Q: Describe the bond angles to be found in each of the followingmolecular structures: (a) trigonal plan...
A: (a)The bond angle in trigonal planar structure is found to be 1200. Example: BH3 has trigonal planar...
Q: (a) If you combine two atomic orbitals on two different atomsto make a new orbital, is this a hybrid...
A: Molecular orbitals are formed by the overlapping of the atomic orbitals of two or more atoms that ta...
Q: What reagents are needed to synthesize the following alcohols?
A: Please find below the pic for left product formation.
Q: Two chemists at Dupont found that ICH2ZnI is better than diazomethane at converting a C=C bond to a ...
A: The C – Zn bond is observed in Simmons – smith reagent which has the same polarity as the C – Mg bon...
Q: Draw all the products of the following reaction:
A: Please find below all the possible products of the above reaction.
Q: Writethe names of the reactants and products for the net ionic reactions for the following reactions...
A: a. When Dimercury diacetate and magnesium chloride are mixed in water, no precipitation occurs becau...
Q: What are products of the following reactions?
A: The products of the given reaction can be shown below
Q: calculate the relative rates of effusion of c^12 O^16, C^12 O^17 and C^12 O^18 . name some advantage...
A:
Q: what are the quantum numbers for the last electron in 41Nb ?
A: An atom is mainly composed of three sub-particles; electrons, protons and neutrons. These particles ...
Q: Which carbocation in each pair is more stable?
A: a) Since in compound 1 the carbocation can do resonance with double bond as it is in conjugation wit...
Q: Give the correct IUPAC name for the following compounds
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts fo...
Q: Draw all the enol tautomers for each of the ketones
A: The enol form has a structure containing a carbon – carbon double bond and a hydroxyl group.
Q: Show how each of the following compounds can be prepared using the given starting material, any need...
A: Since you have posted question with multiple sub-parts, we are entitled to ans the first 3 only.
Q: A room temperature white solid is mixed with a room temperature clear liquid resulting in a clear, c...
A: A room temperature white solid is mixed with a room temperature clear liquid resulting in a clear, c...
Q: What is the expected oxidation states for 60Nd?
A: An atom is composed of three main sub-atomic particles; electrons, neutrons and protons. The atomic ...
Q: A sample of an ideal gas has a volume of 2.24 L at 283 K and 1.14 atm. Calculate the pressure when t...
A: Given information: Pressure at initial stage (P1) = 1.14 atm Volume at initial stage (V1) = 2.24 L T...
Q: Use average bond enthalpies (Table 8.3) to estimate ΔH forthe atomization of benzene, C6H6:C6H6(g)--...
A: C6H6(g)--->6 C(g) + 6 H(g) Since in the above reaction we need energy to break 6 C-H and 3 C=C an...
Q: Using any alkene and any other reagents, how would you prepare the following compounds?
A: Hello. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If y...
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant ...
A: According to the Bronsted Lowry acid-base concept, an acid is a substance which loses H+ ions to for...
Q: What stereoisomers are obtained from each of the following reactions?
A: Since you have posted multiple sub-parts, we are entitled to answer the first three only.
Q: 5. When the equation С2НА O2 CO2 + H20 + heat is balanced, the proper sequence of coefficients is 1,...
A: Balanced chemical equation of a reaction is written according to law of conservation of mass. Genera...
Q: Enter a balanced equation for the dissociation of LiBrLiBr.
A: A strong electrolyte is a solution/solute that completely, or almost completely, ionizes or dissocia...
Q: In the formate ion, HCO2-, the carbon atom is the centralatom with the other three atoms attached to...
A: (a) The formate ion (HCO2-) is formed by the sharing of electrons between one carbon atom, one hydro...
Q: Match the formula on the left with its description on the right.
A: Ozone, O3 is a layer present in stratosphere which protects the Earth’s surface from the Sun’s Ultra...
Q: Titration of 22.0 mL of a solution of citric acid (H3C6H5O7) required 15.3 mL of 0.180 M NaOH to rea...
A: Titration of 22.0 mL of a solution of citric acid (H3C6H5O7) required 15.3 mL of 0.180 M NaOH to rea...
Q: The redox reactions taking place in an alkaline battery are shown below. What is the overall cell po...
A: The overall cell potential of the cell is determined by using following formula
Q: The pressure of the atmosphere decreases with increasing altitude in the Choose all that apply. The ...
A: The pressure of the atmosphere decreases in all layers with increasing altitude due to decrease in t...
Q: A 32.5 g piece of aluminum (which has a molar heat capacity of 24.03 J/(°C·mol)) is heated to 82.4°C...
A: Given: Specific heat of water = 4.18 J/g. oC Molar heat capacity of aluminium = 24.03 J/°C·mol Mass ...
Q: Rank the following from strongest acid to weakest acid: CH3CH2N+H3 CH3CH=N+H2 CH3C=N...
A: The molecule which will have most electronegative element which will take the bonded electron form N...
Q: name the structure
A: The alkene consists of five carbon atoms. The double bond is present between the second and third ca...
Q: Name the compound
A: Some rules to follow for writing names organic compounds: Identify the longest carbon chain. This c...
Q: Propose a mechanism for the following reaction:
A: Please find below the reaction mechanism
Q: What is the hybridization of the central atom in (a) SiCl4,(b) HCN, (c) SO3, (d) TeCl2?
A: Before bond formation the mixing of atomic orbital takes place and results to form orbitals of same ...
Q: Describe the alkyne you should start with and the reagents you should use if you want to synthesize ...
A: Please find below the reaction pics to synthesise the mentioned compounds.
Q: How do you solve question 6?
A: The given compound is shown below:
Q: Are the following pairs of structures resonance contributors or different compounds?
A: Hey, since there are multiple sub parts questions posted, we will answer first three questions. If y...
Q: What is the molecular formula for a monocyclic hydrocarbon with 14 carbons and 2 triple bonds?
A: Since it is the hydrocarbon. Hence, it will have only Carbon and hydrogen. Now we know that an alkan...
Q: The drawing below shows the overlap of two hybrid orbitalsto form a bond in a hydrocarbon. (a) Which...
A: (a) This given orbital structure shows us that it consists of two carbon atoms and four Hydrogen at...
Q: The iodine bromide molecule, IBr, is an interhalogen compound.Assume that the molecular orbitals of ...
A: (a) Which valence atomic orbitals of I and of Br are used to construct the MOs of IBr? Answer- From ...
Q: Assign relative priorities to the groups or atoms in each of the following sets:
A: (a)Given set of atoms and groups are as follows, Priority of each of these groups or atoms can be g...
Q: older. Or import from another browser. Import favorites [Review Topics] [References] Some fuel-effic...
A: The amount of gasoline (V) is required to drive the car 9.00 miles is calculated as shown below wher...
Q: Each of the following reactions has two nucleophiles that could add to the intermediate formed by th...
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you. ...
Q: The figure that follows shows ball-and-stick drawings of threepossible shapes of an AF3 molecule. (a...
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and s...
Q: Draw the products of the following reactions. If the products can exist as stereoisomers show what s...
A: Since you have posted multiple sub-parts, we are entitled to answer the first three.
Q: A container was charged with hydrogen, nitrogen, and ammonia gases at 120°C and the system was allow...
A: Given: The given equilibrium reaction at constant temperature is,
Q: How can the following compounds be prepared using ethyne as the starting material?
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only. ...
Q: Ammonia, NH3, reacts with incredibly strong bases to producethe amide ion, NH2-. Ammonia can also re...
A: To find the bond angle we need to find the geometry first for which we will need hybridisation. Sinc...
Q: What reagents should be used to carry out the following syntheses?
A:
Q: Operator Example. Apply the following operators on the given functions: (a) Operator d/dx and functi...
A: (a) Applying the operator d/dx on the function x2 is given below.
Q: Predict the H¬C¬H and C¬C¬C bond angles inpropyne:
A: The H-C-H and C-C-C bond angles in propyne are to be predicted.
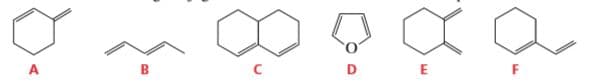
Which of the following conjugated dienes will not react with a dienophile in a Diels–Alder reaction?
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images


