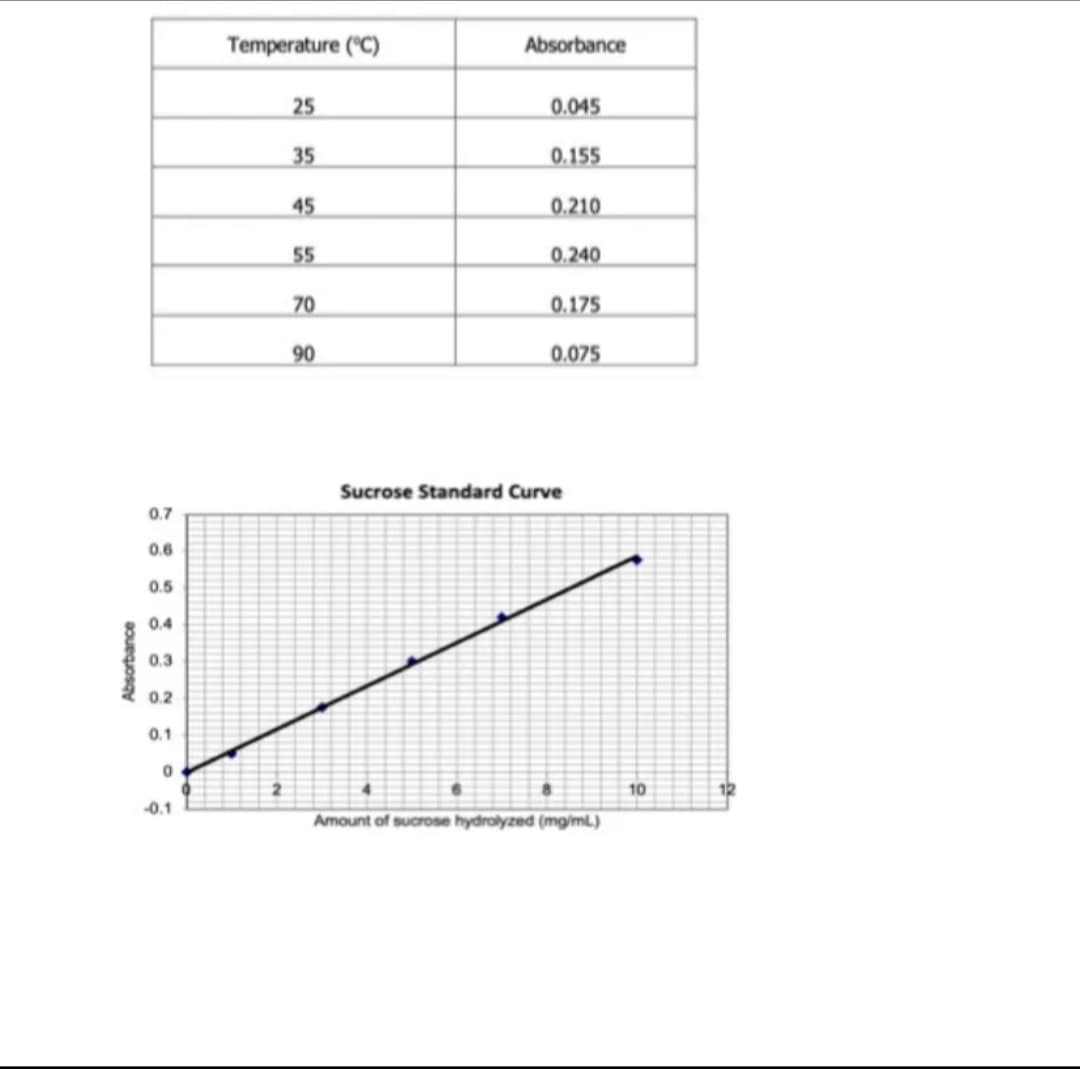
A sample of yeast extract has been analyzed of its invertase activity. The effect of temperature on the invertase activity was monitored using the same dinitrosalicylic acid assay used in your experiment in the laboratory. Refer to the data below. What is the optimum temperature of invertase in deg C?
Q: There are eight chemical reactions that occur in the citric acid cycle process. The reactions of the…
A: Cellular respiration is the process how biochemical energy is generated from food. It involves the…
Q: *Is B-D-glucosamine a reducing sugar? HO CH₂OH OH О ОН NH₂ B-D-glucosamine
A: Sugars are of two types-reducing and non-reducing. Reducing sugars have an anomeric carbon has an…
Q: 1.1 Explain in detail, the diagrams in terms of the principle of bioenergetics. WAK products А…
A: Bioenergetics is the field in biochemistry that concerns how cells transform energy and hence…
Q: During ribosomal protein synthesis, the growing peptide chain OA) Is synthesized C-terminal to…
A: Proteins are synthesized by the ribosomes in the cytoplasm. The ribosomal machinery synthesizes the…
Q: 3. 1,3-bisphosphglycerate has a high energy bond that can be used to phosphorylate ADP to make ATP…
A: Delta G or the standard free energy of a reaction indicates the tendency of a reaction to move…
Q: 2. The Km of a particular enzyme is 2.0 x 105 M. If the initial velocity (Vo) is 0.16 mmol/min for…
A: For a one-substrate enzyme-catalyzed reaction, the Michaelis-Menton equation shows the quantitative…
Q: The textbook author admits the number of protons pumped from the matrix to inter-membrane space by…
A: Oxidative Phosphorylation: In a sequence of redox reactions, electrons are transferred from one…
Q: Inhibiting the reverse transcriptase of HIV is a common method for treating HIV infections. a) The…
A: HIV is an RNA virus which has reverse transcriptase enzyme that can convert the RNA to DNA.
Q: Describe the changes that occur in each step of the mechanism.
A: Enzymes are high molecular weight proteins that catalyse biochemical reactions. Proteases are…
Q: Transfer to PRPP Please show mechanism steps and arrow pushing.
A: Nucleophiles are electron donating groups . They participate in biochemical reactions by donating a…
Q: In the tertiary structure of a protein, glutamine can interact with O valine; hydrophobic…
A: Proteins are bio molecules with vast diversity in their structure. They exhibit four levels of…
Q: Plant vacuoles must also maintain an optimum pH for its acid hydrolases. True False
A: Enzymes are high molecular weight proteins that catalyse biochemical reactions. Their catalytic…
Q: For class I MHC, which of the following statements are correct? P. They are expressed on all…
A: MHC are proteins encoded by a huge complex of genes called the major histocompatibility complex…
Q: Explain IN DETAIL the reactions and processes of alcoholic and lactic acid fermentation. Include…
A: The process of cellular respiration leads to catabolism of pyruvate after the glycolytic pathway.The…
Q: The amount of ATP that glycerol can produce is O less than 50% one glucase unit O 50% of one glucase…
A: Simple fat or triglycerides are fatty acid esters of glycerol. Glycerol is naturally occurring…
Q: The structure shown is an example of a type of macromolecule (carbohydrate, lipid, protein, or…
A: The biological macromolecules that constitute a cell are proteins, nucleic acids, lipids and…
Q: Can you write possible mineralization and biotransformation reactions for glucose (C6H12O6) ?
A: Biotransformation is the biochemical process (metabolism) taking place in some life form that alters…
Q: At pH 5, what is the net charge of the pentapeptide Val-Glu-Cys-Ser-Lys? O-1 O 0 O +1 O +2
A: The amino acids contain ionizable groups. The ionic form of theses ionizable groups depends on the…
Q: Starch is an example of a O none of the other answers are correct O fat O carbohydrate O protein…
A: The four classes of biological macromolecules are proteins, nucleic acids, lipids and carbohydrates.…
Q: Based on the amino acids present in the segments between the breakers, what do you predict the…
A: A protein's function depends on its structure. There are four levels of protein structure: primary,…
Q: Although insulin initially acts through a tyrosine kinase receptor it also subsequently results in…
A: Receptor tyrosine kinase (RTKs) :- 2nd major type of cell surface receptors. Ligand may be:-…
Q: 2. Mechanisms of absorption of monosaccharides from the intestinal lumen into the enterocyte and…
A: Monosaccharides are smallest or basic subunit of carbohydrates by which disaccharides,…
Q: Explain from a chemical stand point why ATP has a high phosphoryl-transfer potential. Make sure to…
A: ATP or Adenine Triphosphate is nucleotide made up of ribose sugar attached to Adenine and phosphate.…
Q: When the movement of lipids in biological membranes was measured, it was observed that: A) Saturated…
A: The cell membrane is a phospholipid bilayer. According to the Fluid Mosaic Model, the cell membrane…
Q: Which of the following statement is FALSE about enzymes? Enzymes are unconsumed during a biochemical…
A: Enzymes are biological catalysts that are utilized by metabolic reactions occurring in the living…
Q: I. Essay: Explain the following question with the use of illustrations. Briefly describe them with…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three sub-parts…
Q: Based on its R-group, how would the amino acid arginine be classified? O hydrophobic O neutral polar…
A: There are 22 different standard amino acids present. The amino acids are classified in different…
Q: Which of the following statements concerning complete protein hydrolysis is incorrect? Primary…
A: Protein hydrolysis is process of breaking down proteins into its components. Hydrolysis reactions…
Q: What is meant by intrinsic GTPase activity? Exchange of GDP for GTP on the a-subunit of the G…
A: GPCRs interact with G proteins in the plasma membrane. G proteins are specialized proteins that can…
Q: Please help me with finding the hypotheses that are being testing in each of the three enzyme…
A: Starch is a storage-type homopolysaccharide that contains two types of glucose polymers: amylose:…
Q: If there are still contaminating proteins with your LDH fraction after going through 3 different…
A: Proteins are high molecular weight biomolecules made up of amino acid residues linked via peptide…
Q: Use the relationships revealed by a Lineweaver-Burk plot and the table of enzyme performance to…
A: Parameters such as Km and Vmax are used for comparing enzyme activities. If we know the initial rate…
Q: If the concentration of solutes in the fluid surrounding the cell is less than the cell's cytoplasm…
A: Osmosis is the movement of water across the semipermeable membrane driven by the difference in…
Q: 1. What is the name of the nucleobase in the nucleotide structure? a. guanine b. adenine c.…
A: Nucleic acids are composed of nucleotides (sugar, nitrogenous base & phosphate group). Nucleic…
Q: A molecule that is required by enzymes in order for the enzyme to be active is called a OA)…
A: Enzymes are high molecular-weight protein molecules that catalyse biochemical reactions. The…
Q: When performing cloning experiments, it is not always necessary to treat sources of DNA with the…
A: Isoschizomers are enzymes that can share the same recognition site in restriction digestions.
Q: HO -NH₂ NH NH HN S-CH3 ΝΗ O The peptide chain has OH residues. The name of the peptide using single…
A: Peptides are small sequences of amino acids that are joined together through peptide bonds. Peptides…
Q: In an experiment to determine the degree of saturation in 0.43 g of an unknown lipid, the following…
A: The fatty acids in triglycerides can have one or more double bonds in the hydrocarbon chain. Fatty…
Q: 1. Enzymes A) tilt the reaction equilibrium in favor of product formation B) tilt the reaction…
A: The enzymes are biological catalysts that increases the rate of biochemical reactions. Most enzymes…
Q: A. Draw the chemical repeat unit of corn starch. B. What type of material is the corn starch/water…
A: Cornstarch consists of amylose and amylopectin. Both are polymers of glucose, but have different…
Q: 4. What is the sum of all the hydrogens at the highlighted carbon atoms in the given structure? A. 7…
A: The given structure is a hydrocarbon. By balancing the valency of carbon, we can calculate the…
Q: You are a research scientist studying miRNA processing. You currently know everything about the…
A: Dicer is also known as helicase with RNase motif. It belongs to RNAse III family. Dicer cleaves…
Q: Draw the fractional binding curve with protein that bind to a molecule of ligand L.
A: A ligand is a molecule that binds to a receptor. The specificity of the ligand varies. One ligand…
Q: 1. I Draw the amino acids Asn and His and show the most prevalent interaction that would occur…
A: Asparagine is a polar uncharged amino acid, while histidine is a polar charged amino acid.…
Q: 5. Which of the following statements is/are correct regarding allosteric regulation? a) Allosteric…
A: Allosteric enzymes increase or decrease the reaction based on response to the regulatory molecule…
Q: Identify the biochemical role that each plays within a biochemical transformation:…
A: Coenzymes and cofactors are non protein components that are essential for a protein function. Most…
Q: it is widely accepted that proteins can organize themselves according to a range of stable…
A: Introduction Proteins are the most abundant macromolecules in our body. proteins are made up of…
Q: In hemoglobin from patients with sickle cell anemia, the hemoglobin ß-chains have a single amino…
A: Sickle cell anemia is a genetic disease that results in the formation of the sickle shape of RBCs.…
Q: What will be the result for each qualitative test if the intact casein contains the following…
A: Proteins are folded peptides. Peptides are made up of amino acid residues linked via a peptide bond.…
Q: How many reduced molecules (NADH, FADH2, NADPH) will be generated by converting lineoleic acid…
A: The fats are stored in eukaryotes as triglycerides that are broken down using the enzyme Lipase to…

Step by step
Solved in 2 steps

- You are studying a biochemical pathway and isolate Neurospora mutants I, II, and III.Mutant I can grow if you supplement the medium with Z.Mutant II can grow if you supplement the medium with X, Y, or Z.Mutant III can grow if you supplement the medium with X and Z, but not with Y. Draw a biochemical pathway that shows the correct order for compounds X, Y, and Z and for the enzymes that each mutant is defective for.Sydney Brenner isolated Salmonella typhimurium mutants that were implicated in the biosynthesis of tryptophan and would not grow on minimal medium. When these bacterial mutants were tested on minimal medium to which one of four compounds (indole glycerol phosphate, indole, anthranilic acid, and tryptophan) had been added, the growth responses shown in the following table were obtained. Mutant Minimal medium Anthranilic acid Indole glycerol phosphate Indole Tryptophan trp-1 − − − − + trp-2 − − + + + trp-3 − − − + + trp-4 − − + + + trp-6 − − − − + trp-7 − − − − + trp-8 − + − − + trp-9 − − − − + trp-10 − − − − + trp-11 − − − − + Give the order of indole glycerol phosphate, indole, anthranilic acid, and tryptophan in a biochemical pathway leading to the synthesis of tryptophan. Indicate which step in the pathway is affected by each of the mutations.Is the following statement correct or incorrect? It is only possible to employ enzymes that are present naturally in yeast or bacteria for biocatalysis.
- The substrate which was used to investigate the enzymatic activity of wheat bran phosphatase was p-nitrophenyl phosphate. It is converted to p-nitrophenol and inorganic phosphate by this enzyme. P-nitrophenol has a pKa of 7.1 and is colorless, while its conjugate base, p-nitrophenolate has a yellow color. The enzyme assay was performed at pH 5.1 after the 0.5 N KOH is added at the end of each enzyme assay, the pH of the assay mixture is raised to 9.1. Why was the base added?A researcher is interested to test the antibacterial activity of a Philippine Plant crude extracts against Beta-lactamase producing Klebsiella pneumonia isolated in a hospital setting. He will use the following concentrations of crude extract, 500mg/ml, 250mg/ml and 100mg/ml. Positive control: Tetracycline 10mg/ml and DMSO as the negative control. Given this research scenario kindly construct a research framework.What reaction would you expect when performing a positive control in the oxidase assay? What would it mean if a known oxidase-positive bacterium did not cause the expected reaction?
- After testing the potency of different β-lactamase inhibitors against purified OXA-M290 enzyme, the antimicrobial activity of combinations of β-lactam antibiotics and β-lactamase inhibitors is tested against the E. coli KGH1 strain that produces OXA-M290. The results of these studies can be used to identify the best antibiotic to treat an infection caused by this E. coli strain. Broth microdilution tests are carried out with a panel of β-lactam antibiotics and β-lactamase inhibitors to determine their MICs against E. coli KGH1. These tests are carried out in a 96-well microplate. Three β-lactam antibiotics (amoxicillin, ceftazidime, and ertapenem) and three β-lactamase inhibitors (sulbactam, tazobactam, and vaborbactam) are tested. The rows in the microplate correspond to the following antibiotics and inhibitors: Row A: amoxicillinRow B: amoxicillin + sulbactamRow C: ceftazidimeRow D: ceftazidime + sulbactamRow E: ertapenemRow F: ertapenem + sulbactamRow G: ertapenem + tazobactamRow H:…For the production of a secondary metabolic by Streptomyces coelicolor, a fed batch was performed. At the end of the single batch phase, the following conditions were reached in the reactor: V=10000L, cell concentration X=10g/L and product concentration P=0.1g/L. The feeding was then started with constant flow F= 200L/h, for 100h. Knowing that the substrate concentration in the feeding medium was SF= 80g/L and in the fermentation medium it was practically null, calculate: a) The final concentration of cells and productb) If the reactor were fed with a constant dilution rate (D), what should be the value of D used to reach the same cell concentration obtained in the situation with constant flow?μp= 0.01 g of product/ (g of cells.h)Y x/s = 0.15g cells/g of substrateGiven this research scenarios kindly construct a research framework. A researcher is interested to test the antibacterial activity of a Philippine Plant crude extracts against Beta-lactamase producing Klebsiella pneumonia isolated in a hospital setting. He will use the following concentrations of crude extract, 500mg/ml, 250mg/ml and 100mg/ml. Positive control: Tetracycline 10mg/ml and DMSO as the negative control.
- For an experiment where you will using different concentrations of avocado catalase to determine how that affects the rate of the reaction with its substrate, hydrogen peroxide, what would be the general protocol? What is the control in the experiment?Pyridoxal phosphate (PLP) is a coenzyme for the enzyme ornithine aminotransferase. The enzyme was purified from cells grow in PLP = deficient media as well as from cells grown in media that contained pyridoxal phosphate. The stability of the two different enzyme preparations was then measured by incubating the enzyme at 37°C for different lengths of time and then assaying for the amount of enzyme activity remaining. The following results were obtained. (a) Why does the amount of active enzyme decrease with the time of incubation? (b) Why does the amount of enzyme from the PLP deficient cells decline more rapidly?200 ml of a 2% protein solution containing an enzyme that you want to purify. Half of the sample is subjected to method A, consisting of fractionated precipitations and 5 ml of final solution are obtained, with a concentration protein equal to 5 mg / ml and enzymatic activity equal to 2000 U / ml. The other half is subjected to method B, consisting of ion exchange chromatography, and a final solution of 10 ml, with protein richness equal to 10 mg / ml and with an activity enzymatic also equal to 2000 U / ml. You want to know: a) Which of the two methods has provided the purest enzyme. b) By which of the methods the greatest amount of enzyme has been obtained.

