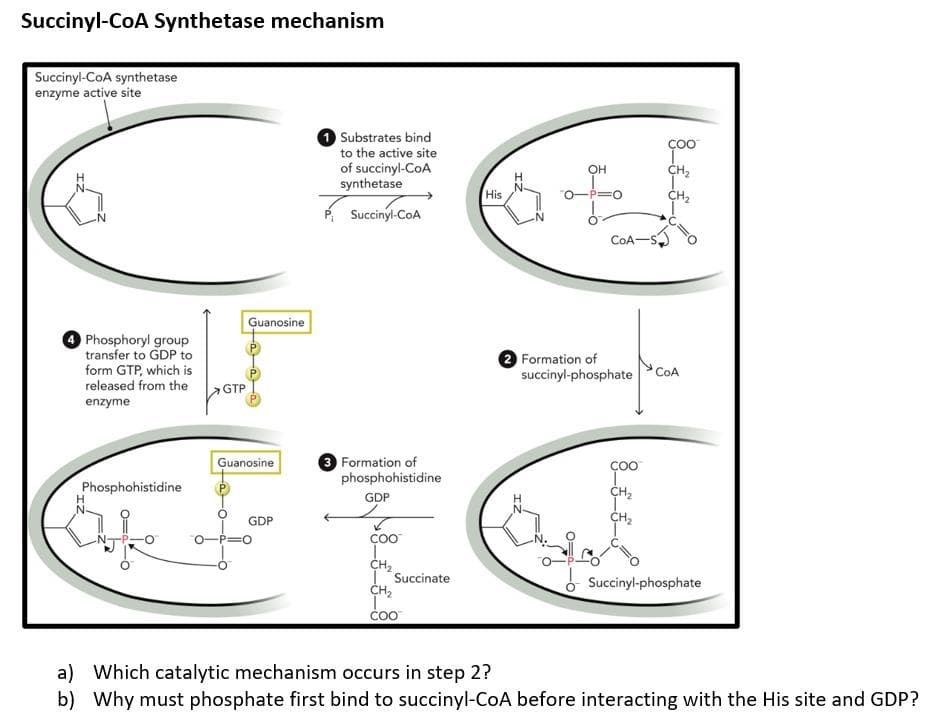
a) Which catalytic mechanism occurs in step 2? b) Why must phosphate first bind to succinyl-CoA before interacting with the His site and GDP?
Q: 10. Isoniazid (MAO inhibitor) is prescribed to the patient with Parkinson discase. What does cause…
A: Parkinson's disease is commonly seen in adults after the age of 50. Parkinson's disease is known to…
Q: Which of the following statements is correct about oxidative pentose phosphate pathway? Group of…
A: Oxidative pentose phosphate pathway: . This is the first phase in pentose phosphate pathway in which…
Q: Which of the following glycosidic linkages is hydrolyzed by the a-amylase?
A: α amylase enzyme belongs to the enzyme group of amylase. Amylases hydrolyses the α-1,4-glycosidic…
Q: In cholesterol, to which ring of the steroid system is the hydroxyl group attached? B O A
A: cholesterol is a sterol and is the primary compound from which synthesis of many steroid hormones,…
Q: Explain the biochemical and subsequent medical consequences of a deficiency of Myoadenylate…
A: Myoadenylate deaminase insufficiency is a metabolic muscle disorder in which the muscle cell's…
Q: what transport proteins are involved is getting ca2+ out of cytosol
A: Proteins are composed of amino acids, which are bound together by peptide linkage. Amino acids…
Q: Polysaccharides may gel through a variety of different mechanisms. Which of these statements is…
A: Carbohydrates are divided into 3 classes monosaccharides, disaccharides, and polysaccharides.…
Q: Using DEAE-cellulose as ion exhange resin, indicate the starting and ending pH for the narrowest…
A: DEAE-cellulose contains an diethylaminoethyl group and is positively charged at neutral pH.
Q: Competitive inhibitors are: Select one: O a. bind to another part b. causing the enzyme t bind to…
A: Inhibitors are molecules that bind to enzyme and block it's activity. Enzymes inhibitors are…
Q: Visceral fat loss is often associated with increased insulin sensitivity. Assuming that with the…
A: D. -10.6
Q: In the following diagram, A and B are two Okazaki fragments generated during DNA replication. Solid…
A: Okazaki fragments are short stretches of DNA on the lagging strand, which is synthesized in the…
Q: Only one of the statements below is correct; which one? O Two solutions are hypotonic when they have…
A: Tonicity is a parameter of the effective osmotic pressure gradient, which is the difference in water…
Q: Describe the mutational event that produces the MYC oncogene in Burkitt’s lymphoma. Why does the…
A: Burkitt lymphoma is a type of non-Hodgkin lymphoma that affects adults and children. NHL is a…
Q: The table below provides kinetic information when ADH is reacted with ethanol alone, NAD+ alone, and…
A: Enzyme kinetics is the study of an enzyme catalyzed biochemical reactions. Usually it is studied by…
Q: HO CH, H. CH2 OH I-
A: An amino group and an acid group-containing organic molecules are called Amino acids. when…
Q: Question 6 Match the following lipids with their functions v Bile acids A. signaling molecules…
A: Lipids are biomolecules that include fats, waxes, oils, hormones, and certain components of…
Q: In the iodine clock experiment, if you increase the volume of vitamin C used, would the reaction…
A: The iodine clock experiment or reaction is the very famous experiment for the understanding of…
Q: What is the most stable nitrogenous base pairing?
A: Base can be purines (adenine and guanine) two ring structure and pyrimidines (cytosine and thymine).…
Q: Which of the following named reduced coenzyme Select one: O a. NAD O b. FAD O c. ATP O d. NADH2
A: NAD : Nicotinamide adenine dinucleotide FAD : Flavin adenine dinucleotide ATP : Adenosine…
Q: What are the description of the ff? A. Enzyme influence on reaction velocity B. Effect of…
A: Enzymes are biological catalysts (also called biocatalysts) that accelerate biochemical reactions in…
Q: Match the following descriptions to the given choices Synthesized from a steroid molecule A. Vitamin…
A: Vitamin is an organic substance and an essential micronutrient that an organism needs in small…
Q: Question 4 Match the following descriptions to the given choices v Synthesized from a steroid…
A: Vitamins are essential nutrients that are important for the protection of body from diseases.…
Q: Identify chemical reactions involved in food preparation and preservation. Explain how these…
A: The oldest method of food preservation includes drying, refrigeration and fermentation…
Q: 9. Which of the following statements about trypsin, chymotrypsin, and elastase are true? A. They are…
A: Trypsin, Chymotrypsin and Elastase all three are protein digesting enzyme which uses its active site…
Q: Bigger size sample would result to better quality spectra compared to smaller sample with small size…
A: Attenuated Total Reflection - Fourier Transform Infrared Spectroscopy is a technique to study the…
Q: Question 8 All of the following have terpene structure, except Cholecalciferol Squalene Carotenoids…
A: Terpenes generally are composed of two, three, four, or six isoprene units. Many important terpenes…
Q: In the Biuret Assay for protein concentration determination, the role of sodium potassium tartrate…
A: The biuret test is a chemical test that can be performed to determine whether an analyte has peptide…
Q: When do plants usually undergo mitochondrial respiration?
A: Mitochondrial respiration is commonly known as cellular respiration. This is the process of energy…
Q: reaction
A: Hess law is mainly used to find out specific unknown enthalpy change. It is particularly useful for…
Q: Explain which of the following substances ATP, CoA-SH, FAD and NAD+ have the subunits in their…
A: The nucleic acid polymer has nucleotide as its monomeric unit. synthesis of nucleotides is an…
Q: Why DNA melting is required in PCR? Briefly explain how PCR can be used to detect DNA mutation.
A: PCR is polymerase chain reaction used to amplify the DNA . It was first developed by Kary Mullis. It…
Q: What part of the protein sequence leads to different functions
A: Proteins are peptides , composed of monomers of polymers,Protein function is directly related to the…
Q: Write an equation to describe the catabolism of an aerobic hydrogen oxidizer
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What is the molecular weight of a linear polysaccharide consisting of 7 galactose monomers and 1…
A: Glucose and galactose are monosaccharides that are also referred to as simple sugars. They represent…
Q: Illustrate and summarize ketogenesis.
A: Ketogenesis is one of the biochemical pathways. The process of ketogenesis occurs in the liver. The…
Q: production of Ribose 5-phosphate in the Pentose Phosphate Pathway is needed in the synthesis of…
A: Pentose phosphate pathway, also called the hexose monophosphate shunt or phosphogluconate…
Q: Acetyt CoA Oxaloscetate CoA NADH Citrate NAD Isocitrate Malste Pumarate NAD NADH FADH, FAD a-…
A: TCA cycle is the tricarboxylic acid cycle which is second step in cellular respiration that occurs…
Q: Explain the mechanism of Warburg effect and how it benefits cancer cells
A: Cancer means uncontrolled cell growth. This uncontrolled cell growth may cause a lump of cell or…
Q: In plants, under what solution conditions are Calvin cycle and gluconeogenesis enzymes most active?…
A: Gluconeogenesis is the process of Synthesis of glucose from non Carbohydrate sources like aminoacids…
Q: Match the following lipids with their functions
A: Lipids are the various organic compounds which are insoluble in water. These are- fats, waxes,…
Q: For the electron transport chain, all are inhibitors except: Select one: O a. Antimycin A O b.…
A: Most of the free energy released during the oxidation of glucose to carbon dioxide is retained in…
Q: (continued) B. From a metabolic engineering perspective, why did the researchers heterologously…
A: Zymomonas mobilis is a gram negative facultative anaerobic bacteria. It ferments the pyruvate into…
Q: Question 4 Match the each enzyme deficiency with their corresponding disease B-hexosaminidase A A.…
A: Enzyme deficiency results in certain metabolic disorders and results in serious diseases.
Q: Food Sample marshmallows Pumpkin seeds cracker Dried cranberries рорсorn Rice cake almond
A: Calorific value of foods is based on their nutrient content. Calorific value of foods is the total…
Q: 7. Describe the falient features and functions of steroids.
A: Steroids are biologically active compounds. Steroids are found in plants, animals and fungi.…
Q: Which of the following refers to the test performed 2 hours after an overnight-fasted patient was…
A: Different tests are used to test the blood glucose level.
Q: 8. Which of the following amino acids in hemoglobin accepts proton H+ when red blood cells reach…
A: Hemoglobin is a conjugated protein with four polypeptide chains. It contains two identical alpha and…
Q: You receive a tube containing 18.8 nmol of lyophilized (freeze dried) primers to be used in PCR. How…
A: Primers are a short nucleic acid sequence that provides a starting point for DNA synthesis. I…
Q: The steps of glycolysis between glyceraldehyde 3-phosphate and 3-phosphoglycerate do NOT involve:…
A: Glycolysis is defined as the process of breaking down the compound called "glucose" to create…
Q: Question 23 18:1cA9 O w-9 fatty acid O oleic acid
A: Polyunsaturated fats, such as omega-3 fatty acids, are a form of fat that body cannot produce.…
Step by step
Solved in 3 steps

- Distinguishing the Mechanisms of Class I and Class I Aldolases Fructose bisphosphate aldolase in animal muscle is a class 1 aldolase, which forms a Schiff base intermediate between substrate (for example. fructose-1, 6-bisphosphate or dihydroxyacetone phosphate) and a lysine at the active site (see Figure I8.12). The chemical evidence for this intermediate conies from studies with aldolase and the reducing agent sodium borohydride, NaBH4. Incubation of the enzyme with dihydroxyacetone phosphate and NaBH4 inactivates the enzyme. Interestingly, no inactivation is observed if NabH4 is added to the enzyme in the absence of substrate. Write a mechanism that explains these observations and provides evidence for the formation of a Schiff base intermediate in the aldolase reaction.Extending the Mechanism of Methylmalonyl-CoA Mutase to Similar Reactions Based on the mechanism for the methylmalonyl-CoA mutase (see problem 14), write reasonable mechanisms for the following reactions shown.Regulation of Glutamine Synthetase by Covalent Modification Suppose at certain specific metabolite concentrations in vivo the cyclic cascade regulating E. coli glutamine synthetase has reached a dynamic equilibrium where the average state of GS adenylylation is poised atn=6. Predict what change in nwill occur if: [ ATP ] increases, PIIA/PIID increases, [ -KG ]/[ Gln ] increases, [ Pi ] decreases.
- Modeling the Regulation of AcetyI-CoA Carboxylase Based on the information presented in the text and in Figures 24.4 and 24.5, suggest a model for the regulation of acetyl-CoA carboxylase. Consider the possible roles of subunit interactions, phosphorylation, and conformation changes in your model.Using the ActiveModel for aldose reductase, describe the structure of the TIM barrel motif and the structure and location of the active site.The mechanism for the conversion from alpha kytoglutarate into succinyl CoA by alpha-ketoglutare dehydrogenase is analogous to the pyruvate dehydrogenase mechanism. Draw and show the major intermediates and arrow pushing for the enzyme-catalyze process for the conversion of alpha-ketoglutarate to succinyl CoA. Note: the carbanion of thiamine pyrophosphate nucleophillically attacks C-2 of the alpha-ketoglutarate, i.e., the carbonyl carbon of that substrate.
- The products of pyrimidine base catabolism, β-alanine and β-aminoisobutyrate, can be further degraded to acetyl-CoA and succinyl-CoA, respectively. Can you suggest the types of reaction required to accomplish these transformations?ATP is a (+) allosteric effector, and CTP is a (-) allosteric effector of theenzyme ATCase. Both of these heterotropic effectors bind to the regulatorysubunits on ATCase. The substrates of ATCase, aspartate and carbamoylphosphate, bind the enzyme active site with positive cooperativity (i.e.,they exert a “+” homotropic effect on activity). As the concentrations ofthe substrates change from values where [S] ≪ KM to values where [S] issaturating ([S]≫ KM), how will the binding constants for each of the twoallosteric effectors change? In other words, does ATP bind ATCase withhigher affinity when [S] is low or high? Does CTP bind ATCase with higheraffinity when [S] is low or high?Please draw out mechanism The formation of fructose 1, 6- biphophste from dihydroxyacetome phospahte (DHAP) and Glyceraldehyde 3- phosphate (GAP) catalyzed by alsolase.
- Explain biochemical pathways mechanistically. Describe the β-oxidation pathway. Describe the conversion of propionyl CoA to succinyl CoA. Describe the substrate activation processes in the urea cycle (CPS 1 and arginosuccinate synthetase).Digestion of plant materials generates phytanate (derived from chlorophyllmolecules; (a) Explain why phytanate cannot be catabolized by β oxidation.(b) Instead, phytanate is linked to CoA, then a peroxisomal dioxygenaseconverts phytanoyl-CoA to 2-hydroxyphytanoyl-CoA. Draw the structure of this intermediate. (c) 2-Hydroxyphytanoyl-CoA lyase generates an aldehyde (pristanal) and formyl-CoA. Draw the structure of pristanal.Some bacteria use the citric acid cycle intermediate, α-ketoglutarate, plusacetyl-CoA, as the starting point for lysine biosynthesis. The first partof this biosynthetic pathway uses the same chemical strategy found inthe citric acid cycle. Propose a four-step pathway for the conversion ofα-ketoglutarate to 2-oxoadipate. Draw the three missing intermediates, andindicate the chemistry involved in each reaction. Include any cofactors thatyou think might be required for specific steps.

