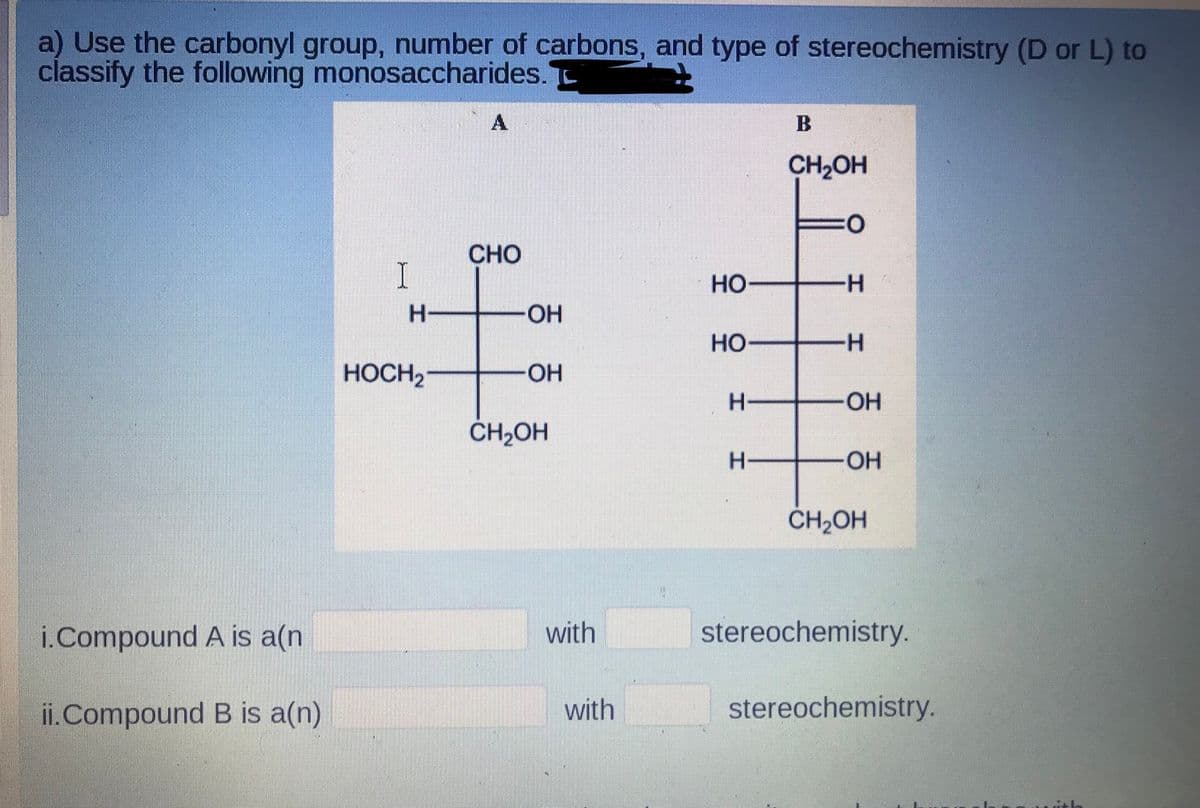
a) Use the carbonyl group, number of carbons, and type of stereochemistry (D or L) t classify the following monosaccharides. B CH2OH 0: CHO но -H- HO- Но -H- HOCH2- -HO- HO- ČH,OH ČH2OH i.Compound A is a(n with stereochemistry. ii.Compound B is a(n) with stereochemistry.
Q: The hydrolysis of a substrate, S, by an enzyme has been studied in the lab. The following initial…
A: Consider the reaction where a substrate 'S' is converted to its product 'P' by an enzyme. The rate…
Q: Aside from Giemsa, what other stains could be used in staining blood for blood parasites? What is…
A: Giemsa staining method is widely uses for staining blood for blood parasites.
Q: There are 4 classes of biochemical families (Carbohydrates, Proteins, Lipids, and Nucleic Acids).…
A: Carbohydrates are classified as monosaccharides, oligosaccharides, and polysaccharides. The…
Q: Which of the following cell types can do glycolysis, anaerobic respiration, glycogen biosynthesis,…
A: Gluconeogenesis is a process that converts non-carbohydrate substrates into glucose.
Q: Which of the following cofactors is NOT used by the pyruvate dehydrogenase complex? a. NAD+ b. FAD…
A: The pyruvate formed by the process of glycolysis is converted into acetyl-CoA. The reaction is…
Q: What does NOT describe a starch?
A: Starch is a polysaccharide that is produced by most of green plants for the purpose of storage of…
Q: To set up a standard curve you would have had to set up a dilution series. Draw up a table showing…
A: Serial dilution is the process of converting a dense solution towards a more useable concentration…
Q: Determine the p50 for variant A to the nearest 5 torr (i.e., if the p50 was 12, you would write 10).…
A: The oxygen haemoglobin dissociation curve plots the proportion of haemoglobin in its saturated form…
Q: The product formed in the first substrate-level phosphorylation reaction in glycolysis is…
A: 1. Glycolysis is a process in which one mole of glucose is partially oxidised into 2 moles of…
Q: If OAT takes ornithine and alpha-ketoglutarate as (a) substrates, draw the structures of the…
A: Aminotransferases are group of enzymes that catalyzes the transamination reaction between amino acid…
Q: 7. Which of the following is true of water in the hydration layer of proteins? a. It has a lower AS…
A: Note : Hi! Thank you for the question. We are authorized to answer one question at a time. Since you…
Q: What is the total ATP produced from complete oxidation of 10 molecules of glucose asumming that the…
A: Glycolysis is the process in which glucose is converted into pyruvate, with the production of ATP,…
Q: Briefly explain how ester hydrolysis experiments helped to provide evidence for the mechanism of…
A: Carboxylic acid and free alcohol are formed by an ester hydrolysis reaction that breaks an ester…
Q: Calculate the frictional coefficient of a molecule of DNA of 20 base pairs in water at 20C; assume…
A: DNA or deoxyribonucleic acid is a polynucleotide chain made of monomeric units of nucleic acids.…
Q: Predict which fractions will contain actin and which will contain myosin, and predict the degree of…
A: All types of muscle tissue contain actin and myosin. Muscle contractions and movements are…
Q: During a 20-minute workout, a person burns 350 calories. What is the number of grams of glucose…
A: In-order to solve this problem, first we to find the amount of ATP generated in the given muscle…
Q: What are the three major pathways that eventually become entry points of molecules into the Krebs…
A: In the Krebs cycle, acetic acid or its equivalent provides energy to the organism through oxidation,…
Q: The synthesis of phosphocreatine is unfavorable. If dur- ing extended periods of rest, e.g.,…
A: Phosphocreatine (PCr) - It is a phosphorylated creatine molecule that works as transferable reserve…
Q: Upon digestion of starch, isomaltose (an isomer of maltose), one of its degradation products, is…
A: Isomaltose is a disaccharide similar to maltose, but with an α-(1-6)-linkage instead of an…
Q: Which of the following is an anomer of a-D-galactopyranose? CHOH OH H. он он CH,OH OH он H он H H он…
A: Galactose is aldohexose. It is a C-4 epimer of glucose. The cyclic six-membered ring is called…
Q: 10 b. How many different proteins composed of 100 amino acids could possibly exist?
A: The building blocks for the synthesis of proteins are amino acids. 20 different amino acids…
Q: 1.Describe briefly the concept of capillary electrophoresis and sate two ways that it is similar to…
A: Electrophoresis is a laboratory technique used to separate charged molecules like nucleic acids, and…
Q: Which of the following is least soluble in hexane?
A: Fatty acids (FA) are aliphatic chain with carboxylic acid group, which is non-polar molecule. This…
Q: Which use active transport? * water Osugars, e.g., sucrose Omicronutrients, e.g., nitrates O O O
A: Transportation is a necessary, physiological, and natural process that occurs in all higher…
Q: Which enzyme activity of the glycogen debranching enzyme is operating during the release of glucose…
A: The amylo-1, 6-glucosidase catalytic action of glycogen debranching enzyme permits it to hydrolyze…
Q: Which protein(s) are retained on Ni²+-HiTrap Chelating HP? Self-assessment: not retained: retained:…
A: An amino acid residue is joined to a peptide molecule in order to form a protein, which occurs…
Q: Movement of water is controlled by which factor?
A: Movement of water occurs through the xylem cells of plants to reach all parts of plant body . As…
Q: Name at least three substrates for the hexokinase enzyme. Why are humans considered…
A: Part 8. The three substrates for hexokinase enzymes are: glucose-6-phosphate dehydrogenase…
Q: Based on the observations below, make a brief statement on the identity of the sample. Test…
A: Introduction: Carbohydrates are an important source of energy used by all living things. It serves…
Q: Failure of anti-oxidant function results in the hydroxylation of an aromatic acid of Enzyme Z and…
A: Enzymes are highly specialized proteins that have extraordinary catalytic power, greater than that…
Q: efly
A: There are various methods of studying the Enzyme-substrate complex like magnetic resonance…
Q: 2. Use your knowledge of amino acids (and the R groups) and tertiary structures of proteins to…
A: The disease, which is referred to as sickle cell anaemia is caused because of mutation. In this…
Q: Calculate AGo' for the reaction NADH + pyruvate → NAD+ + lactate You are given: Faradays constant F…
A: Electrons from NADH and a proton are used to reduce pyruvate into lactate in lactic acid…
Q: Long explanations are not needed. Direct answers would suffice. a. In the TCA cycle, succinate…
A: The hormone glucagon exerts its effect via stimulation of adenylyl cyclase that leads to production…
Q: 10. Match each chromatographic method with the characteristics of the protein that you would expect…
A: Introduction: Chromatography is an analytical technique in which compounds in a mixture are…
Q: When the cell needs both NADPH and ATP, the most likely utilization of the Pentose Phosphate Pathway…
A: The pentose phosphate pathway synthesizes NADPH and pentose sugars required by the cells. The…
Q: For each molecule of glucose, how many times does the CA cycle proceed? a. thrice b. once c.…
A: The Krebs cycle, also known as the Citric acid cycle, is a sequence of enzyme-catalyzed events that…
Q: Write the basic concepts in metabolism
A: Cells are machinery structures which carry out various complex controlled biochemical reactions in…
Q: Which of the following pairs of amino acids can have intermolecular hydrogen bonding between the…
A: The secondary structure of proteins is stabilized by hydrogen bonding. mainly it…
Q: ACTIVITY 6.1 Complete the table below Source Example of Carbohydrate Function
A: Carbohydrates are a form of macronutrient that can be found in a variety of foods and drinks.…
Q: product of the reaction:
A: Lipids are biological molecules that are insoluble in water but soluble in non-polar…
Q: Glycolysis is regulated primarily by _______________. a. three strongly exergonic, non-equilibrium…
A: The pathway that transform glucose to pyruvic acid is known as glycolysis. The free energy that is…
Q: Do coconut oils contain cholesterol? Justify your answer.
A: Excess cholesterol is generated by a variety of variables, including the fat components of the diet…
Q: In the Krebs Cycle (Citric Acid Cycle), A 4-carbon compound with a 2-carbon unit to yield a 6-carbon…
A: Kreb cycle/ tricarboxylic acid cycle/ citric acid cylce - occur in matrix of mitochondria.
Q: For an enzymatic reaction, the following data were obtained for two different initial enzyme…
A: Enzymes are protein molecules that increase the rate of reactions by decreasing the activation…
Q: Contrary to legend, camels do not store water in their humps, which consist of large fat deposits.…
A: Tripalmitoylglycerol is formed by three palmitoyl-CoA molecules. As a result, it is required to…
Q: pls solve this problem with in 15 to 30 min
A: 1 Hemiacetal in general , when aldehyde can react with alcohol forms hemiacetal. The linear…
Q: lease help me answer questions 71 & 77 71. Which of the following are examples of kinetic energy? A.…
A: Introduction: Kinetic energy: The term energy is the ability to do work or cause change. There are…
Q: Using one of the lower level structures, explain how the protein’s effects one of the higher level…
A: Proteins are biomolecules composed of amino acids. Unlike other biomolecules, proteins have…
Q: How does the mouth achieve initial digestion of carbohydrates? Read and analyze the question and…
A: Carbohydrate digestion begins in the mouth with the mechanical action of chewing followed by the…
Step by step
Solved in 2 steps

- How many stereoisomers are possible for the following monosaccharide? a. 4 b. 8 c. 16 d. 2 e. 1Give the molecular and structural formulae for: –2 examples of structural isomers of monosaccharides –3 examples of stereoisomeric monosaccharides. (Do not include glucose, fructose, galactose)(a) What are the two principal polysaccharides in starch? (b) Which of these polysaccharidescontains unbranched chains of the monosaccharides?
- Identify the monosaccharide represented by each of the following structures. Name each compound.Which of the following define the stereochemistry of alanine (as per the structure shown)? Note: Functional groups arranged horizontally are facing towards the front, and the functional groups arranged vertically are facing towards the back. a) d- b) S- c) R- d) I-what is the name of the illustration complex lipids?
- Name the identity of the carbohydrates.Given the structure of disaccharide below: (a) is it reducing? (b) will it react positively with Benedict's reagent and (c) what specific glycosidic linkage is the name of the disaccharide?Classify the following lipid (choose all that apply for the overall structure, not the individual residues). a. wax ester b. fatty acid c. polyunsaturated d. trans alkene(s) e. steroid f. monounsaturated g. triglyceride h. cis alkene(s) i. saturated

