A student is told that a solution of 1.03 g of an unknown ionic compound dissolved in 10.0 g of water freezes at -1.64 oC. She is told to assume the solution has ideal behayior. She is also told the unknown is one of the compounds given below. Which of the ionic compounds isher unknown? The freezing point of water is 0 oC and Kf = 1.86 oC/m. Group of answer choices aluminum nitrate, Al(NO3)3 (MM = 213 g/mol) strontium perchlorate, Sr(CIO4)2 (MM = 286.6 g/mol) cesium iodide, Csl (MM = 259.8 g/mol) barium sulfate, BaSO4 (MM = 233.4 g/mol) Expert Answer O
A student is told that a solution of 1.03 g of an unknown ionic compound dissolved in 10.0 g of water freezes at -1.64 oC. She is told to assume the solution has ideal behayior. She is also told the unknown is one of the compounds given below. Which of the ionic compounds isher unknown? The freezing point of water is 0 oC and Kf = 1.86 oC/m. Group of answer choices aluminum nitrate, Al(NO3)3 (MM = 213 g/mol) strontium perchlorate, Sr(CIO4)2 (MM = 286.6 g/mol) cesium iodide, Csl (MM = 259.8 g/mol) barium sulfate, BaSO4 (MM = 233.4 g/mol) Expert Answer O
Chemistry & Chemical Reactivity
9th Edition
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter13: Solutions And Their Behavior
Section: Chapter Questions
Problem 71GQ: An aqueous solution containing 10.0 g of starch per liter has an osmotic pressure of 3.8 mm Hg at 25...
Related questions
Question
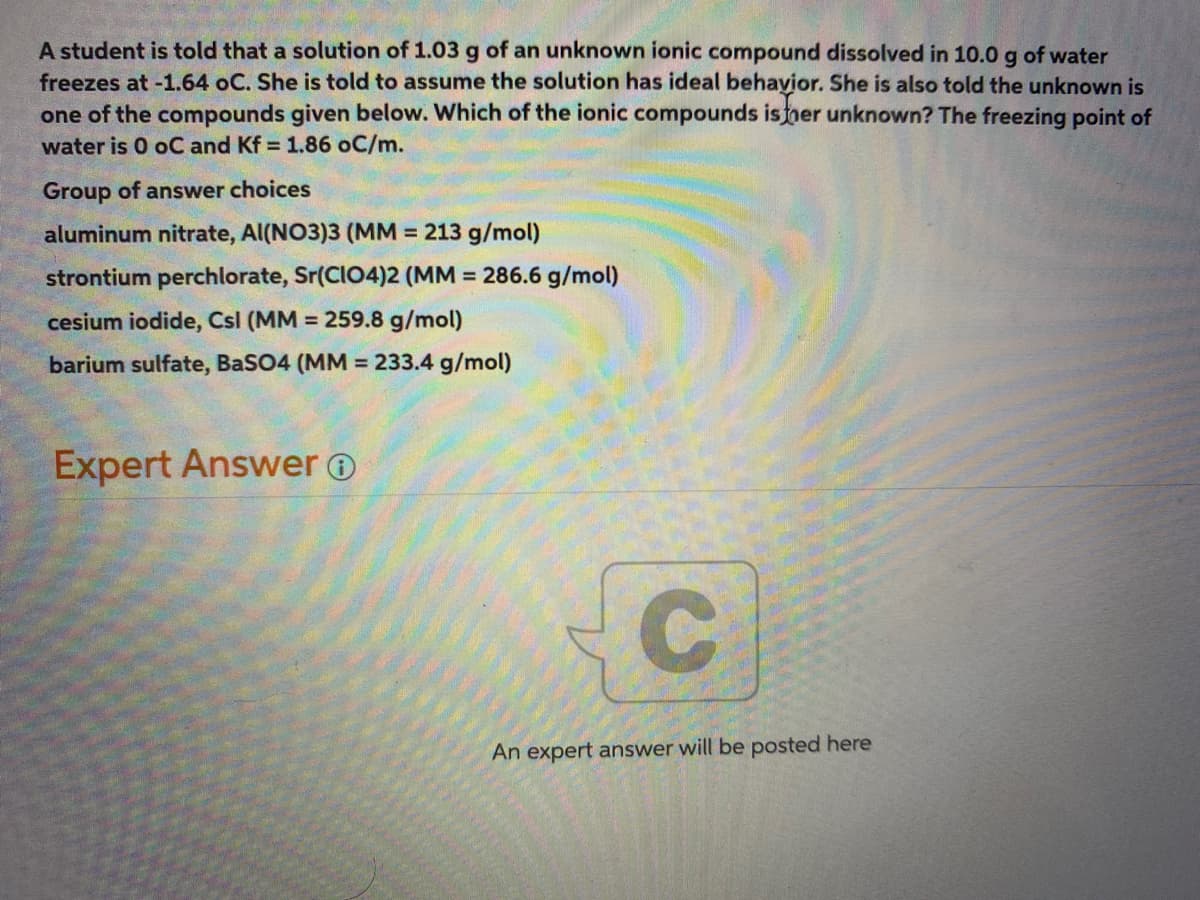
Transcribed Image Text:A student is told that a solution of 1.03 g of an unknown ionic compound dissolved in 10.0 g of water
freezes at -1.64 oC. She is told to assume the solution has ideal behayior. She is also told the unknown is
one of the compounds given below. Which of the ionic compounds isner unknown? The freezing point of
water is 0 oC and Kf = 1.86 oC/m.
Group of answer choices
aluminum nitrate, Al(NO3)3 (MM = 213 g/mol)
strontium perchlorate, Sr(CI04)2 (MM = 286.6 g/mol)
cesium iodide, Csl (MM = 259.8 g/mol)
barium sulfate, BaSO4 (MM = 233.4 g/mol)
%3D
Expert Answer o
An expert answer will be posted here
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning