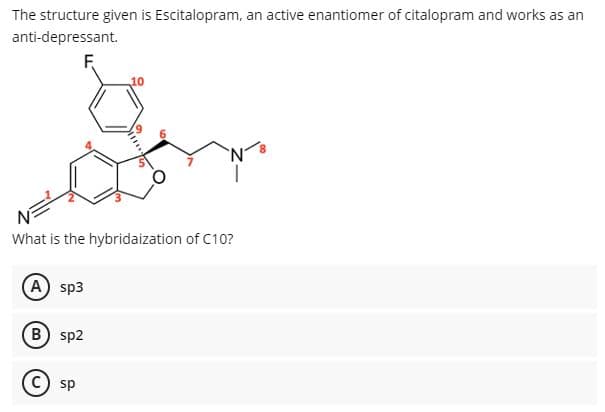
The structure given is Escitalopram, an active enantiomer of citalopram and works as an anti-depressant. What is the hybridaization of C10? A sp3 B) sp2 sp
Q: By convention, a chemical bond between two atoms with an electronegativity difference of 2.0 is whic...
A: Electronegativity is defined as tendency of atom to attract shared pair of electrons.
Q: The oxidation of Fe 2+ to Fe 3+ can be induced by hydrogen peroxide in an acid medium. Write the hal...
A: Given that, a balanced redox reaction is 2Fe2+ + H2O2 + 2H+ → 2Fe3+ + 2H2O. We have to write down t...
Q: topic: matter do you think that each sample occupies space? write the reason(s) for each sample wa...
A: Yes, it does because whatever we see when you open ur eyes is matter and what you feel when you clo...
Q: A nitrogen sample occupies 246 mL at STP. If the same sample were collected over water at 25°C and 6...
A: For the phase change of the ideal gas the temperature, pressure, and volume of the initial state and...
Q: when you pour one cup of tap water into a transparent bottle, and add one small drop of food colorin...
A: Given: You pour one cup of tap water into a transparent bottle and add one small drop of food colour...
Q: In a calorimetry experiment similar to the one performed today, a student used 50.0 mL of 1.0 M NaOH...
A: Since you have posted multiple questions, we are entitled to answer the first only. 5) Given: Concen...
Q: Reactants Net Ionic Equation: 3a CoCl(aq) + KOH (6M) -> Co lag) + 2 0H (aq)> Co(04),(6) Co(OH)26) +H...
A: Here we have to find the incorrect net ionic equations.
Q: All of the following are added to five different containers of water, each with the same mass of wat...
A: Which resulting solution would yield the lowest freezing point?
Q: Write ways of synthesizing the following compounds using acetyl ion ions. If the synthesis of any gi...
A:
Q: Predict the major product(s) you would obtain from sulfonation of 4-bromobenzoic acid.
A:
Q: Ph Ph но Ph Ph Ph Ph 1.0 eq TIOH, DCE -Ph A Ph Ph Но Propose a reasonable mechanism using Baldwin Ru...
A: 5 - exo- tet is more favorable over 6 endo trig . This is well known as Baldwin's rule.
Q: hat is the rate of formation of oxygen if the rate of decomposition of dinitrogen pentoxide is 2.748...
A: Given : 2 N 2O 5( sol'n) → 4NO 2( sol'n) + O 2( g)
Q: In an experiment to study the formation of HI(g), H2(g) + I2(g) ⇌ 2 HI(g) At equilibrium, [H2]...
A:
Q: d) increasing the pressure in the reaction vessel e) decreasing the temperature of the system
A: For the preparation of Ammonia the haber process is scientifically used and is given by when one of ...
Q: 8) Identify the nucleophile and electrophile in each reaction. А. H. O.. CI CI B.
A: Electrophile : As the name specify electro means electron and phile means loving, it means the speci...
Q: Draw the major product(s) of nitration of benzoic acid.
A:
Q: Which of the following is less soluble in water, ethanol (C2H5OH) or BaSO4.
A:
Q: In a calorimeter experiment, NaOH and HNO3 underwent a neutralization reaction and produced 0.075mol...
A: Neutralization reactions are generally exothermic and thus ΔH is negative.
Q: At a given temperature, 4.595 atm of F2 and 5.404 atm of Br2 are mixed and allowed to come to equili...
A: Given : We have to calculate Kp for the reaction.
Q: The following set of data was obtained by the method of initial rates for the reaction: S2O82-(aq)...
A:
Q: Carboxylic acids are known to exhibit a higher C=O stretch frequency in the gas phase than in the co...
A:
Q: Give the IUPAC name of the compound below.
A:
Q: Consider the reaction, CH3CI(g) + 3CI2(g) → CCI4(g) + 3HCI(g) (a) Express the rate of reaction with ...
A: We are to write the rate of reaction for given reaction CH3Cl + 3Cl2 → CCl4 + 3HCl and, [HCl] = 0.02...
Q: During heating, some solution of NaCl splattered out of the evaporating dish. How would this affect ...
A: Given: During heating, some solution of NaCl splattered out of the evaporating dish.
Q: The alloy brass contains lead as an impurity in the form of lead sulphate (PbSO,). By dissolving bra...
A: Since you have asked multiple questions, we will answer only first question for you. In order to get...
Q: A mixture of gaseous CO and H2, called synthesis gas, is used commercially to prepare methanol (CH3O...
A:
Q: Write the letter of the pair of compounds that illustrates the given isomerism. CH3 H,C CH CH H2N H3...
A: Functional isomerism=> Compounds have same same molecular formula but different functional group....
Q: decrease the freezing poi taloe 17. A solution of glucose in water is prepared so that the concentra...
A: Answer -
Q: A sulfide iron was formed by combining 2.233 g of iron (Fe) with 1.926 g of sulfur (S). What is the ...
A:
Q: A(g) + 12 B(g) = 2 C(g) Determine the expression for the rate of the reaction with respect to each ...
A:
Q: What are the distinguishing features of gas and vapor poisoning compared with other types of poisoni...
A: A question based on catalysis that is to be accomplished.
Q: Calculate the [H+ ] in solution of the following pH: a. 3.00 b. 10.53.
A: Given : We have to calculate [H+ ] for the following
Q: Predict the major product(s) you would obtain from sulfonation of 1-bromo-2-methylbenzene. • You do ...
A: Given that : We have to predict the major product(s) obtained from the sulfonation of 1-bromo-2-meth...
Q: What is the final temperature (in degrees Celsius) when 1940 J of energy are put into a 250 millilit...
A: We have to predict the final temperature.
Q: Complete the Kw expression for the autoionization of water. Kw 1.00 x 10-14 =
A: Please find your solution below : Autoionisation of water is a reaction in which water forms hydroni...
Q: 3. An empty 1.0 was filled with 5.0 moles of oxygen and 4.0 moles of nitrogen. If the amount of NO2 ...
A:
Q: For the following chair conformations of 1,2-dibromocyclohexane, determine the orientations of the t...
A:
Q: Ethane
A: We have to propose a synthesis from ethane to butan-2-one. The required synthesis is shown below
Q: H3C CH3 H3C OCH3 CH3
A: This is a synthesis type transformation reaction , passing through several steps . Necessary reagen...
Q: Rationalize the following observations. (1) From Fe(CO)s to Fe(CO):(PPhs)2, absorptions in the IR sp...
A: To know the answer of these questions, we have to know bonding pattern and structure of the molecule...
Q: 0.550-gram sample of a nondissociating enzyme was dissolved in water at 25°C to yield 50.0 mL of sol...
A: Using osmotic pressure formula we can calculate molecular mass of enzyme. Formula is - ...
Q: 3. Oxazolidines are important synthetic precursors for the production of drugs. The reason is that t...
A: Here we are given with organozinc reagent, we have to utilize it and have to make the reaction.
Q: Considering the Ammonium ion, construct the SALCS of this cation and also the MO orbital diagram for...
A:
Q: 2The reductant in Walden reductor, is a. any first group element O b. metallic silver OC KMNO4 O d. ...
A:
Q: Ksp
A:
Q: A piece of aluminum with a mass of 30 grams was heated in a steam jacket to 98oC and then plunged in...
A:
Q: Part C A carbon skeleton for cyclohexane is shown below. Where on that skeleton would the substituen...
A:
Q: Consider the gas-phase reaction of nitric oxide with hydrogen at 1280°C 2NO(g) + 2H2(g)→N2(g) + 2H2O...
A: The gas phase reaction of nitric acid and hydrogen at 1280 °C is given as 2 NO (g) + 2 H2 (g) → N2 (...
Q: when you mix 20ml sugar and 50ml water in a 250ml beaker, what is the volume of the sugar and water ...
A:
Q: Discuss the theory behind potentiometric pH determinations. Then compare the advantages and disadvan...
A: Potentiometric pH meter is used to measure the difference in electric potential between the pH elect...
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Tamiflu(shown below) is an antiviral medication used to treat influenza. How many asymmetric centers are present in the this structure?Thapsigargin is a natural product with promising anticancer properties Question: How many sp2 hybridized C's are present?Cymbalta (shown below) is an antidepressant. How many asymmetric centers are present in this structure?
- Propose structures for molecules that meet the following descriptions:a. Contains two sp2-hybridized carbons and two sp3-hybridized carbons.b. Contains only four carbons, all of which are sp2-hybridized.c. Contains two sp-hybridized carbons and two sp2-hybridized carbons.A) For a molecule with three chiral carbons the possible number of stereoisomers is: 4, 8, 6 or 2 B)Which of the following has a S configuration: (Image)Draw three-dimensional representations of the following compounds. Which have asymmetric carbon atoms? Which have no asymmetric carbons but are chiral anyway? Use your models for parts (a) through (d) and any others that seem unclear.1,3-dichloropropadiene
- which carbon is more electronegative : a sp3d hybridized carbon b sp2 hybridized carbon c sp hybridized carbonDo the following compounds have any asymmetric centers? b. Are the compounds chiral? (Hint: Make models.) 1. CH2=C=CH2 2. CH3CH = C = CHCH3How are they identical, if in the first newman projection CH3 and CH3 are opposite from each other, but then in the second newman projection Br and CH3 are opposite from each other. Wouldnt they be diastereomers since one of the chiral centers changed?
- (a)(2R,3S)-2,3-dibromohexane, draw any planes of symmetry.Propose structures for molecules that meet the following descriptions: Contains two sp2 hybridized carbons and two sp3 hybridized carbons.Specify the hybridization (sp, sp2, sp3, sp2d, sp3d, spd2 etc.) of the circled atoms in the structures below a. Hybridization of the circled carbon is _______________; of the circled nitrogen is b.Hybridization of the circled (C=O) oxygen is _____________; of the circled (C-N-C) nitrogen is ______________; of the circled sulfur is c.Hybridization of the circled carbon on the left of C≡C is _____________;of the circled (CH2) carbon is ______________; of the circled (Si=O) silicon is______________; of the circled (C=O) oxygen is

