Q: A 65-kg person is accidentally exposed for 240 s to a15-mCi source of beta radiation coming from a s...
A: (a) Calculate the activity of the radiation source in disintegrations per second: Calculate the act...
Q: Draw a stepwise mechanism for each reaction.
A:
Q: Synthesize m-chlorophenol from benzene.
A: Given:
Q: Calculate the pH of a solution containing 1 x 10-7 M of hydronium ions [H3O+]. pH = 5.0 pH = 6.0 pH...
A: Since the pH of any solution is given by pH = -log[H3O+] where [H3O+] = concentration of H3O+ ions
Q: Consider the following equilibrium, for which Kp = 7.53×10−2 at 480 ∘C: 2Cl2(g)+2H2O(g)⇌4HCl(g)+O2(g...
A:
Q: What product is formed when each compound undergoes thermal electrocyclic ring opening or ring closu...
A: These reactions are pericylic reactions. When molecule possess (4n+2)π electrons then its thermal re...
Q: justify the ingridients of benadryl cream
A: The Benadryl cream is actually an itch stopping cream. It is used to relieve itching and pain tempor...
Q: Please determine the number of IR-active C-O stretching vibrations for the following molecules (plea...
A: SOLUTION: Step 1: Cis-Fe(CO)4Cl2 has C2v symmetry. It has 4 IR active C-O stretching vibrations and ...
Q: Draw the major product for each reaction showing the stereochemical outcome (syn or anti). Identify ...
A: (c)
Q: Rank the following ammonium ions in order of increasing pKa.
A: pKa is defined as the negative logarithm of Ka. More is the acidity of the compound, more is it’s K...
Q: What product (including stereochemistry) is formed in each of the following intramolecular reactions...
A: NaOEt acts as a base and abstracts the most acidic H. Then it undergoes intramolecular reaction to f...
Q: (a) What is the empirical formula of cellulose? (b) Whatis the monomer that forms the basis of the c...
A: The cellulose is an organic polymer which is formed of many repeated monomer units. The formation of...
Q: Please answer all
A: Since you have asked multiple question, we will solve the first question for you. If you want any ...
Q: Draw a qualatative potential energy diagram for rotation about the C3-C4 bond in 2-methylpentane. Sh...
A: Conformers of 2-methylpentane are,
Q: A medical solution had a concentration of 30 mg/ml. How many ml would need to be delivered to dispen...
A: The concentration of medicine solution is given in mg/ml Hence it means the concentration is given b...
Q: Show how the following compounds can be synthesized from benzene: a. N,N,N-trimethylanilinium iodide...
A: a. The synthesis of the given compound from benzene is given below. The given compound is, N,N,N -tr...
Q: Draw each stereogenic center using a Fischer projection formula.
A:
Q: The neutralization of a strong acid and strong base has an enthalpy change, ∆H°, = -55.9 kJ/mol. The...
A: The relationship between the ΔG0 and equilibrium constant is given by RTln(K) = -ΔG0 where R = gas...
Q: A physician orders 700,000 units of penicillin. The goal of penicillin is labeled 250,000 units/cc. ...
A:
Q: Synthesize A from diethyl malonate and any needed organic compounds and inorganic reagents.
A: Di-ethyl-malonate or DEM is an ester of malonic acid. The product A can be formed by the following ...
Q: for the following, please identify the titrant and analyte!!! Prepare a titration for a 5mL bora...
A: The of any titration is to find out the concentration of the unknown solution using known concentrat...
Q: The amino acid glycine (H2N—CH2—COOH) can participatein the following equilibria in water:H2N—CH2—CO...
A: EXPLANATION: A) part Glycine behaves in equilibrium with H2O. H2N—CH2—COOH + H2O ⇌ H2N—CH2—COO- + ...
Q: Consider the following reaction at equilibrium:2CO2 (g) --><-- 2CO (g) + O2 (g) ∆H° = -514 kJL...
A: According to LeChatleir's principle, "When a system at equilibrium is subjected to a change in eithe...
Q: Rocket engines built by NASA use fuels like liquid hydrogen and liquid oxygen. Hobbyist rocketeers u...
A: SOLUTION: Step 1: ΔG0 can be calculated from the standard free energy changes of formation from the...
Q: For the reaction 2HI(g)⇌H2(g)+I2(g) the equilibrium constant Kc=1.8×10−2 at 700 K. A. What is the ...
A:
Q: Derive general expressions for (a) the partition coefficient, KoD,in an organic (1) aqueous–(2) LLE ...
A: SOLUTION: Step 1: The partition coefficient is defined as the particular ratio of the concentration...
Q: Mr. D.B. Has poison ivy. The doctor orders daily decreasing doses of prednisone for treatment. This ...
A: Since the mass of each tablet is = 10 mg Hence the number of tablets need to be taken = mass of med...
Q: Draw a stepwise mechanism for the following reaction.
A: This reaction is the hydrolysis of Amide in basic medium. Here, one tetrahedral intermediate is form...
Q: Consider the elements Li, K, Cl, C, Ne, and Ar. From thislist, select the element that (a) is most e...
A: (a) Electronegativity of the elements increases along the period from left to right and decrease dow...
Q: Discuss how catalysts can make processes more energyefficient.
A: A catalyst is substance that can be added to a chemical reaction without getting consumed in a chemi...
Q: Indicate whether the following balanced equations involveoxidation–reduction. If they do, identify t...
A: (a) 2 AgNO3(aq) + CoCl2(aq)---> 2 AgCl(s) +Co(NO3)2(aq) In this reaction the oxidation states of ...
Q: Consider the following reaction at equilibrium:2SO2 (g) + O2 (g) --><-- 2SO3 (g) ΔΔ H° = -99 k...
A: The reaction given is 2SO2 (g) + O2 (g) --><-- 2SO3 (g) Δ H° = -99 ...
Q: Farnesyl diphosphate is cyclized to sesquiterpene A, which is then converted to the bicyclic product...
A: The steps involved in the mechanism of the given reaction are as follows: The first step is the remo...
Q: A reaction is made up in the following way: 19 mL of 4.8 M acetone + 14 mL of 1.4 M HCI + 18 mL of 0...
A: Given: 19 mL of 4.8 M acetone 14 mL of 1.4 M HCl 18 mL of 0.0057 M I2 17 mL of water
Q: Draw a structure for each of the following: a. m-chloromethylbenzene c. o-nitroaniline ...
A: a. The structure of m-chloromethylbenzene is drawn below.
Q: An a,ß-unsaturated carbonyl compound can be prepared by a reaction known as a selenenylation-oxidati...
A: LDA is used to abstract the ‘H’ from the less hindered side to form the carbanion. Since both the si...
Q: A nuclear reaction in which a larger nuclei is split apart into smaller nuclei is called a _____ rea...
A: A) combustion : The combustion reaction is a process in which the reactant is burned or reacted with...
Q: Spontaneity criteria of simple and nonsimple system in terms of Gibbs and Helmholtz free energies
A: For a spontaneous process, change in Gibbs free energy is negative because the energy of the system ...
Q: A reaction is made up in the following way: 16 mL of 4.3 M acetone + 16 mL of 1.1 M HCI + 16 mL of 0...
A: Since concentration = moles / volume in L Hence moles of I2 taken = concentration of I2 X volume of...
Q: A patient was injected with 5.00 mL of a 3.0gm/10mL codeine solution. How many grams of codeine were...
A: Since the medicine is marked as 3 g/10 mL i.e 3 grams of codeine is present in every 10 mL of soluti...
Q: What is the rate for the second order reaction A → Products when [A] = 0.451 M? (k = 0.761 M⁻¹s⁻¹)? ...
A: For any reaction, the rate of reaction depends on the concentration of reactants only And since the...
Q: Choose from the options A-E and explain briefly by illustrations the chemistry behind each answer. P...
A: Whenever an alkyl halide is treated with a base, there is a possibility of the formation of more tha...
Q: Homework
A: Given structure;
Q: The Keq for the equilibrium below is 0.112 at 700.0 °C.SO2 (g) + 1/2O2 (g) --><-- SO3 (g)What ...
A: The reaction with given equilibrium constant value is SO2 (g) + 1/2O2 (g) --><-- SO3 (g) Henc...
Q: Draw all stereoisomers formed in each reaction.
A: At the both end of the double bond, the anti-addition of halogen is occurred.
Q: At 1000.0 K, the equilibrium constant for the reaction2NO (g) + Br2 (g) --><-- 2NOBr (g)is Kp ...
A: The reaction with given equilibrium constant value is 2NO (g) + Br2 (g) --><-- 2NOBr (g) Henc...
Q: Differentiate between carnot heat engine and refrigerator.
A: Given: Difference between heat engine and refrigerator.
Q: mass of evaporating dish in grams is 43.7 grams. what is the mass in: kg lbs tons dg
A: Since 1 Kg = 1000 g Hence mass in Kg = mass in g / 1000 => mass in Kg = 43.7 / 1000 = 4.37 X 10-...
Q: What two carbonyl compounds are needed to synthesize each of the following compounds, using a Robins...
A: (a)
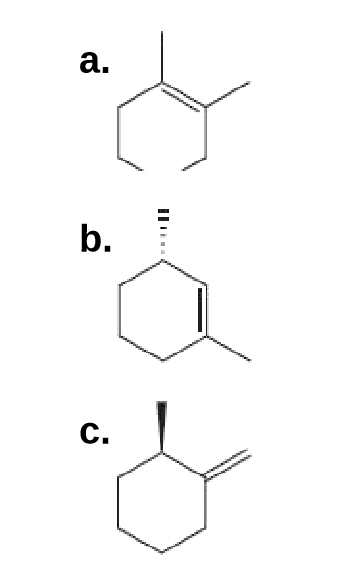
Draw the structure (including stereochemistry) of an alkyl chloride that forms each
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images


