A sample of a mixture of oxalic acid, H2C2O4, and sodium chloride, NaCl, has a mass of 4.554 g. If a volume of 29.58 mL of 0.550-M NaOH is required to neutralize all the H2C2O4, calculate the mass percent of oxalic acid in the mixture. Oxalic acid and NaOH react according to this equation: H,C,0,(aq) + 2 NaOH(aq) → Na,C,O,(aq) + 2 H,0(€)
A sample of a mixture of oxalic acid, H2C2O4, and sodium chloride, NaCl, has a mass of 4.554 g. If a volume of 29.58 mL of 0.550-M NaOH is required to neutralize all the H2C2O4, calculate the mass percent of oxalic acid in the mixture. Oxalic acid and NaOH react according to this equation: H,C,0,(aq) + 2 NaOH(aq) → Na,C,O,(aq) + 2 H,0(€)
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter4: Reactions In Aqueous Solution
Section: Chapter Questions
Problem 49QAP: A solution of potassium permanganate reacts with oxalic acid, H2C2O4, to form carbon dioxide and...
Related questions
Question
Please answer every letter, thank u
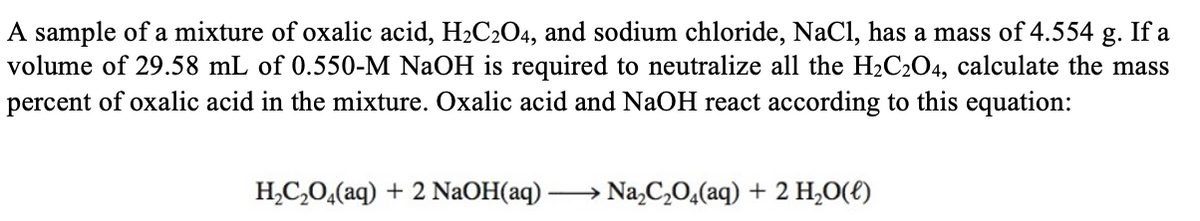
Transcribed Image Text:A sample of a mixture of oxalic acid, H2C2O4, and sodium chloride, NaCl, has a mass of 4.554 g. If a
volume of 29.58 mL of 0.550-M NaOH is required to neutralize all the H2C2O4, calculate the mass
percent of oxalic acid in the mixture. Oxalic acid and NaOH react according to this equation:
H,C,0,(aq) + 2 NaOH(aq) -
Na,C,0,(aq) + 2 H,0(E)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning