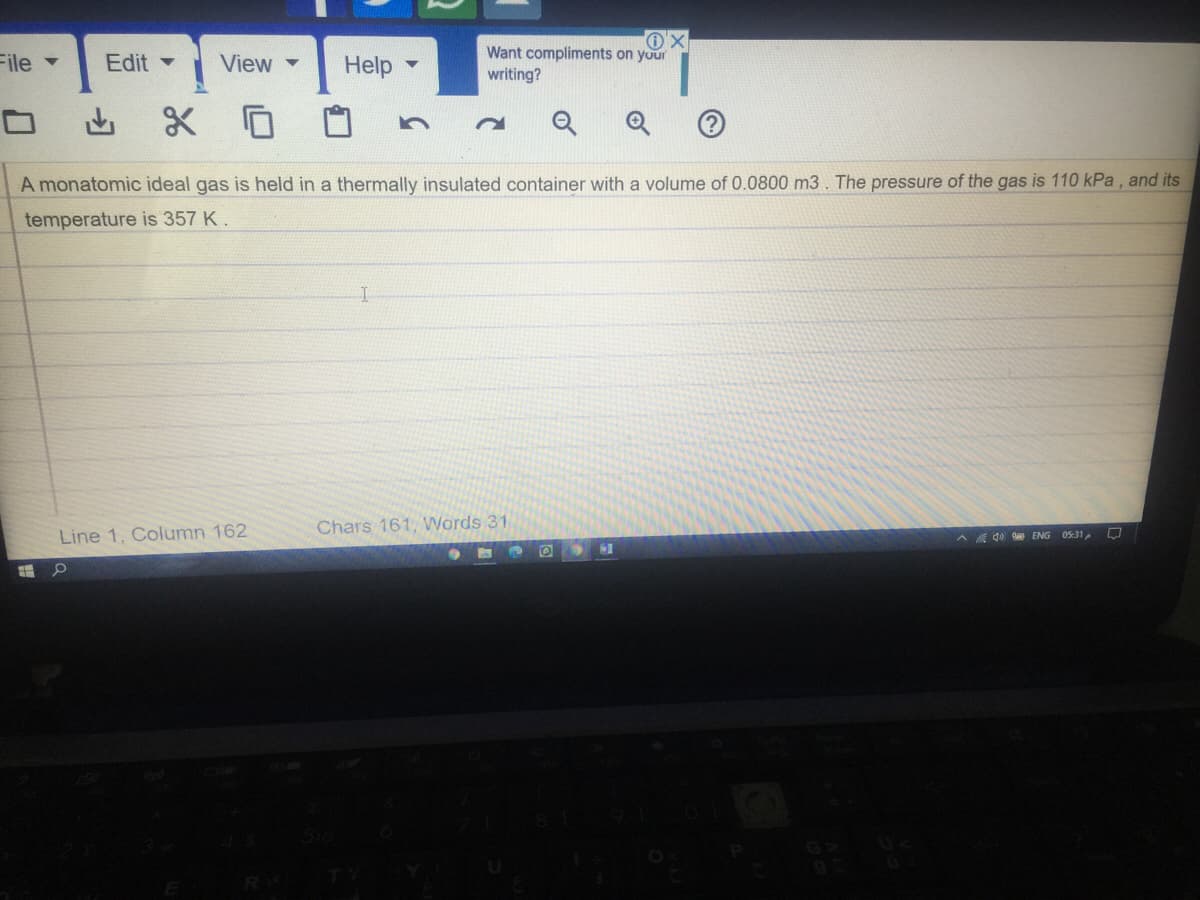
A monatomic ideal gas is held in a thermally insulated container with a volume of 0.0800 m3. The pressure of the gas is 110 kPa, and its temperature is 357 K.
A monatomic ideal gas is held in a thermally insulated container with a volume of 0.0800 m3. The pressure of the gas is 110 kPa, and its temperature is 357 K.
Chapter4: The Second Law Of Thermodynamics
Section: Chapter Questions
Problem 71AP: A system consisting of 20.0 mol of a monoatomic ideal gas is cooled at constant pressure from a...
Related questions
Question
100%
No need for explanation just find
A) what volume must the gas be compressed to increase its pressure to 150kpa
B) at what volume will the gas have 280K?
I’ll rate, please ASAP.
Transcribed Image Text:Want compliments on your
File
Edit -
View
Help
writing?
ロ
A monatomic ideal gas is held in a thermally insulated container with a volume of 0.0800 m3. The pressure of the gas is 110 kPa , and its
temperature is 357 K.
Chars 161, Words 31
Line 1, Column 162
A A 40 ENG 05:31
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you



College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College



College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College

University Physics Volume 1
Physics
ISBN:
9781938168277
Author:
William Moebs, Samuel J. Ling, Jeff Sanny
Publisher:
OpenStax - Rice University