Q: How many moles of sodium hydroxide are there in 1.00 kg of sodium hydroxide (Given Na=22.99 g/mol; O...
A: Given mass of sodium hydroxide = 1kg = 1000g
Q: At a given temperature, 4.595 atm of F2 and 5.404 atm of Br2 are mixed and allowed to come to equili...
A: Given : We have to calculate Kp for the reaction.
Q: Rationalize the following observations. (1) From Fe(CO)s to Fe(CO):(PPhs)2, absorptions in the IR sp...
A: To know the answer of these questions, we have to know bonding pattern and structure of the molecule...
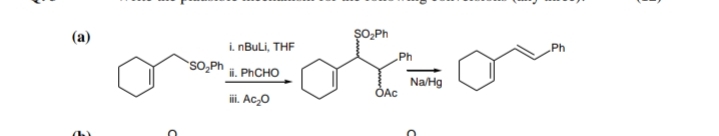
Q: Propose a mechanism for the reaction shown below. ÇI OH ONH2 + CI Нeat NO2 NO2
A: NOTE: The product given is incorrect and the correct product will have NH2 group in the place of OH....
Q: 1. a. What is the pH of a solution prepared by adding 25.0 mL of 0.10 M acetic acid (Ka = 1.8 x 10-5...
A:
Q: Rewrite this measurement with a simpler unit, if possible. kg-m 4.3 3. m
A: Given measurement is : Rewrite this measurement with a simpler unit, if possible = ?
Q: Consider the reaction IO−4(aq)+2H2O(l)⇌H4IO−6(aq);Kc=3.5×10−2IO4−(aq)+2H2O(l)⇌H4IO6−(aq);Kc=3.5×10−2...
A:
Q: The gas-phase reaction X(g) +Y(g) -> XY(g) has an overall energy change of +45 kJ. The activation en...
A:
Q: Balance the following Equation : Fe3O4 (s) + CO (g) = Fe (s) + CO2 (g)
A:
Q: C3H8 + 5 O2 → 3 CO2 + 4 H2O a) If I start with 5 grams of C3H8, what is my theoretical yi...
A:
Q: The carbocation electrophile in a Friedel-Crafts reaction can be generated in ways other than by rea...
A:
Q: Be + 2 HCl --> BeCl2 + H2 My theoretical yield of beryllium chloride was 10.7 grams. I...
A: Since you have posted multiple questions as per guidelines we can answer only one per session . If y...
Q: (d) Explain the observed stereo-selectivity in the formation of product using Chelation model. OMe Z...
A: The mechanism of this reaction is done in the below.
Q: Suggest the following transformation: i. Synthesis of 1,2-diphenylethanol using only benzene as the ...
A: For converting benzene to 1,2-diphenylethanol the reaction is as follows
Q: 5. Among many of the drugs studied for the treatment of cancer is the carboxamide derivative Poly (A...
A: Major product.
Q: Compare your experimental observation on the disruption of phycocyanin folding in the presence of ac...
A:
Q: A piece of aluminum with a mass of 30 grams was heated in a steam jacket to 98oC and then plunged in...
A:
Q: What does a theoretical yield of 1.38g mean ?
A: Given, Theoretical yield = 1.38 g Required, What is the meaning of theoret...
Q: Complete combustion of a mole of natural gas produces a hotter flame than a mole of LPG. Select the ...
A: Natural gas is composed of methane and LPG is composed of propane. Both gas burn with blue flame. He...
Q: HN. NBS, ROOR, hv Br AICI,
A: Given : We have to check whether the given conversion is right or wrong.
Q: A mixture was found to contain 1.50g NH4CI, 0.80 g NaCI, and 1.20 g SiO2. What is the percentage of ...
A: Given: Mass of NH4Cl = 1.50 g Mass of NaCl = 0.80 g Mass of SiO2 = 1.20 g
Q: 1H NMR 2(CHa) CH2 PPM
A: Spectral Analysis - We observe two peaks in the above 1 H NMR spectrum 1) one due to the CH2 hydroge...
Q: Question 20 he effective cation exchange capacity is the total capacity of a soil to hold exchangeab...
A: True/False
Q: A solution is made by mixing 13,0 g of solid barium hydroxide and 50.0 mL of 100 M nitric acid. Writ...
A: Neutralization reaction is reaction where an acid and base react to form water and salt Here we are...
Q: Indicate the orbital where each lone pair is found. (Encircle lone pair and label) Answer Letter (a...
A: We have to predict the type of orbitals each lone pair present in.
Q: For the following equilibrium, K= 6.2 x 104: Pb2+ + 2CI PbCl2 Which of the following is true?
A:
Q: Carbon NMR Homework 200 180 160 140 120 100 80 60 40 20 ppm
A: The given compound is C6H4Br2.
Q: how how the forward and reverse reaction rates respond to having some HOCl suddenly removed from the...
A: The reaction rate depends on the concentration of the reactant but the reaction rate does not depend...
Q: 5. Propose the structure of a compound that exhibits the following 'H NMR data: (c) C4H100 0.90 8 (6...
A: The given compounds mainly consists of C, H, and O. As heteroatoms are present in these compounds, ...
Q: 2 NaCl + CaO → CaCl2 + Na2O What is my theoretical yield of sodium oxide if I start with 20 gr...
A: The balanced reaction given is, => 2 NaCl + CaO → CaCl2 + Na2O Given: Mass of CaO reacting = 20 g...
Q: Find the pH of 0.99 M solution of LIOH.
A: pH is power of hydrogen or potential of hydrogen. pH scale is used to determine the nature of a solu...
Q: 1. Explain why you only see four absorptions. Include why you only see 2 absorptions in the C-O regi...
A: We use 13C NMR spectroscopy to find the structure of the compound using 13C carbon which is NMR acti...
Q: Carbon-hydrogen bonds exhibit a range of different chemical reactivity that depends on molecular str...
A: Primary carbon: A primary carbon is a carbon atom bound to 1 other carbon atoms. Secondary carbon: A...
Q: 2. Draw out the forward synthesis (fill in the boxes) only for the following transformation (it'll b...
A: Here we have to make a desired forward synthesis for below conversion .
Q: what was johns dalton initial research focus was and what he eventually contributed to early atomic ...
A: Johns dalton first introduced atomic theory in chemistry.He gave three laws.These are i-Law of Conse...
Q: The initiation step of radical halogenation of alkanes involves homolytic bond formation. Select the...
A: Radical halogenation is the substitution of hydrogen atom of alkane by a halogen atom.
Q: Sloichiometry: Mole to Mass Problems Mg +2HCI → MgClz +2 H2 1. a. How many grams of HCl are consumed...
A: Since you have asked multiple question, we will solve the first question for you.If you want any spe...
Q: CH3 CH3-CH2-C-CH2-CH3 HNO3 KMNO4 A B H,SO4 H2SO4/Heat CH2-CH3 HNO3 H2/Pt or C FeCl3 Cl2 A (Para Prod...
A: Given reactions are : Predict the major products of the reactions = ?
Q: (a) Given that the solubility product of rhodochrosite, MNCO3, is 10-93, calculate the equilibrium c...
A: In a solution that contains a solute having low solubility, the ions tend to form precipitate. An eq...
Q: HO PPM
A: The given NMR spectra correspond to the alcohols and iodides with variable number of carbon atoms.
Q: 7. In each set of structures, arrange_the labeled bond s in the order indicaleed Set 1 a Increasing ...
A: We have to tell about the bond energy. We know that if a bond having greater s-character, then the b...
Q: Why is it importance to understand the difference of an acid and base solution? What benefits does i...
A: Given : We have to tell why there is need to understand the difference between acid and base.
Q: A stainless steel alloy is to be analyzed for its chromium content. A 3.30 g sample of the steel is ...
A: Given Mass of sample steel = 3.30 g volume of solution = 250 ml Mass of BaCrO4 = 0.145 g M.m (molar ...
Q: Identify each of the following groups as an activator or deactivator and as an o,p-director or m-dir...
A: Given : We have to explain whether the gives groups are activator or deactivator.
Q: Some N2 gas is mixed with some O2 gas, and The picture shows a representative sample of the mixture....
A:
Q: The structure given is Escitalopram, an active enantiomer of citalopram and works as an anti-depress...
A:
Q: b) You wanted to produce 2-nitrobenzene-1,4-dicarboxylic acid. For that purpose, you started the rea...
A: We can solve this question by considering the directing nature of the substituents it means that in ...
Q: A container is filled with 5g of gas at STP. It is heated to 35°C with pressure being kept constant....
A: Given: Mass of gas = 5 g. Initial conditions are STP conditions i.e. 1 atm and 273.15 K. And final t...
Q: Is the name correct? 5-(2,2-dimethylethyl)nonane If not, give the correct name.
A:
Q: Predict the major product(s) you would obtain from sulfonation of o-dichlorobenzene. • You do not ha...
A: Aromatic electrophilic substitution reaction: Benzene and other aromatic compound gives mainly aroma...
Step by step
Solved in 3 steps with 3 images

- The attractive force between two ions in Na20, Fc is 8.64x10-9N. What is the bond length a0 (in nm) given that valence of the charged Na+ is Z1=+1, for O-, is Z2 =-2, the charge of a single electron q is 0.16x10-18 C and the proportionality constant k0=9x109 V.m/C? Note that K=k0(Z1q)(Z2q) and that 1 V.C = 1 J.What is the value for Kb Na2C6H6O6?Write a short note on the history of inoganic chemistry.

