A urethane is a molecule in which a carbonyl group is part of an ester and an amide (it is an amide in one direction and an ester in the other direction). Propose a mecha- nism for the reaction of an isocyanate with an alcohol to form a urethane. -N=C=0+ ELOH -NH-C-OEt Phenylisocyanate Ethanol A urethane
A urethane is a molecule in which a carbonyl group is part of an ester and an amide (it is an amide in one direction and an ester in the other direction). Propose a mecha- nism for the reaction of an isocyanate with an alcohol to form a urethane. -N=C=0+ ELOH -NH-C-OEt Phenylisocyanate Ethanol A urethane
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter22: Reactions Of Benzene And Its Derivatives
Section: Chapter Questions
Problem 22.48P: When certain aromatic compounds are treated with formaldehyde (CH2O), and HCl, the CH2Cl group is...
Related questions
Question
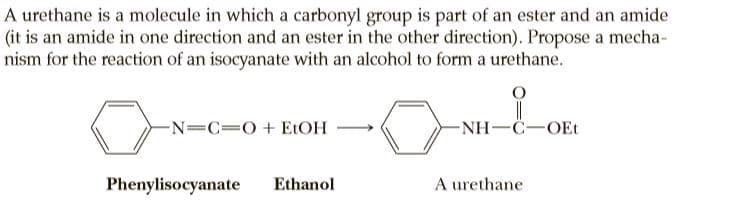
Transcribed Image Text:A urethane is a molecule in which a carbonyl group is part of an ester and an amide
(it is an amide in one direction and an ester in the other direction). Propose a mecha-
nism for the reaction of an isocyanate with an alcohol to form a urethane.
-N=C=0+ ELOH
-NH-C-OEt
Phenylisocyanate
Ethanol
A urethane
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning
