Q: 12. Most monomers are organic materials, atoms are joined in O A. Hydrogen bonding. O B. Covalent…
A:
Q: 4) Trimethyl Aluminium exists as a: A)Dimer b) Monomer d) Hexamer e) Tetramer
A:
Q: 6. Polyethene, aka Polyethylene, is what type of polymer? a. Addition b. Condensation C. Polyamide…
A: Polymers are macromolecules composed of large molecules which can be broken into simpler chemical…
Q: C. Identify the tensile groups in the following molecules: HN "CH, HO. H;C -OH CH, NH,
A: In this question, we will Identify the tensile groups in each of the given compounds. You can see…
Q: The structural isomers C2H5OH and CH3OCH3 would be expected to have the same values for which of the…
A: To select which value for the structural isomer C2H5OH and CH3OCH3 is same Structure isomers have…
Q: Are polymers today becoming more sophisticated in structure?
A: Polymers are the macromolecules that are formed by the repeated units of monomers.
Q: There are E23 molecules in 40.4 grams of antifreeze, ethylene glycol (C2H60).
A: This problem can be solved by knowing that one mole contains 6.022×1023 particles(atoms, ions,…
Q: Define the term biodegradable?
A: Fuel fertilizer, compost, and biogas are all made from biodegradable waste. Biodegradable waste…
Q: Draw the molecular structures of the monomers of:(i) PVC(ii) Teflon
A:
Q: How does the structure of a Polymer affect its function.
A: Here we have to explain how does the structure of a polymer affect its function? We can explain it…
Q: How much ethanol will be consumed to provide 7 x 107 kJ/yr?
A: Given, Molar mass of ethanol = 46 g/mol 1 mole of ethanol reacts with 3 moles of oxygen to produce…
Q: Find the structure of teflon (polytetrafluoroethylene). how is it similar to the structure of…
A: The structure of teflon is,
Q: 8. Molecular compounds tend to ... Mark only one oval. have high melting points have low boiling…
A: Molecular compounds are held together by weak forces. These forces are intermolecular forces. These…
Q: Melamine and formaldehyde combine to form the polymer, Melmac®. How is Melmac® classified and what…
A: Melamine undergo condensation polymerization with formaldehyde to give melmac.
Q: 1. LBA 2. Bromomethane
A: step1. Enolate formation in presence of the base LDA step2. SN2 attack of enolate on alkyl halide
Q: Describe the formation and structure of polymers
A: Polymer : it is the substance which is made of a large number of similar units . when many monomeric…
Q: All polymers contain only one type of monomers. O True O False
A: Given : All polymers contains one type of monomers.
Q: Give one example each of(i) addition polymers(ii) condensation polymers(iii) copolymers.
A: Example of addition polymer, condensation polymer and copolymers has to be given below,
Q: 9. List down the demanded properties of biodegradable polymer materials for biomedical applications
A: We do have some properties of biodegradable polymer materials which are very important for…
Q: What type(s) of intermolecular forces are expected between CH3CH2CHO molecules? H H- Indicate with a…
A:
Q: Give the correct name for structures (1) and (2). (1) (2)
A: The nomenclature of the compounds are given below
Q: (a) Identify the type of polymer given in the following figure. (1) (ii) (b) Elucidate the term…
A: Polymer are very large molecules which is made up of many repeating molecular unit which is…
Q: b. 1. TFCI pyridine 2. NaSCH3 OH С. 1. EtMgCI но Br 2. aq H*
A: TfCl make OH a good leaving group while NaSCH3 is a good base in comparison to a nucleophilie,. So,…
Q: 5. Differentiate between a thermoset and a thermoplastic.
A: Given : Thermoset and thermoplastic. To find: Difference between Thermoset and thermoplastic.…
Q: What are copolymers? What do they consist of?
A: To find: What are copolymers What do they consist of?
Q: Fill in the following blanks with appropriate sentences. 1. The process of moving oil from one…
A: To fill in the blanks: 1. The process of moving oil from one region to another is called…
Q: Describe the important features of the structure of a polymeric solid, such as rubber.
A: Introduction: Polymeric solids are made up of long chain molecules , wrapped around each other.The…
Q: 1. What is the general formula for saturated hydrocarbons? 2. How is the formula changed if one…
A: Alkanes are saturated hydrocarbon whereas alkene and alkyne are unsaturated hydrocarbon. Weight of…
Q: SH2 + H20 +
A:
Q: HO
A: Applying concept of IUPAC nomenclature of organic molecules.
Q: 13. The development of modern plastics started in 1920s using raw material extracted from coal and…
A:
Q: Briefly explain the purpose of each item in the distillation set-up a. Boiling chips – b. DCM –…
A: Distillation is one of the separation techniques. It is used to separate the components from the…
Q: Which is most likely soluble in water? O none of these O decane (C10H22) octane (C8H18) O CB14…
A:
Q: Differentiate between thermoplastic and thermosetting polymers. Give one example of each.
A: Thermoplastic polymers = Thermoplastic polymers are those which can be softened, reshaped at…
Q: Using intermolecular forces, predict which of the following compounds would be most soluble in…
A: The solubility of a compound is determined based on the like dissolve like rule; a polar compound…
Q: H3C OCH3
A: Given,
Q: Identify the Classification of Hazardous Wastes. example: Dichloromethane - Halogenated organic…
A: The waste that has substantial or potential threats to public health or the environment is known as…
Q: 1. Differentiate between thermoplastic and thermosetting polymers.
A: Thermosetting plastics and thermoplastics are both polymers, but they behave differently when…
Q: What is the vesper structure for H2Te
A: VSEPR Structures are drawn according to the theory which says that electron-pairs arrange themselves…
Q: Describe how this "dried product" is often different from a crystallized product. (Describe the two…
A: Answer: Crystallization is the process of converting a liquid substance into its solid form.
Q: b. Are polymers today becoming more sophisticated in structure? Why or why not?
A: A question about more sophistication in the structure of recent polymers.
Q: .Explain the term polymerization with two examples.
A:
Q: which of these compounds have the highest boiling point? and which one is the lowest? (does it have…
A: To solve this problem we have to know about the Boling point .
Q: One kind of polyester is a condensation copolymer formed between terephthalic acid and ethylene…
A: The monomers given are,
Q: The boiling point of hydrocarbons decreases with increase in branching. Give reason.
A: The boiling point of hydrocarbons decreases with an increase in branching. Their reason has to be…
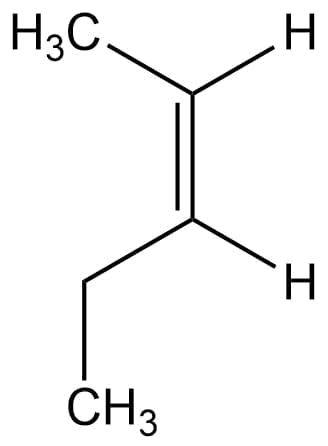
name the structure
Trending now
This is a popular solution!
Step by step
Solved in 2 steps


