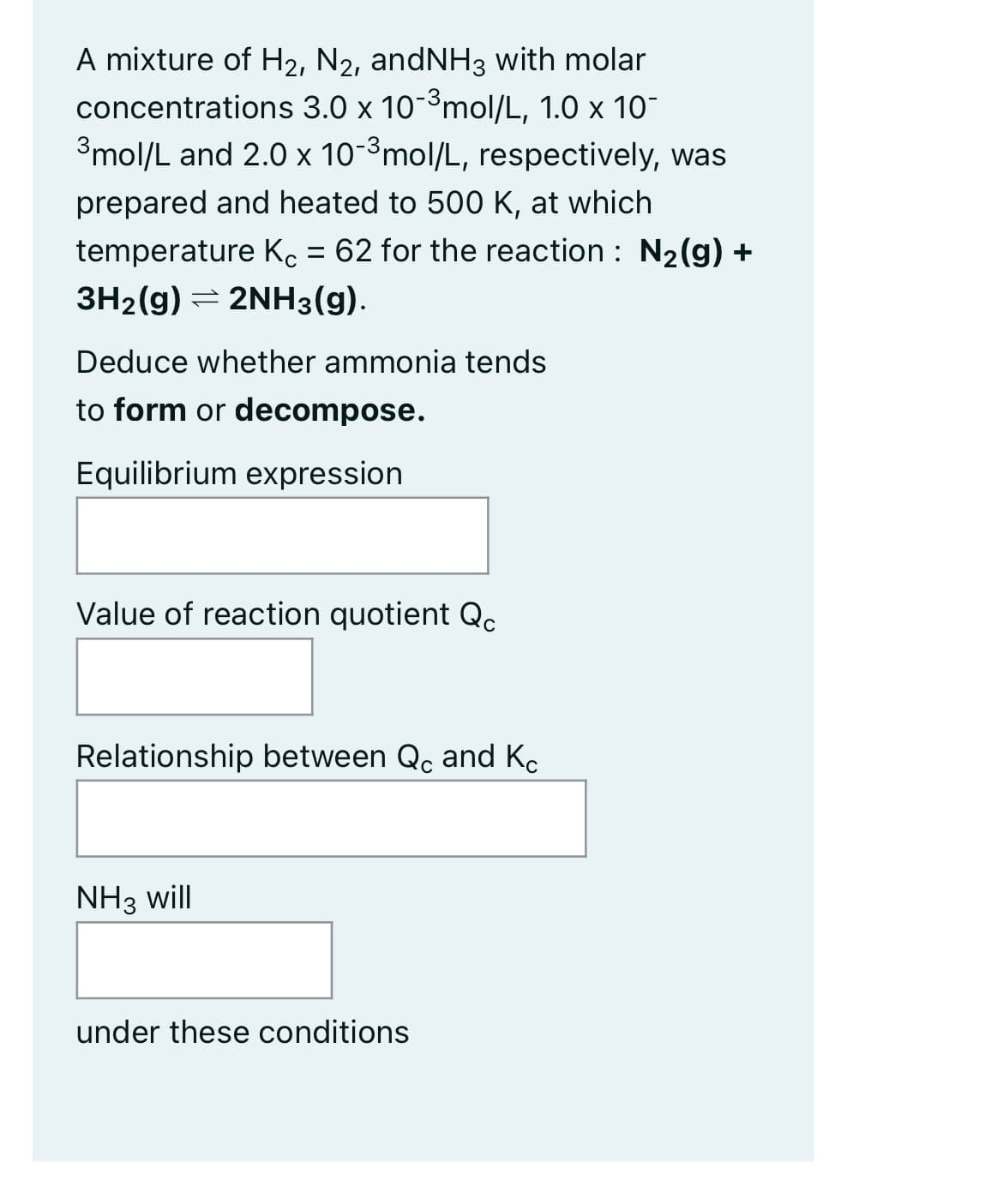
Transcribed Image Text:A mixture of H₂, N2, andNH3 with molar
concentrations 3.0 x 10-³ mol/L, 1.0 x 10-
3mol/L and 2.0 x 10-³ mol/L, respectively, was
prepared and heated to 500 K, at which
temperature Kc = 62 for the reaction : N₂(g) +
3H₂(g) = 2NH3(g).
Deduce whether ammonia tends
to form or decompose.
Equilibrium expression
Value of reaction quotient Qc
Relationship between Qc and Kc
NH3 will
under these conditions
![Phenol, C6H5OH, is a weak organic acid:
C6H5OH(aq) = С6H5O¯(aq) + H+ (aq)
Although somewhat toxic to humans, it is
widely used as a disinfectant and in the
manufacture of plastics. What is the equilibrium
hydrogen ion concentration in a 0.16M acid
solution? What is the pH of the solution? Ka of
C6H5OH = 1.3 x 10-10
Kc 1.3 x 10-10-
C6H50*H*
Initial
0
0
Change
Equilibrium
[H+] =
pH=
C6H5OH
0.16M](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F7f1e4f7e-3ee3-4328-9071-c5dd7af83341%2Fe3e8c3bc-93f0-40a4-a18b-a5e988ded213%2Fodlsfro_processed.jpeg&w=3840&q=75)
Transcribed Image Text:Phenol, C6H5OH, is a weak organic acid:
C6H5OH(aq) = С6H5O¯(aq) + H+ (aq)
Although somewhat toxic to humans, it is
widely used as a disinfectant and in the
manufacture of plastics. What is the equilibrium
hydrogen ion concentration in a 0.16M acid
solution? What is the pH of the solution? Ka of
C6H5OH = 1.3 x 10-10
Kc 1.3 x 10-10-
C6H50*H*
Initial
0
0
Change
Equilibrium
[H+] =
pH=
C6H5OH
0.16M
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning