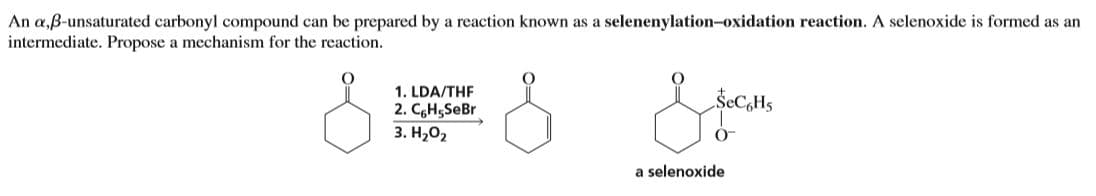
An a,ß-unsaturated carbonyl compound can be prepared by a reaction known as a selenenylation-oxidation reaction. A selenoxide is formed as an intermediate. Propose a mechanism for the reaction. 1. LDA/THF 2. CgH5SeBr 3. H,02 ŠECH5 a selenoxide
Q: Draw the products formed when benzoyl chloride (C,H;COCI) is treated with each nucleophile: (a) H20,…
A: Since you are posted with multiple sub-parts. As per the rule, I am answering the first three…
Q: 6. What is the best choice of reagent(s) to carry out the ? transformation shown here? Ph HO. Ph A)…
A: We have given three organic reaction we have to find major product obtained from these reactions.
Q: 2-Pentylcinnamaldehyde, commonly called flosal, is a perfume ingredient with a jasmine-like odor.…
A:
Q: Which compound is the major product of the reaction sequence shown? (1) NANH,NH3, -35°C (2) CH3 CH2-…
A: 1) first reaction is acid base reaction. 2) second reaction is SN2 reaction 3) third reaction is…
Q: H,CH2C H,CH2C CH3 H,CH,C H,C H JAN JEN JIN JUN 22. produces an a-diketone upon reaction with cold,…
A:
Q: 8] Propose a mechanism to account for the following reaction H2SO4
A: Ethers are species that has a oxygen atom attached to two alkyl groups. Ethers are polar in nature…
Q: Acid-catalyzed dehydration of B-hydroxy carbonyl compounds occurs by the mechanism discussed in…
A: The mechanism for the given reaction is given below
Q: Show the product expected when the following unsaturated 8-ketoester is treated with each reagent.…
A: The product of the given reaction can be shown as
Q: The Baylis–Hillman reaction is a DABCO (1,4-diazabicyclo[2.2.2]octane) catalyzed reaction of an…
A:
Q: Please provide a way to synthesize the target compound using the reagents given below once Target…
A: The conversion reaction is given in the following step.
Q: 1. Propose a mechanism to rationalize the following transformations. TSOH a) HO H2SO4 THE b)
A: Mechanism of organic reaction.
Q: Decytospolides A and Bare fungal natural products that are toxic to some cancer cells, In a…
A: Concept : 1) NaH is used as base for deprotonation. 2) Grignard reagent is used as nucleophile here…
Q: Propose a plausible mechanism for the following transformation. NaH CI HO 13.38e This substitution…
A: Organic reaction mechanisms
Q: Identify A, B, and C, intermediates in the synthesis of the five-membered ring called an…
A: The A,B and C cpmpounds are as given below:
Q: The Baylis–Hillman reaction is a DABCO (1,4-diazabicyclo[2.2.2]octane) catalyzed reaction of an…
A:
Q: Select the reagent that you would use to convert an alkene to an epoxide. mCPBA 03, then DMS…
A: The epoxide is a cyclic ether with a ring structure consisting of three atoms that somehow form an…
Q: Draw the products formed when benzoic anhydride [(C6H5CO)2O] is treated with each nucleophile: (a)…
A: a.
Q: Chemistry Draw the product formed when phenylacetaldehyde (C6H5CH2CHO) is treated with reach…
A: Detail mechanistic pathway is given below to find out the product in every case
Q: 6. Propose a mechanism for the following transformation. H2SO4 H20 ОН
A:
Q: is a valuable synthetic intermediate because each of its Epichlorohydrin three carbons contains a…
A: Allyl chloride can be prepared from propene by the free radical mechanism as shown in the next step.
Q: 1.3. Propose a synthetic method from benzene the following molecule. NO Give the major product(s) of…
A: Answer of the question given below
Q: Mustard gas, Cl¬CH2CH2¬S¬CH2CH2¬Cl, was used as a poisonous chemical agentin World War I. Mustard…
A: Sulfur in mustard gas is more nucleophilic than other corresponding oxygen compounds. This is due to…
Q: 1. Cl2, H20 2. NaOH Another mechanism for the formation of epoxides is through the formationofa…
A: There are number of functional group associated with organic compounds which impart specific…
Q: 1. Predict the major product(s) from the treatment of acetone with the following compounds: (a) [I…
A: Solution Acetone, a colorless liquid conjointly called dissolving agent, could be a solvent utilized…
Q: Show how m-toluidine can be converted tom-toluidineCH3 NH2the following compounds, using any…
A: a.
Q: The synthetic product, after being subjected to Hofmann elimination conditions, may result in the…
A: The formation of Olefin’s from Amine can be effectively achieved by Hoffmann Elimination reactions.…
Q: Devise a stepwise mechanism for the following reaction. The reaction does not take place by direct…
A:
Q: Acid-catalyzed dehydration of β-hydroxy carbonyl compounds occurs by the mechanism. With this in…
A: Given reaction is: Step wise mechanism of the given reaction is :
Q: When a cyclic ketone reacts with diazomethane, the next larger cyclic ketone is formed. This is…
A: To draw a mechanism for the below ring-expansion reaction.
Q: Nicotine can be made when the attached ammonium salt is treated withNa2CO3. Draw a stepwise…
A: The first step involves slow dissociation of sodium carbonate as a base. The first step involves the…
Q: What steps are needed to convert benzene into p-isobutylacetophenone, a synthetic intermediate used…
A: First we need to react the benzene with the below mentioned compound in presence of AlCl3 Than the…
Q: Draw the mechanism explaining the following transformation of the reactant into the observed major…
A: This is an example of reductive amination reaction. Reaction of primary amine with formaldehyde in…
Q: What is the major product of the reaction of 1 mol of propyne with each of the following reagents?…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: OH CH₂CNHCH3 CH3 C d OH CHCHNHCH₂ CH3 Ephedrine can be synthesized via reductive amination plus…
A:
Q: Quinapril (trade name Accupril) is used to treat high blood pressure andcongestive heart failure.…
A: The reaction mechanism along with the products is given below.
Q: A Mannich reaction puts a R R-NCH2 - group on the α-carbon of a carbon acid. Propose a mechanism…
A: For a Mannich reaction to take place the presence of a hydrogen atom at the carbon atom adjacent to…
Q: Draw a stepwise mechanism for the following reaction. This reactioncombines two processes: the…
A: A mechanism for the given reaction can be shown as,
Q: 34. Propose a mechanism for the following reaction: H + H20 CH, CH2CH2CH2 он
A: In this question, we will draw the mechanism steps for the formation of cyclic ether from the given…
Q: 40. Which sacleophile would afford the greatest amount of the substitution product shown below? Br…
A:
Q: Below is a reaction scheme for the transformation of 2-phenylethan-1-ol to the corresponding…
A:
Q: The reaction sequence shown in Scheme 5 demonstrates the synthesis of a substituted benzene…
A:
Q: Which compound is the major product of the reaction sequence shown? (1) SOC2 (A) (B) HO. (2)…
A: Given is two step reaction.
Q: An amide with an NH2 group can be dehydrated to a nitrile with thionyl chloride (SOCl2). Propose a…
A: Thionyl chloride when reacts with 1° amide, a nitrile can be produced. A four step mechanism can be…
Q: Match each set of reagents reacting with to a major product below. Answers may be Br repeated. The…
A: NaI in acetone : f Explanation : NaI give I- ion and this ion give SN2 Reaction with faster rate…
Q: Fluticasone, the chapter-opening molecule, can be prepared by the following reaction. Draw a…
A:
Q: Specify the reagent you would use in each step of the following synthesis: step 1 step 2 CI Reagents…
A:
Q: 3.4 Propose a mechanism for the following reaction: CI NaNH, Liq. NH3 NH₂
A: In the given reaction the chlorobenzene reaction sodium amide to form aniline. The reaction is…
Q: What reducing agent can selectively reduce aldehydes, but not esters? KMnO4 OsO4 LiAlH4 NaBH4
A: It is asked which reducing agent can selectively reduce aldehydes, but not esters. The options given…
Q: Lexan can also be prepared by the acid-catalyzed reaction of diphenyl carbonate with bisphenol A.…
A:
Step by step
Solved in 2 steps with 1 images

- The Baylis–Hillman reaction is a DABCO (1,4-diazabicyclo[2.2.2]octane) catalyzed reaction of an a,b-unsaturated carbonyl compound with an aldehyde toform an allylic alcohol. Propose a mechanism for the reaction. (Hint: DABCO serves as both a nucleophile and as a base in the reaction.)The Baylis–Hillman reaction is a DABCO (1,4-diazabicyclo[2.2.2]octane) catalyzed reaction of an a,b-unsaturated carbonyl compound with an aldehyde to form an allylic alcohol. Propose a mechanism for the reaction. (Hint: DABCO serves as both a nucleophile and as a base in the reaction.)The poison gelsemoxonine can be isolated from the leaves of a plant native to southeastern Asia (Gelsemium elegans). A key step in a synthesis of this natural product involves a thermally initiated (70°C) sigmatropic rearrangement of the compound shown below (R=protecting group) (J. Am. Chem. Soc. 2011, 133, 17634–17637). Draw the curved arrows for the mechanism of the formation of gelsemoxonine. Include lone pairs in your answer.
- Chemistry Draw the product formed when phenylacetaldehyde (C6H5CH2CHO) is treated with reach reagent: f. (CH3)2CHNH2, mild acid g. (CH3CH2)2NH, mild acid h. CH3CH2OH (excess), H+ i. piperidine, mild acid j. HOCH2CH2OH, H+Which of the following statements about terminal alkynes is FALSE?I I. A geminal dihalide is produced by the hydrohalogenation reaction.II. The proton in the terminal carbon is acidic but just slightly.III.They create an aldehyde when they react with H2O, H2SO4, and HgSO4.IV. A silver acetylide is formed after treatment with alcoholic AgNO3.These reagents can produce ketones with alkynes A. BH3, THF, H2O2 B. KMnO4 C. O3 D. H2SO4, H2O, HgSO4 choices:A,DB,CA,B,CA,B,C,D
- gas, Cl¬CH2CH2¬S¬CH2CH2¬Cl, was used as a poisonous chemical agentin World War I. Mustard gas is much more toxic than a typical primary alkyl chloride. Itstoxicity stems from its ability to alkylate amino groups on important metabolic enzymes,rendering the enzymes inactive.(a) Propose a mechanism to explain why mustard gas is an exceptionally potent alkylatingagent.(b) Bleach (sodium hypochlorite, NaOCl, a strong oxidizing agent) neutralizes and inactivates mustard gas. Bleach is also effective on organic stains because it oxidizes coloredcompounds to colorless compounds. Propose products that might be formed by thereaction of mustard gas with bleach.This is Wittig Rxn: Whatever mechanism you choose to draw is fine since you can leave the base as B: Draw the arrow pushing mechanism using the compounds below – Constant: 4-nitrobenzyl benzaldehydebase 1: triethylaminebase 2: NaOHbase 3: K2CO3ylide: Acetonyltriphenylphosphonium chloridePropose a mechanism for the reaction of benzyl acetate with methylamine. Label theattacking nucleophile and the leaving group, and draw the transition state in which theleaving group leaves.
- 8. Propose a mechanism for this transformationWhat is the major product of the reaction of 1 mol of propyne with each of the following reagents? a. HBr (1 mol) e. aqueous H2SO4, HgSO4 h. H2/Lindlar catalyst b. HBr (2 mol) f. R2BH in THF followed by i. sodium amide c. Br2 (1 mol)/CH2Cl2 H2O2/HO-/H2O j. the product of part i followed by d. Br2 (2 mol)/CH2Cl2 g. excess H2, Pd/C 1-chloropropaneBoron tribromide (BBr3) cleaves ethers to give alkyl halides and alcohols.R¬O¬R′ + BBr3 ¡ R¬O¬BBr2 + R′BrR¬O¬BBr2 + 3 H2O ¡ ROH + B(OH)3 + 2 HBrThe reaction is thought to involve attack by a bromide ion on the Lewis acid–base adductof the ether with BBr3 (a strong Lewis acid). Propose a mechanism for the reaction ofbutyl methyl ether with BBr3 to give (after hydrolysis) butan-1-ol and bromomethane.



