A scientist needed to determine the concentration of a potassium hydroxide solution. To do this, they titrated 10.0 mL of the base against 0.150 M sulfuric acid. The titration was repeated multiple times to improve the accuracy. Determine the concentration of the potassium hydroxide solution. a Choose the correct titration runs, and calculate the average titer of acid. (Enter your answer to four significant figures.) Average titer of acid = mL Show/Hide Help Trial Run Titer Run 1 Titer Run 2 Titer Run 3 Titer Run 4 Titer 23.70 mL 22.65 mL 22.75 mL 22.70 mL 23.75 mL
A scientist needed to determine the concentration of a potassium hydroxide solution. To do this, they titrated 10.0 mL of the base against 0.150 M sulfuric acid. The titration was repeated multiple times to improve the accuracy. Determine the concentration of the potassium hydroxide solution. a Choose the correct titration runs, and calculate the average titer of acid. (Enter your answer to four significant figures.) Average titer of acid = mL Show/Hide Help Trial Run Titer Run 1 Titer Run 2 Titer Run 3 Titer Run 4 Titer 23.70 mL 22.65 mL 22.75 mL 22.70 mL 23.75 mL
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter4: Stoichiometry: Quantitative Information About Chemical Reactions
Section: Chapter Questions
Problem 139SCQ: Two students titrate different samples of the same solution of HCl using 0.100 M NaOH solution and...
Related questions
Question
answer and explain step by step
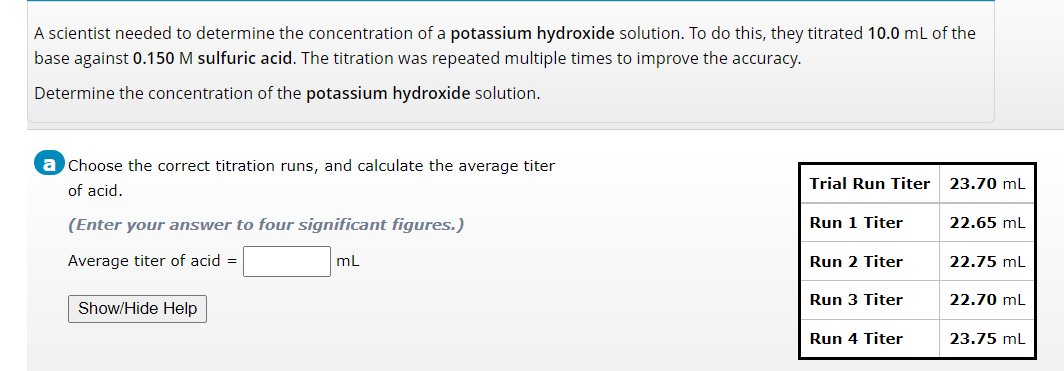
Transcribed Image Text:A scientist needed to determine the concentration of a potassium hydroxide solution. To do this, they titrated 10.0 mL of the
base against 0.150 M sulfuric acid. The titration was repeated multiple times to improve the accuracy.
Determine the concentration of the potassium hydroxide solution.
a Choose the correct titration runs, and calculate the average titer
of acid.
(Enter your answer to four significant figures.)
Average titer of acid =
mL
Show/Hide Help
Trial Run Titer
Run 1 Titer
Run 2 Titer
Run 3 Titer
Run 4 Titer
23.70 mL
22.65 mL
22.75 mL
22.70 mL
23.75 mL
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning