Q: How functional group determines reactivity ?
A: Organic compounds are the compounds which are mainly composed C and H atoms. The branch of chemistry…
Q: Label the following pairs of compounds as: a. identical b. constitutional isomers c. neither
A: If the compounds have the same molecular formula but have a different bonding arrangement then they…
Q: Label attached pair of alkenes as constitutional isomers, stereoisomers, or identical.
A: The given isomers can be named as follows,
Q: Considering rotation around the bond highlighted in red in each compound, draw Newman projections…
A:
Q: Draw all constitutional isomers formed by monochlorination of attached alkane ?
A: In monochlorination process, the hydrogen atom in an alkane compound is replaced by the chlorine…
Q: Explain why CH3CH,NHCH3 has a higher boiling point than (CH3);N, even though they have the same…
A: When vapor pressure of a substance becomes equal to the atmospheric pressure then the temperature at…
Q: A
A:
Q: Which compound in each pair has the higher boiling point?
A: Boiling point is the temperature of a compound at which a liquid is changes into gas phase. The…
Q: Give the IUPAC name for each compound.
A: (a) CH3-CH2-CH2-CH2-CH(CH3)-CH2-CH3 IUPAC name : 3-methylheptane (b) IUPAC name :…
Q: Q1. Name each of the following structures by the IUPAC system? B. C.
A: Given compounds are : IUPAC names of the given compounds = ?
Q: chemical compound whose structure is based on C and has many H
A: Organic compounds
Q: Given each of the structures provided, write the corresponding IUPAC name. (a) (b) (c)
A:
Q: B. Give the IUPAC names for each of the following molecules.
A: While giving IUPAC name to a compound, consider the longest chain and start numbering from highest…
Q: Draw the structure corresponding to following IUPAC name. cyclobutylcycloheptane
A: The structure of the given IUPAC name can be drawn as
Q: Ili
A: The given compound, (1S,2S,4R)-1-isopropyl-2,4-dimethylcyclohexane is in planar form. It contains…
Q: Total number of stereoisomer :-
A: Stereoisomerism called spatial isomerism in which molecules have same molecular formula but differ…
Q: Convert each of the following structures into its more stable chair form. One structure represents…
A: The compounds which have the sample molecular formula but differ in the structural formula are…
Q: What force(s) is(are) responsible for the higher heat of vaporization of benzene compared to…
A: Heat of vaporisation is the amount of energy needed to convert the liquid into gas without rising…
Q: Draw the two chair conformations of menthol, and tell which is more stable
A: Menthol is the derivative of cyclohexanol. Chemical formula for menthol is C20H10O and IUPAC name…
Q: Give the IUPAC names of the following : a) b) c) tot
A:
Q: Br f.
A: The compound has only IUPAC name
Q: 'CH3
A: Given that compound contains a ten carbon cycloalkane, hence the name of the compound is derived…
Q: Label the following pairs of compounds as: a. identical b. constitutional isomers c. neither Place…
A:
Q: Vrie the IUPAC name for the following organic compounds: a) b) H3C H3C- CH3 CH3 H3C CH3 H3C- -CH3…
A: Since you have asked multiparts questions, we will solve the first three sub parts questions for…
Q: Compound A has a heat of combustion of -2300 kJ/mol. Compound B, a stereoisomer of Compound A, has a…
A:
Q: pnysical ame molecular formula but different structures and different chemical prop Ethanol Dimethyl…
A:
Q: 11
A: Note : Assign the R,S nomenclature to the organic compounds. Then write the substitution name first…
Q: B. For each write the correct IUPAC name b)
A: As per the rule, only the first three parts can be answered
Q: .A There is no relationship between them .B equal in boiling point .C higher than the boiling point…
A: Boiling point is defined as the amount of heat required to vaporize a liquid. this depends on…
Q: B. For each write the correct IUPAC name
A: Given compounds are : Give the IUPAC name of the following = ?
Q: Given each of the structures provided, write the corresponding IUPAC name. (a) (b) (c) (d)
A: International Union of Pure and Applied Chemistry that is also known as IUPAC. It is used for naming…
Q: how many isomers of chlorobutane and there Synthetic formula
A: There are total four possible isomers of chlorobutane.
Q: Answer below question for A and B depicted in the ball-and-stick models. Que: What is the IUPAC name…
A: IUPAC nomenclature Identify the longest carbon chain Identify the functional groups and…
Q: What does Zaitsev's rule state? As the degree of substitution around the C=C of an alkene decreases,…
A: We have to tell which option is correct from given choices of options in accordance with zaitsev…
Q: Draw the structure corresponding to following IUPAC name. sec-butylcyclopentane
A: sec-butylcyclopentane 1. Secondary carbon should contain one Hydrogen atom 2. Butyl has four…
Q: Which Newman projection corresponds to the compound below:
A: Given : structure of organic molecule.
Q: draw most stable and least stable conformations for each
A: Given that we have to draw,these molecules having less stable and more stable Newmaan projections.
Q: Label each as aromatic, antiaromatic, or nonaromatic
A: Condition Aromatic. 1) Compound should be planer that means each carbon should be SP2 hybridised…
Q: give me the proper IUPAC Names for the compounds represented by A), B), C) and D)
A:
Q: Circle the compound that would have the lowest energy at its most stable conformation and box the…
A: We have to circle the compound would have the lowest energy at its most stable confirmation as…
Q: Classify each carbon–carbon double bond as isolated or conjugated.
A: a. Both the double bonds are conjugated. b. Both the double bonds are conjuagted. c. a,b is…
Q: Alkenes are not soluble in water because they are
A: The correct reason for alkenes not being soluble in water is to be selected- polar hydrophilic…
Q: CI
A:
Q: Which alkyne has the smaller heat of hydrogenation, HC=CCH,CH,CH, or CH,C=CCH,CH,? Explain your…
A: Heat of hydrogenation is enthalpy of catalytic hydrogenation. Heat of hydrogenation is depend on…
Q: . A. Draw ethane using wedge and dash notation in its highest energy conformation. B. Give the name…
A: The energy of a molecule is inversely related to the stability of that molecule which means the…
Q: Which alkyne has the smaller heat of hydrogenation, HC ≡ CCH2CH2CH3 or CH3C ≡ CCH2CH3? Explain your…
A: An internal or more substituted alkyne is more stable than a terminal or less substituted alkyne.…
Q: Which has a higher melting point between tetracosane and eicosane? Explain
A: To know this we have to know the structure of the compounds . With increasing in molecular weight of…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

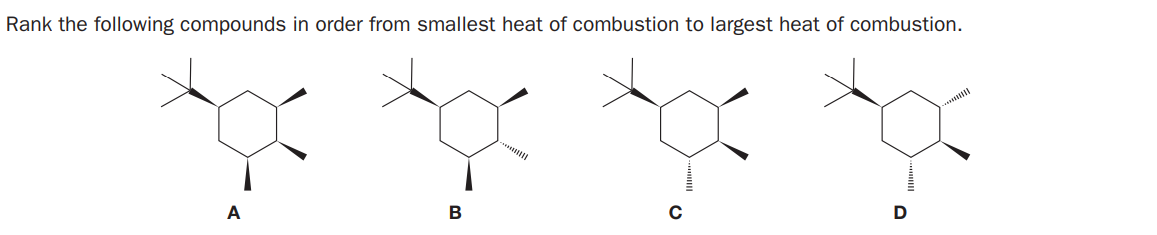
- How do you rank alkenes in order of their heats of hydrogenation? How do you know an alkene's heat of hydrogenation?Rank the following compounds from highest boiling to lowest boiling:Rank the following compounds according to heat of combustion with 1 being greatest exothermicity and 4 least exothermicity of combustion.
- a. Which is the most stable: 3,4-dimethyl-2-hexene, 2,3-dimethyl-2-hexene, or 4,5-dimethyl-2-hexene? b. Which compound has the largest heat of hydrogenation? c. Which compound has the smallest heat of hydrogenation?(a) which if the structure of trans-1,2-dimethylcyclopentane? (b) which is the most stable conformation of 1-bromo-2-ethylcyclohexane? (c) which is the least stable conformation of 1-bromo-2-ethylcyclohexane? (d) which is the more stable configuration of 1,3-dimethylcyclopentane? *Et = ethylLable out / Draw out all the strains each cyclohexane below contains
- Draw both chair confirmations of the substituted cyclohexane drawn below. Be certain to actually do a ring flip. Also be certain to very clearly distinguish axial from equatorial positions. Then circle the more stable conformation. Draw a planar representation of a stereoisomers which would have a higher heat of combustion and lower heat of combustion.Draw the most stable conformation of (a) ethylcyclohexaneState Cyclohexane with One Substituent ?

