а. 4Be + 2 He → + X on b. 3927CO +X → 56 25MN + 42ɑ с. 14,Ве +'on 1416C + X d. 2811Na + 2,H → X + 'H e. 4019K → B + X е. 137 55CS → 137 '56Ba +X
Q: Average morality of NaOH= Dillo M+ 0,1|3 M + 0.1124 M 0.3354M 3. EO.112 M
A: Precision is the degree of closeness between the various experminental data obtained. Precision can…
Q: 17. ZnSO4 + Lİ2CO3 →_ ZnCO3 + LizSO4 18. V205 + _ CaS → СаО + —VzSs 19. Mn(NO2)2 + _ BeCl2 →…
A: The given unbalanced equations are as follows --- ZnSO4 + Li2CO3 --> ZnCO3 + Li2SO4…
Q: Concentration of Solutions Children's toys and jewelry were assessed for their contamination by…
A: Solution: Given : molecular wt. of As = 74.92 g/mol…
Q: Considering the chemical equation :below C2H3Br3 + 11 O2 → 8 CO2 + 6 H2O + 4 6 Br2 What is the…
A: The maximum amount of Br2 is determined using mole mole analysis on given chemical reaction.
Q: 23. Ammonia is synthesized from hydrogen and nitrogen. N2 (g) + 3 H2(g) - -> 2 NH3 (g)lf 5.00 L of…
A: Given that: N2(g) + 3H2(g) ....> 2NH3(g) Volume of N2(V) = 5.00L T = 298 K P = 3.00 atm
Q: 2. Mass of crucible and cover, second heating 51.2235 52.2083 3. Mass of covered crucible plus…
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: +3Ca (OH) acs)→ 6H20(1) + Cas (POw). (s) b) +3CACOH) 2cs)
A:
Q: 17. 75.21 °C, Ca(s) + 2 H,SO, () → CaSO,(s) + SO2 (g) + 2 H20((0) 18. 982 K, C¿H2 (9) + 5 N,0(g) → 2…
A: Given Reaction C2H2(g) +5N2O(g) →2CO2(g) +H2O(g) +5N2(g)To calculate ∆G° at 982 k∆G° = ∆H°-T∆S°∆H° =…
Q: Saved cept- Calculate mass of Mg to produce hydrogen gas Magnesium metal is reacted with…
A:
Q: Ozlg) + 4H*(ag) + 4e = 2H,0 +1.23 Br2(aq) + 2e = 2Br (aq) +1.07 NO, (ag) + 4H*(ag) + 3e = NO(g) +…
A:
Q: A chemist adds 365.0 mL of a 2.1 mol/L nickel(II) chloride (NiCl,) solution to a reaction flask.…
A: Molarity is the moles of substance present per unit volume. Molarity = moles of solute / volume By…
Q: HO. 36 o and 5 rT O 21 o and 5 t 50 o and 5 n
A: The question is based on the concept of chemical bonding. we have to calculate how many Sigma and Pi…
Q: Dinitrogen monoxide (N₂O; nitrous oxide) is a greenhousegas that enters the atmosphere principally…
A: Isotopes possess same number of protons but differ in number of neutrons thus, they have different…
Q: Magnesium burns in air with a dazzling brilliance to produce magnesium oxide: 2Mg (s) + O2 (g) 2M9O…
A: From the given reaction we can see that 2 moles of Mg burns to produce 2 moles of MgO hence moles…
Q: Cu(k) + HNO3 → Cu(NO3)2 + NO2(g) + H2O(s) at 735 mmHg pressure at 28 ˚C, 4.95 cm3 of copper (d= 8.95…
A:
Q: A 0.0715 g sample of a material was analyzed for thiourea (CS(NH2)2, MM = 76.12). It was found to…
A:
Q: 8Ca → 3. 65 Sc 65 4. Be + He C + -> P + H 31 15 32 15 5. 16S + Hе 35 6. 1ci +
A: Dear student, As you have asked multiple question but according to guidelines we will solve first…
Q: Br= ? % NaBr =y g i g of AgBr(s) = y×187.77 102.89 ×143.32 %3D 58.44 87.77 xx100 = 0.5260 g % NaCl…
A: NaCl + AgNO3 → AgCl + NaNO3 NaBr + AgNO3 → AgBr + NaNO3 Let the moles of AgCl formed be x and…
Q: 4NH,() + 50,()= 4NO() + 6H,O(g) Procice Exercises 6.1 4NH,(2) + 50.(e) 4NO() + 6H,OI b. H,CO,(ag) =…
A: Equilibrium constant can be expressed in terms of concentration ( Kc ) or pressure ( Kp). Kc is the…
Q: 1.Mnzt +NAOH 12.Mn2++ HNO2 3. Mnzt+ NHs 4.Mnzt +H2S 1. N²++ NaOH 2.Nizt+ HNO3 3. NH3+Ni24 4.…
A:
Q: CIF 3(9) AH = ? 2.) CIF9) + F2(g) AH = - 49.4 kJ 20F2(9) Ozlg) + 2F2(9) AH= 205.6 kJ Cl,0(9) +…
A:
Q: Question 8
A:
Q: A sample of propane gas, C3H8, was combusted to produce energy according to the equation: C3H8…
A: Given : Molecules of CO2 produced = 1.35 x 1024 Density of water = 1 g/cm3 Also, molar mass of…
Q: The percentage of argon gas in dry air A. 0.035% B. 0.065% C. 20.9% The percentage of other gases in…
A: Composition of gases in dry air is 1 O2 20.9% 2.N2 78.1% 3 Ar 0.93%
Q: MCQ 27: Avogadro's constant is the numb A. 6.02 x 1023 В. 6.03 х 1032 C. 1.2 x 10 D. 1.23 x 10 2
A: The question is based on mole concept. we have to identify the value of avogadro number.
Q: 13. 60 grams of L:OH is Gadded G.9gof waher at 25 The temperature incuesses to 45.5°, What is the…
A: LiOH →Li+ + OH- Heat releases due to dissociation of LiOH in water.
Q: Safari File Edit View History Bookmarks Window Help * 20% [4) Mon 4:19 PM Q. A www-awn.aleks.com…
A: Given data:
Q: Considering the chemical equation below: 4 C3H5BR3 + 17 02 12 CO2 + 10 H20 + 6 Br2 What is the…
A:
Q: pH = 7,36 PCO₂=36 mmol/l 2 SB= 19,5 mmol/l BB=39 mmol / BE=-5 mmol/l
A: The concept is in respect to Arterial blood.
Q: of a Hydrate 23.43 29.24 26.31 1. Mass of crucible 2. Mass of crucible and hydrate 3. Mass of…
A: The question is based on the concept of molecular formula determination. we have been given…
Q: What is the concentration of a 62.0 % (w/w) concentrated nitric acid (HNO3, Mr = 63.0 g/mol)…
A: Given: % (w/w) of HNO3 = 62.0 % Density of solution = 1.32 g/cm3 = 1.32 g/mL…
Q: Cgrafito + 2H2 CH4(9) CH4(g) + 202(g) CO2(9) + 2H2Ou) ACH°= -802.37 kJ/mol Cgrafito + O2(g) CO2(9)…
A: Applying Hess's law and with help of data given it is determined that formation∆HCH4.
Q: (a) 174Hf→ 170yb + ? chemPad XX→ b) 60 Co→ 60 Ni+? chemPad XoXº ↓ Help Greek Help Greek
A: a) H174f→Y170b + H24e b) C60o→N60i + e-10 c) B210i→P210o + e-10 d) P239u→U235 + H24e
Q: At elevated temperature NaHCO3 is quantitatively converted to Na2CO3. By calcining 0.3592 g of an…
A:
Q: The reaction for the Haber process, the industrial production of ammonia, is N2(8) + 3H2(8) –…
A:
Q: 14 14 4. 6C7N+ O a. 14 O b. 0 1H OC. 1e 0 Od le d. 1N
A: To solve this problem we have to complete the given radioactive reaction .
Q: Draw the organic products of each of the following reactions.
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: given that 4 NH3 + 5O2 -> 4NO + 6H2O if 3.00 mol NH3 were made to react with excess of oxygen gas,…
A: The Equation is given as 4 NH3 + 5O2 -> 4NO + 6H2O The amount of H2O formed has to be…
Q: Example Sample: NaCl+NaBr + inert = 1.000 g excess AGNO; U Ag: 107.87, Cl: 35.45 AgCl(s) + AgBr(s) =…
A: According to the law of chemical equivalence, in a chemical reaction, the number of equivalents that…
Q: (c) 174Hf – 170yb + ? chemPad Help Greek - (d) 192pt - 1880s + ? chemPad Help Greek - 1L
A: Loss of 1 Alpha particle causes decrease in mass number (A) by 4 units and decrease in atomic number…
Q: For the reaction 4AB = 2A2+ 4B, Kc =6.90.11 5 Therefore Kc for the A2+ 2B= 2AB IS (äbäi 1) 0.381…
A: For the given reaction 1, the expression of Kc will be Kc1 = [A2 ]2 X [B]4 / [AB]4 = 6.90 where [A2…
Q: 7.50 g of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have…
A: Given:Mass of CO2 = 10.86 g.Mass of H2O = 5.33 g.Molar mass of CO2 = 44 g/mol.Molar mass of H2O = 18…
Q: CUCN OR SN/HCI AIC (2) SNHC options. 80212 CCN Cent KMnOt → B HNO3 H2804 Zn(Hg) Hel Brzihn <CH₂H…
A:
Q: Sulfate (SO2) is typically determined by dissolving in H₂O followed by precipitation as BaSO4 via…
A:
Q: What is -30°F in K? Зок O 30K O 303 K O-30 K O 239 K 04K
A: To convert F to K, Subtract 32 from the Fahrenheit temperature. Multiply this number by 5. Divide…
Q: Calcium ΔHof (kJ/mol) ΔGof (kJ/mol) So (J/mol K) Ca (s) 0 0 41.4 Ca (g) 178.2…
A:
Q: what is Dimitrii Mendeleev (1869)
A:
Q: Given = 3.0 M Ca(oH)2 & 2.5L Looking For = g CacOH)z mol = M x L = (3.0MX25 L) mol= 7,5 mol Cal0H )2…
A:
Q: Suppose we performed Lab 1 examining two different metals: A and B. Metal B has a larger molar mass…
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to…
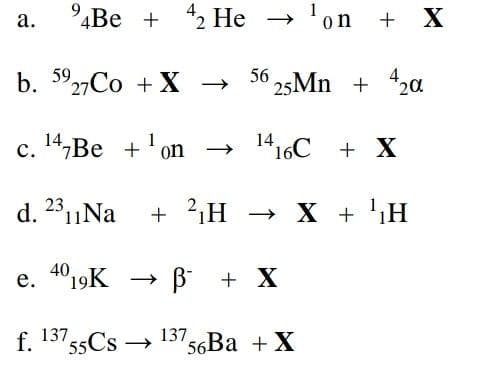
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- An important process for the production of acrylonitrile (C3H3N) (U.S.production is greater than 109 lb) is given by the following reaction: 2C!H"(?) + 2NH!(?) + 3O#(?) H⎯⎯⎯J 2C!H!N(?) + 6H#O(?)A 150.-L reactor is charged to the following partial pressures at 25°C: ? = 0.500 MPa? = 0.800 MPa ? = 1.500 MPa What mass of acrylonitrile can be produced from this mixture (MPa = 106 Pa)?1) Commercial fuming Sulphuric acid (Oleum-H2S2O6) is 99.9% solution. Please convert it into molarity.2) Find out the Volume (dm3) of product (gas) at RTP when 0.58 M, 150 mL NaOH (aq.) reacts with 350 mL, 0.25 NH4Cl.There is actually something im confused about, when you calculating V0 for S0=200uM --> V = 0.48*200/ 2.42 + 1 Why are you only using the S0 value=200uM one place in the equation and then using the S0 value from given data the second place in the equation? Shouldnt S0 be 200 uM both places in the equation?
- Your body deals with excess nitrogen by excreting it in the form of urea, NH2CONH2. The reaction producing it is the combination of arginine (C6H14N4O2) with water to give urea and ornithine (C5H12N2O2). C6H14N4O2 + H2O ? NH2CONH2 + C5H12N2O2 [Molar masses: 174.2 18.02 60.06 132.2] If you excrete 95 mg of urea, what quantity of arginine must have been used? What quantity of ornithine must have been produced?1)Please express the normal salt (NaCl) concentration in body fluid into molarity (mM).2) Commercial fuming Sulphuric acid (Oleum-H2S2O6) is 99.9%. solution. Please convert it into molarity.3) Find out the Volume (dm3) of product (gas) at RTP when 0.58 M, 150 mL NaOH (aq.) reacts with 350 mL, 0.25 NH4Cl.4) The above reaction has the product Ammonia, which when dissolved in 650 mL ethanol makes an alkaline ethanolic solution. Find its molarity (M) 5) Calculate the adult dose as per the BW of the baby. (Child dose-50 mg and the BW of the baby is 48 lb (British pound) (1lb=0.453 Kg)Inorganic Chem Please help answering this question Use equation V = (N)(q1)(q2)(k) / r where k = 9.0x10^9
- Chemistry In an analysis of the content of carbohydrate present in a glycoprotein, the following results were found: 12.6, 11.9, 13.0, 12.7 and 12.5 g of carbohydrate per 100 g of protein. Taking into account that σ is unknown, the confidence interval for the average value at a 90% confidence level of the carbohydrate content is: Select one:to. 12.5 ± 0.4b. 12.5 ± 0.2c. 12.5 ± 0.5d. 12.5 ± 0.3Potassium dichromate has several industrial applications. To determine the purity of the salt that will be used in different industrial processes, a sample mass equal to 2.660 g was dissolved and quantitatively transferred to a 500.00 mL flask. An aliquot of 25.00 mL of this solution was treated with excess KI and the released iodine was titrated with 0.1000 mol L-1 sodium thiosulfate, spending 27.00 mL. Calculate the purity of the analyzed salt. Data:K = 39.10 O = 16.00 Cr = 52.00 I = 126.9 S = 32.07(a) A commercial 737 jet transporting 143 passengers and 5 crew members from Kansas City (MCI) to Baltimore (BWI) burned 11,800 lb (about 1700 gallons) of Jet A fuel en route. Jet A fuel is kerosine based, consisting primarily of CnH2n+2 hydrocarbons, with n =6 to 16, so the carbon:hydrogen ratio is close to 1:2. During this flight, how much CO2 was released into the atmosphere? Assume the combustion of the fuel was complete, so all the fuel was burned to form CO2 and H2O. Give both the mass of CO2 produced (in kg and in lbs) and the volume it would occupy at 298 K, 1 atm.(b) How much CO2 would be released into the atmosphere if those passengers and crew made the trip instead, in pairs, in hybrid cars at 40 miles per gallon. Assume the density of the gasoline is 0.75 kg L-1 and that carbon and hydrogen dominate the composition in a ratio of 1:2. The road trip is 1082 miles.
- Consider the following data concerning the equation: H2O2 + 3I– + 2H+ → I3– + 2H2O [H2O2] [I–] [H+] rate I 0.100 M 5.00 × 10–4 M 1.00 × 10–2 M 0.137 M/sec II. 0.100 M 1.00 × 10–3 M 1.00 × 10–2 M 0.268 M/sec III. 0.200 M 1.00 × 10–3 M 1.00 × 10–2 M 0.542 M/sec IV. 0.400 M 1.00 × 10–3 M 2.00 × 10–2 M 1.084 M/sec The rate law for this reaction isConsider the following data concerning the equation: H2O2 + 3I– + 2H+ → I3– + 2H2O [H2O2] [I–] [H+] rate I 0.100 M 5.00 × 10–4 M 1.00 × 10–2 M 0.137 M/sec II. 0.100 M 1.00 × 10–3 M 1.00 × 10–2 M 0.268 M/sec III. 0.200 M 1.00 × 10–3 M 1.00 × 10–2 M 0.542 M/sec IV. 0.400 M 1.00 × 10–3 M 2.00 × 10–2 M 1.084 M/sec The rate law for this reaction is a. rate = k[H2O2][I–][H+] b. rate = k[H2O2][H+] c. rate = k[H2O2]2[I–]2[H+]2 d. None of these e. rate = k[H2O2][I–] f. rate = k[I–][H+]Consider the following data concerning the equation: H2O2 + 3I– + 2H+ → I3– + 2H2O [H2O2] [I–] [H+] rate I 0.100 M 5.00 × 10–4 M 1.00 × 10–2 M 0.137 M/sec II. 0.100 M 1.00 × 10–3 M 1.00 × 10–2 M 0.268 M/sec III. 0.200 M 1.00 × 10–3 M 1.00 × 10–2 M 0.542 M/sec IV. 0.400 M 1.00 × 10–3 M 2.00 × 10–2 M 1.084 M/sec The rate law for this reaction is a. rate = k[H2O2]2[I–]2[H+]2b. rate = k[I–]c. None of thesed. rate = k[I–][H+]e. rate = k[H2O2][I–][H+]f. rate = k[H2O2][H+]

