Chapter10: Organohalides
Section10.1: Names And Structures Of Alkyl Halides
Problem 2P: Draw structures corresponding to the following IUPAC names: (a) 2-Chloro-3, 3-dimethylhexane (b) 3,...
Related questions
Question
Question 10
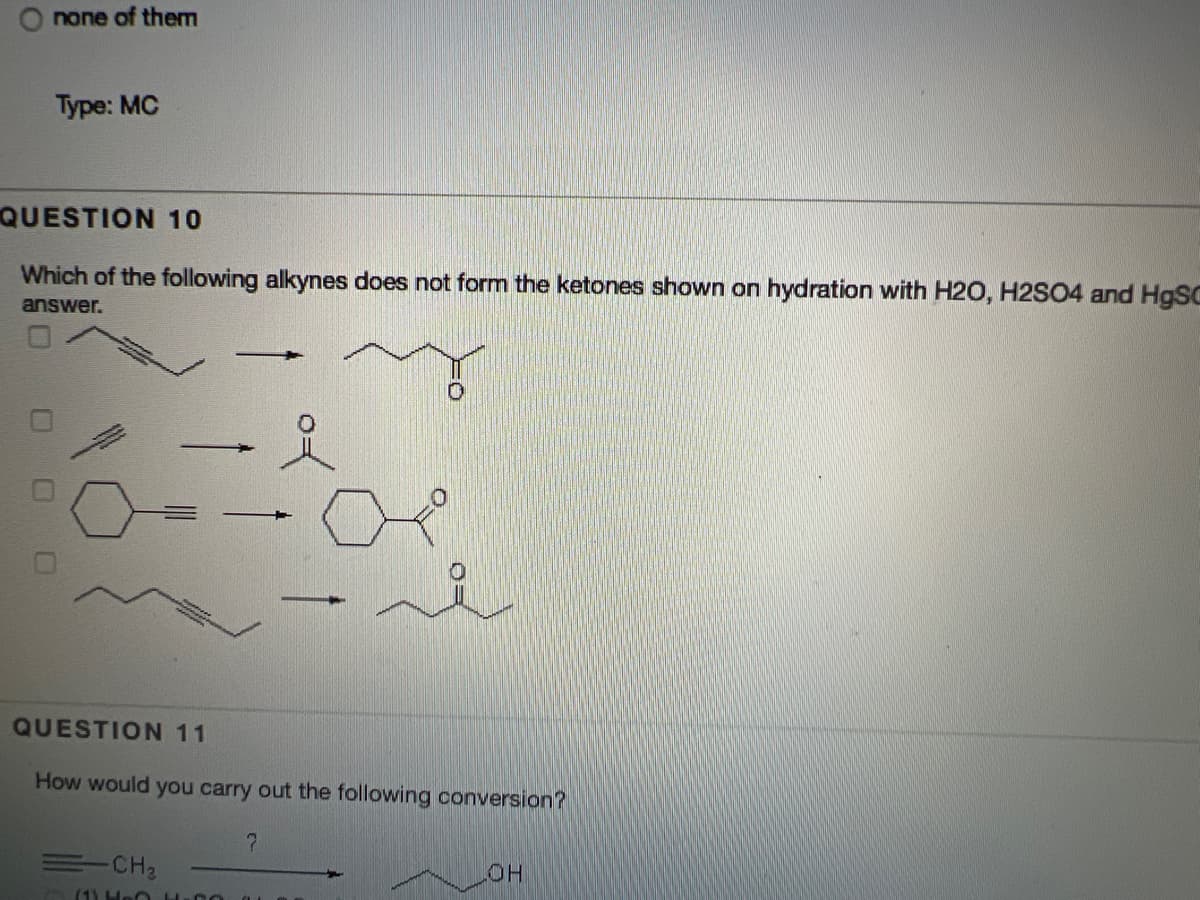
Transcribed Image Text:O none of them
Type: MC
QUESTION 10
Which of the following alkynes does not form the ketones shown on hydration with H2O, H2SO4 and H9SC
answer.
QUESTION 11
How would you carry out the following conversion?
=CH2
HOT
(1) Hen 00
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

