Q: Determine the grams of protein in one cup of soup that has 140 kcalkcal with 7 gg of carbohydrate an...
A: Major sources for body nutrients are protein, carbohydrate and fats when it burns it generates en...
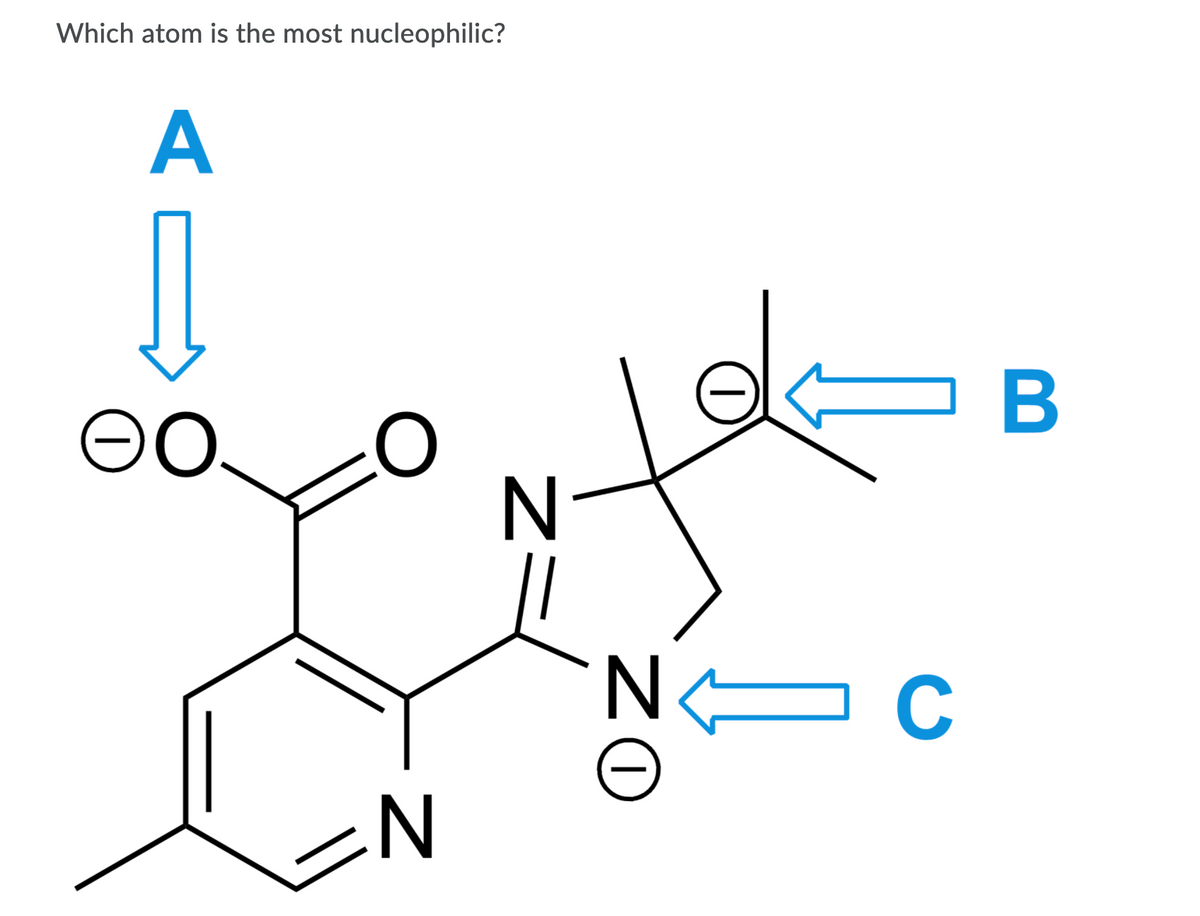
Q: Use the resonance structures of the molecule below to identify the nucleophilic sites C E D ZI
A:
Q: References Use the References to access important values if neec Fill in the compound formulas in th...
A:
Q: Draw the Lewis structures of the polyatomic ions and assign formal charges.
A: Given :- Polyatomic ions = i) NH4+ ii) CO32- To draw :- Lewis str...
Q: 1. A 10.0 mL sample of a diluted solution has a transmittance of 25.1%. Determine the connection of ...
A: The question is related to absorbance based on beer lambert's lawA= ε c L; c=concentration; L= pat...
Q: Aryl and vinyl halides are good in SN2 reaction. True O False
A: Nucleophilic substitution is an important organic reaction in which a leaving group is replaced by a...
Q: The vapor pressure of liquid methanol, CH3OH, is 100. mm Hg at 294 K. A 0.116 g sample of liquid CH3...
A: Since you have posted multiple questions, I am going to answer the first one only as per the guideli...
Q: Isotonic saline solution, which has the same osomotic pressure as blood, can be prepared by dissolvi...
A:
Q: Draw the Lewis structure of PBr,. Include all the lone pairs.
A: Phosphorus have 5 valence electrons three involve in the bond formation with bromine so it have one ...
Q: Identify the major product of the reaction below, and explain a detailed, stepwise mechanism which a...
A: Reaction of secondary alkyl halaid is with the weak nucleophile CH3O- gives SN1 mechanism. So this r...
Q: - Calculate equilibrium constant for the reaction: O,(8) + O(g) 0,(g) Given that: NO,(8) 0,(g) + NO...
A:
Q: What is the formula of 2-methylpropane? A) CH B) CH. C) C.Ha D) CH. E) CH.
A: The given organic compound is as follows: 2-methylpropane: In this organic compound the propane mean...
Q: Identify which of the following pairs of molecules are NOT classified as resonance structures. I) II...
A:
Q: (References] This question has multiple parts. Work all the parts to get the most points. Use the Re...
A: given, [OH-] = 10-pOH
Q: Consider the incomplete structure shown. Draw an alternative Lewis (resonance) structure for the inc...
A:
Q: Iron exists in nature as a mixture of predominantly three isotopes: ⁵⁴₂₆Fe (53.94 amu), ⁵⁶₂₆Fe (55.9...
A: We have to calculate the percentage of rarest of three isotopes.
Q: Compound 10: C10H10O3 || 9 8 7 6 4 3 2 1 d(2 H) d(2 H) s(3 H) s(3 H) 200 150 100 50 CR200701473NY pp...
A: All the spectroscopic method is used to find the structure of any unknown compound. The NMR spectro...
Q: statement is correct?
A: In Analytical Chemistry, an internal standard is defined as a chemical substance that is added to th...
Q: Which of the following methods results in the most sensitive method? A) All 3 methods have equal se...
A: We are given Absorbance versus concentration of aspartame (mg/mL) graph for 3 different methods A, B...
Q: General Chemistry 4th Edition AcQuarrie Rock Gallogly Universi prese he molecule cyanazine, once use...
A: Lewis structure is a method of of representing bonding pattern in a molecule. it represent how diffe...
Q: A 47 gg piece of ice at 0.0 ∘C∘C is added to a sample of water at 7.0 ∘C∘C. All of the ice melts and...
A:
Q: 3 You are given 500 ml of a pH 3 solution of HCI. What is the concentration of [CI] in this solution...
A: HCl is a strong electrolyte and strong electrolytes dissociates completely into its ions .
Q: A mixture is made by combining 1.02 lb of salt and 3.76 lb of water. What is the percentage of salt ...
A:
Q: Will the following salts alter the pH a of distilled water solution? If so, will they be acidic, bas...
A:
Q: A solution of sodium carbonate contains 5.603 g of Na2CO3 in 250.0 mL of solution. Calculate the mol...
A: The question is based on the concept of molarity. it is defined as number of moles of solute pres...
Q: What property of liquids can be used in their separation using distillation?
A:
Q: The kinetic energy of a moving object is computed as KE = V2 mv, if the velocity of a 5 kg object is...
A:
Q: Arrange the substrates in order of increasing Sn2 reactivity with NaCN: (A) Bromoethane, (B) 1-chlor...
A: To solve this problem we have to know about the reaction order of SN2 reaction for alkyl halides . W...
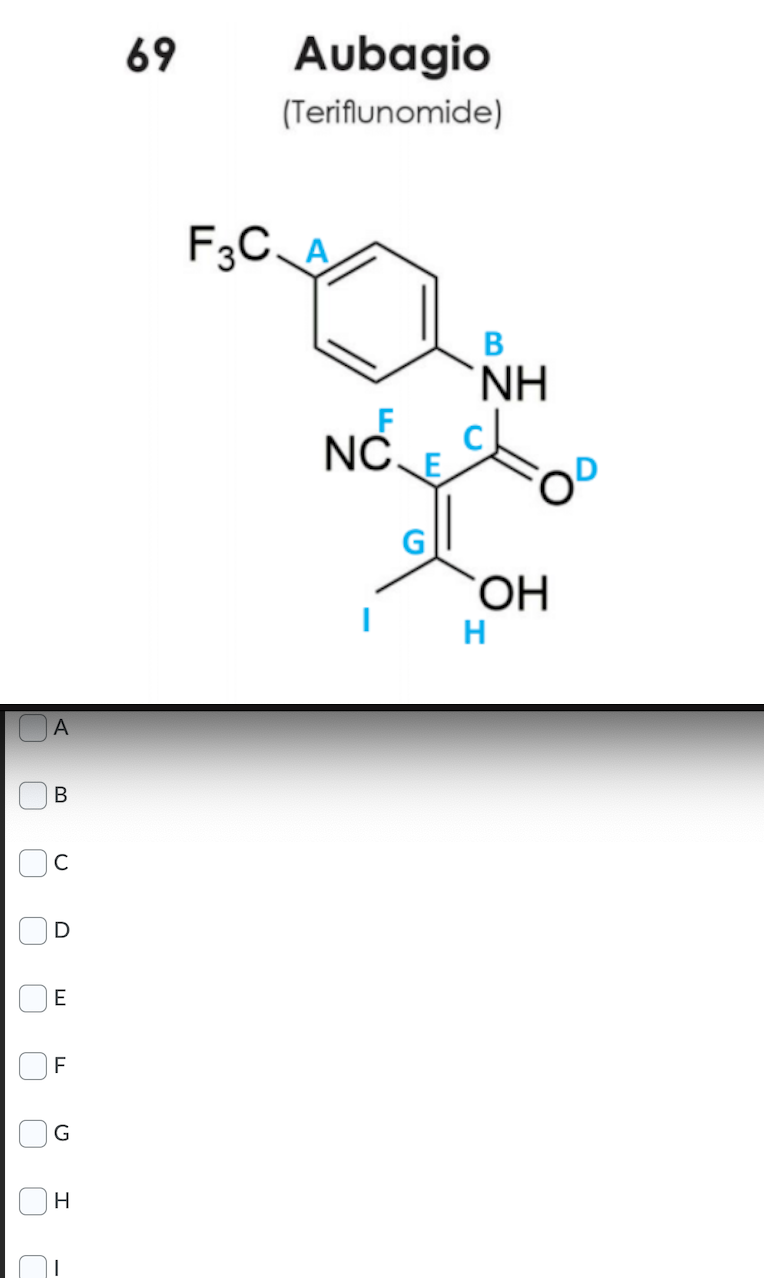
Q: Identify four electrophilic sites in the molecule below. 69 Aubagio (Teriflunomide) F3C. `NH NC. $2....
A: In this question, we will see four electrophilic sites. You can see details explanation and answer ...
Q: Compound 8: C9H10O2 7 4 3 2 1 d(2 H) d(2 H) s(1 H) s(2 H) s(3 H)
A:
Q: While driving your rental car on your vacation in Europe, you find that you are getting 14.1 km/L of...
A: While driving our rental car on our vacation in Europe, we find that we are getting 14.1 km/L of gas...
Q: Calculate the number of atoms in 12.45 grams of iron . 1.342 x 1023atoms 1.342 x 1022toms 0.223 atom...
A: The question is based on mole concept. We have to calculate number of atoms of iron.
Q: (References] Use the References to access important values if needed for this question. Taking logar...
A: The question is based on the concept of logarithmic calculations. We have to calculate log and anti...
Q: Use the References to access important values if needed for this question. The pH scale was designed...
A:
Q: You are given the following: 0.5 M KH2PO 4, 0.5 M K2HPO 4,0.5 M Tris base, 0.5 M Tris HCI and 3 M Na...
A: Interpretation: To find molecular mass of each compound given in the question and prepare the given ...
Q: Give the IUPAC name of the compound, -Br НОос
A: The question is based on the concept of IUPAC naming of the compounds. we have to write the correc...
Q: What is vulcanization of rubber and why it is useful to the construction industry.
A: Introduction : Rubber is a natural polymer and possesses elastic properties .It is also termed as e...
Q: Draw the Lewis structure of CH, Cl,. Include all the lone pairs.
A: Given :- molecular formula = CH2Cl2 To draw :- Lewis structure of CH2Cl2
Q: >Calculate pH of a solution of a 0.10 M CH3COOH solution? Ka = 1.8 X 10-5 How many grams of CH3COOh...
A:
Q: ample tub Deprot. samples Glucose Standards A B E #1 #2 O Standard/samples (r) 0.2 0.2 Nelson'scoppe...
A: The question involves the beer lambert law equation: A=ε c lA is the absorbance; c= concentrationl =...
Q: Bismuth oxide reacts with carbon to form bismuth metal: Bi2O3(s) + 3C(s)→ 2Bi(s) + 3CO(g) When 242 g...
A: 1- First you need to determine the moles of Bi2O3 : Molar mass of Bi2O3 = 465.96gmol-1 Moles = [ m...
Q: Draw the molecular models of the compounds listed below: a) 5-methyl-(2E, 4Z)-2,4-octadiene b) 5-m...
A: Draw the given structure of the following-
Q: The following reaction is at equilibrium in a sealed container. N2(g) + 3H2(g) ⟷⟷ 2NH3(g) ...
A:
Q: Calculate the number of moles in 21.9 grams of Copper(II) hydroxide, Cu(OH)2. 2.75 x 105 mol 4.05 mo...
A: given, weight of copper(II) hydroxide = 21.9 grams
Q: Gold atoms pack in a regular pattern in a three dimensional lattice in pure gold metal. Assume the e...
A: Given: The edge length, a= 0.408 nm Conversion factor : 1.00 cm = 1.00 x 107 nm or 1.00 nm = 1.00 ...
Q: Precipitation from a supersaturated sodium acetate solution. The solution on the left was formed by ...
A: Initially, the solution was a supersaturated solution at 100 oC, with a concentration of 141 g/100 m...
Q: A solution is prepared by dissolving 25.0 g of ammonium sulfate in enough water to make 100.0 mL of ...
A:
Q: (a) How many moles of C atoms are in 1 mol of sucrose (C12H22011)? (1 mol is an exact number.) mol C...
A: (a) • The values provided in the question are:- i) Number of moles of sucrose= 1 mol • Since...
Q: How many moles are in 5.27 × 1024 particles of Na2SO3? 6.75 x 1026 mol 4.75 mol 8.75 mol 8.75 x 1024...
A: We have to calculate the number of moles.
Q: For each factor (on the left list) u can choose one of the three options (on the right). U can choos...
A: Resonance explains the fact that electrons are delocalized.Delocalisef means electrons flow freely...
For 1st picture: which atom is the most nucleophilic?
options are A, B or C.
For 2nd picture: Identify four electrophilic sites in the molecule?
options are give in the pic (choose 4 of them)
Step by step
Solved in 3 steps with 3 images

- which amine is most basic and most nucleophilic?What neutral nucleophile is needed to convert C to D? Imatinib, an effective treatment for certain cancers, is prepared in one step from D.(a) Add curved arrows for each step to show how A is converted to theepoxy ketone C. (b) Classify the conversion of A to C as a substitution,elimination, or addition. (c) Draw one additional resonance structure forB.



