Q: A sample consisting of Na2CO3, NaOH and inert matter weighs 1.179 grams. It is titrated with 0.3 N H...
A:
Q: (a) Draw a skeletal structure of the anabolic steroid 4-androstene-3,17-dione, also called “andro,” ...
A: (a) Steroids are composed of four rings, three six-membered rings and one five membered ring. The t...
Q: Which of the following solutions has the higher boiling point, 0.250 M glucose or 0.150 M NaCl? Expl...
A: The elevation in boiling point of any solution is given by elevation in boiling point = i X Kb X m ...
Q: The normal boiling point of C2Cl3F3 is 47.6 °C and its molar enthalpy of vaporization is 27.49 kJ/mo...
A: The change in entropy in the system in J/K when 19.4 grams of C2Cl3F3 vaporizes to a gas at the norm...
Q: What is specific gravity ? What is the difference between specific gravity and density? What is the ...
A: Certain objects float on water and certain objects sink in water. There are many factors which will ...
Q: The sealed containers of fizzy drinks contain dissolved CO2. This dissolved CO2 is in equilibrium wi...
A: Henry's law: The amount of gas dissolved in a liquid is directly proportional to the partial pressur...
Q: How was the oxidation state for the acetate determined? Does one just need to simply remember this f...
A: Acetate ion is formed from acetic acid i.e CH3COOH when it loses proton
Q: A mixture of hydrogen and nitrogen gases, in a 7.57 L flask at 34 °C, contains 0.388 grams of hydrog...
A: According to ideal gas law, PV = nRT where P = pressure V = volume of vessel = 7.57 L n = moles of g...
Q: Which bond in each pair has the higher bond dissociation energy?
A: As the atomic size increases on moving down in a group of periodic table. Hence the size of S > s...
Q: A buffer is made up of 15.0 mL of 0.1 M HC2H3O2 and 15.0 mL of 0.1 M N a C2H3O2. The pH of the buffe...
A: The term pKa used for acids and it reflects any acid's strength how much strong the acid is. The sma...
Q: Write the formula for each of the following compounds,being sure to use brackets to indicate the coo...
A: The Formula for the following coordination compounds are: (a) hexaamminechromium(III) nitrate [Cr(NH...
Q: The normal boiling point of methanol is 64.7∘C and the molar enthalpy of vaporization if 71.8kJ/mol....
A: We know that , at normal boiling point, Gibb's free energy change is given by, ...
Q: Determine the number of protons and electrons in each ion I¹- A.53 protons, 52 electrons B.53 proto...
A: Given ion is (iodide ion ) Anion is formed by gain of electrons Atomic number of iodine is 53 (num...
Q: The solubility of two slightly soluble salts of M2+ , MAand MZ2, is the same, 4 x 10-4 mol/L. (a) Wh...
A: Solubility product (Ksp):- A solubility product is the equilibrium constant for a chemical reaction...
Q: The maximum number of o bonds that the atom can form is If an atom has sp'd hybridization in a molec...
A: The phenomenon describing atomic orbitals' mixing which generates orbitals identical in energy is re...
Q: For a given reaction, ΔH=69.3kJ/mol, and the reaction is spontaneous at temperatures above the cross...
A: Given ΔH=69.3kJ/mol T= 464K.
Q: What volume of a solution of ethylene glycol, C2H6O2, that is 57.5% ethylene glycol by mass contains...
A: Molarity is defined as moles of solute to the volume of solution. Molarity calculation formula:
Q: For a given reaction, ΔS = +112 J/mol ⋅ K, and the reaction is spontaneous at temperatures above the...
A:
Q: Q5.) A certain reaction has ΔH∘ = + 35.8 kJ and ΔS∘ = 85.5 J/K. 1. exothermic or endothermic? 2. Doe...
A: “Since you have posted a question with multiple sub-parts, we will solve first three subparts for yo...
Q: Under what conditions do zwitterions exist?
A: A zwitterion is adipolar on that has a net charge equal to zero. An amino acid has both acidic and ...
Q: How many molecules are there in a 5.13-mg sample of chlorine trifluoride
A: The formula of chlorine trifluoride is ClF3 Hence the molar mass of ClF3 = Atomic mass of Cl + 3 X ...
Q: Ethylene glycol (C₂H₆O₂) is used as an additive to the water in your automobile to lower its freezin...
A: Since the depression in freezing point of any solution is given by depression in freezing point = i...
Q: Glutamic acid is synthesized by the following reaction sequence. Draw a stepwise mechanism for Steps...
A: The first step in the synthesis of glutamic acid involves the abstraction of acidic proton present b...
Q: The normal boiling point of ethanol (C2H5OH) is 78.3 °C and its molar enthalpy of vaporization is 38...
A: Given: The normal boiling point of ethanol (C2H5OH) = 78.3 °C Molar enthalpy of vaporizati...
Q: For the reaction 2A+B giving 3C +D.what is the magnitude of rate of change for conc. Of D when conc....
A: Reactants' quantity gets decreased during reaction but the products' amount get increased during rea...
Q: (a) Draw two different enol tautomers of 2-methylcyclohexanone. (b) Draw two constitutional isomers ...
A: The carbon which is present adjacent to the carbonyl carbon is α-carbon, and the hydrogen covalently...
Q: What is the energy of a photon corresponding to radio waves of frequency 1.306 106 /s? J
A: The relationship between energy and frequency of photon is given by E = hf where E = energy f = fr...
Q: What volume of butane (CH10) is required to react with 58.6 liters of oxygen gas according to the fo...
A: The reaction given is C4H10 (g) + O2 (g)--------> CO2 (g)+ H2O(g) Since we have 4 C in LHS. Hence...
Q: SO2Cl2 (g) ➞ SO2 (g) + Cl2 (g) What will be the concentration of SO2Cl2 left in the reaction mi...
A: Since we know that, for different order of reactions, the relationship between the concentration of ...
Q: 1. State the effects of substituents on a benzene derivative towards further aromatic substitution. ...
A: SOLUTION: Step 1: When substituted benzene compounds undergo electrophilic substitution reactions, ...
Q: Explain why there is no distinction between low and high-spin arrangements for an octahedral d8 meta...
A:
Q: Consider the tetrasaccharide stachyose drawn below. Stachyose is found in white jasmine, soybeans, a...
A: a) The glycosidic linkages are represented by the acetals containing alkoxy groups attached to anome...
Q: A photon of blue light has a frequency of 7.5 × 10¹⁴ Hz. Calculate the wavelength of this photon (c ...
A: The relationship between speed of light (c) , wavelength (λ) ) and frequency (ν) given mathematical...
Q: Which alkene in each pair has the larger heat of hydrogenation?
A: The more is the substitution on the double bond in an alkene, less is the heat of hydrogenation of t...
Q: Provide the complete mechanism for the following reaction.
A: Given reaction,
Q: A metal lump has a mass of 30.00g. When it is placed in a graduated cylinder containing 20.0 mL of w...
A: The term density exhibits how much mass any material contains at specific volume. Density can be abb...
Q: Rank the protons in the labeled CH2 groups in order of increasing acidity, and explain why you chose...
A: Removal of a proton results into a conjugate base. The stronger the conjugate base, the strong the a...
Q: Given the following compounds and boiling points: Determine the IMFs Order the compounds from wea...
A: Boiling point of any substance is proportional to 2 things in the below mentioned order. 1) The stre...
Q: Which of the following metals would be expected to have the smallest atomic radius? A) Cesium (C...
A: In periodic table, on moving left-right the element's electronegativity as well as atomic number get...
Q: Compund ( A) C6H12 is treated with chlorine in the presence of carbon tetrachloride to given (B)C6H...
A:
Q: Based on the following atomic mass values-'H, 1.00782 amu; ²H, 2.01410 amu; H, 3.01605 amu; 'He, 3.0...
A: Part a: Calculate the mass defect for the given nuclear reaction. The mass defect will be equal to (...
Q: What volume of 0.170 M KMnO4 would be required to titrate 0.38 g of K2[Cu(C2O4)2]⋅2H2O?
A: Given, molar concentration of KMnO4 = 0.170 M mass of K2[Cu(C2O4)2]⋅2H2O = 0.38 g Now, ...
Q: Draw the structure of glycine as a zwitterion
A: The structure of normal glycine is as shown below.
Q: Calculate ∆G° for a reaction for which ∆H° = 24.6 kJ and ∆S° = 132 J/K at 298 K. Is the reaction spo...
A: Given, ∆Ho = 24.6 kJ ∆So = 132 J/K T = 298 K
Q: (a) Explain why there is no distinction between low and high-spin arrangements for an octahedral d8 ...
A:
Q: Answer all questions 1.What are the various ways by which alkenes may be synthesized?2.i. Give two e...
A: Since we are entitled to answer up to 1 question, we’ll answer the first question as you have not me...
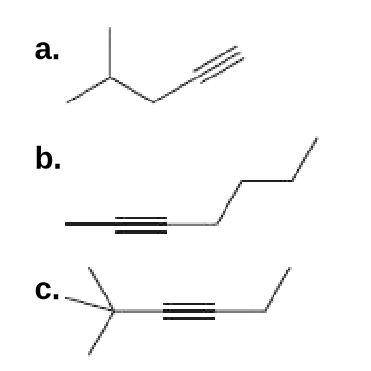
Q: A chiral alkyne A with molecular formula C6H10 is reduced with H2 and Lindlar catalyst to B having t...
A: Chiral alkyne with molecular formula C6H10 The given alkyne with chiral centre could be;
Q: The standard free energy of formation of solid glycineis -369 kJ/mol, whereas that of solid glycylgl...
A: Condensation reaction involves removal of water molecule. Therefore, The value of ∆G0 for the react...
Q: 4. Why does a carbon to oxygen double bond absorption band have a greater intensity than a carbon to...
A: 1. The intensity of the absorption band depends on the change in the dipole moment of the bond which...
What acetylide anion and
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images


