Q: 203.77 miles/sec is converted to cm/nsec
A:
Q: Below, write the balanced chemical equation for the reaction you observed (including state). If a p...
A:
Q: The diagram below represents the layout of a typical single-beam UV-Vis, explain the function of eac...
A:
Q: Туре of Crystal Covalent Metallic Ionic Molecular Substance Network Dispersion Forces Dipole- Dipole...
A: In this question we have to determine the type of crystal solid formed by the given compounds.
Q: What is the function of the flame in AAS? Ionozation of sample boiling of sample A...
A:
Q: Solve the standard entalphies of the following and determine wether the reaction is endothermic or e...
A: 2NH3(g) + 3N2O(g) -----> 4N2(g) + 3H2O(l) CaCO3(s) -------> CaO(s) + CO2(g) C(graphite) + O2(...
Q: II. Analysis. Given below is a schematic diagram for a simple analysis of a novel tetrasaccharide is...
A: D- fructose. B- glucose . C - galactose . As A gives Benedict's test: (+) Seliwanoff's test: (-) Bi...
Q: Question 5 In a typical AAS what is the ouput of the istrument given out as data? % composition Volt...
A: Atomic absorption spectroscopy can be defined as the technique that is used for measuring the concen...
Q: Convert the following measurement. 5.8 x 10 x10 3 3 cm ?
A:
Q: O Heptene
A: In this question we have to choose correct option of compound is represented by above spectra with e...
Q: What is the the concentration of copper and cobalt in mM?
A: The data given is,
Q: The diagram below represents the layout of a typical single-beam UV-Vis, explain the function of eac...
A:
Q: Ammonia, NH3, is produced at high temperatures and pressures in the presence of a catalyst as shown ...
A:
Q: Calculate the molarity of a phosphoric acid (H3PO4) solution that is 84% by mass phosphoric acid and...
A:
Q: A solution of table sugar in water contains 5.25% by mass sucrose. Calculate: a)the mass of sucrose ...
A: To find out mass of sucrose first check mass in 5.25% solution. Use that relation to find mass of su...
Q: A 50.0 mL juice extract is colorimetrically assayed using Nelson’s test. One milliliter (1.00 mL) o...
A: Given: 50 mL of original juice is used. 1 mL of the original solution is used to give absorbance of ...
Q: 1. Give the structures of the products obtained when the following are heated: Ph „Me -OMe b. a. Ph ...
A: The feasibility of the given reaction depends on the nature of the reactants such as diene and dinop...
Q: d)the mole fraction of sucrose and water in 5.25% by mass sucrose
A:
Q: statement
A: When a pure substance changes phase from subcooled liquid to superheated steam, this CAN be reversed...
Q: What is the order of the reaction with respect to P? What is the order of the reaction with respect...
A: The sum of the exponent term in the rate law for a chemical reaction is known as the order of the re...
Q: How to calculate grams of aluminum chloride using 2AI + 6HCI -> 2AICI3 + 3 H2 formed with 135 g of A...
A:
Q: What is the product of the following reaction?
A:
Q: ~60°C trial ~50°C trial ~40°C trial ~30°C trial ~20°C trial Vol. of HCl used (mL) 19.87 12.93 12.37 ...
A: The solution of the question is given below:
Q: O. An acrobat swings from a trapeze with a frequency of 0.5 Hz. What is the length of the trapeze ca...
A: Given : Frequency = 0.5Hz
Q: What happens during a dehydration reaction? Water reacts with the molecule and breaks it down into s...
A: The electronic configuration of carbon is 1s22s22p2
Q: Please prepare the following compound from methane through multi-step synthesis.
A: Here we have to convert methane to cis- cyclohexane-1,2-diol by multistep synthesis reaction.
Q: The following data represented the concentration of ofloxacin present in chicken meat samples: 4.17,...
A: Percentage relative standard deviation is given by formula % relative SD =(standard deviation / me...
Q: The iodination of salicylamide was carried out analogous to Experiment 12 in your “Green Organic Che...
A: Increase in chemical shifts suggest that the proton is deshielded. Hydrogen bond interaction is one ...
Q: A female skater is spinning at 1 rev/s with her arms outstretched so is 3.6 kg/m2. She then pulls he...
A: According to the question, a female skater is spinning at 1 rev/s with her arms outstretched so that...
Q: A solution at 25 °C has [OH-] = 6.7 * 10 -3 . What is the pH of the solution?
A: [OH-] = 6.7 × 10-3 Temperature = 25°C pH of the solution = ?
Q: how much energy must be absorbed or released as heat to change 87.0 g of liquid propanone at 25.00°C...
A: The solution is as follows:
Q: A 5.0 L balloon has a pressure of 1 atm at sea level, where the temperature is 28 ͦC. The balloon w...
A: Initial volume = 5.0 L Initial pressure = 1 atm Initial temperature = 28 °C New temperature = 11 ...
Q: Under certain conditions the rate of this reaction is zero order in hydrogen iodide with a rate cons...
A: The rate of the zero-order reaction does not depend on the concentration of the reactant. For the ze...
Q: Calculate the molar mass (g/mol) of a strong electrolyte (i = 2.16) where 40.87 g of the compound is...
A:
Q: O, - CO 2e) H,O _H,PO4(aq) _H,Ou Ca(Н,РО as(PO)(OH)s + Ozie) _CO: + H,O _H,0 +
A: According to the question, we need to balance the chemical reaction. Reaction 01: C2H18 + 0₂ ----&g...
Q: How to calculate the mass of 2.7 x 1024 molecules of sugar C12H22O11?
A: Molar mass of C12H22O11 = ( 12 ×12) + (22×1) + (11×16) = 3...
Q: initial change equilibrium
A:
Q: 55.0 grams of aluminum powder is mixed with 75.0 grams of ferric oxide, which of the two substances ...
A: In Aluminium thermite process Al acts as a reducing agent, it can reduce metal from metal oxide.
Q: What is the mole fraction of HCl in an aqueous solution that contains 44.2 g of HCl and 88.6 g of wa...
A:
Q: 10.38 Give the IUPAC name for each compound. а. b. с.
A: Note : As per our company guidelines we are supposed to answer only first 3 sub-parts. Kindly repost...
Q: In experiments on the decomposition of azomethane, CH3NNCH3(g) → C6H6(g) + N2(g) the following...
A:
Q: II. Analysis. Given below is a schematic diagram for a simple analysis of a novel tetrasaccharide is...
A: D- fructose B - glucose C- galactose A-ribose From TLC profile we can see that four carbohydrate...
Q: Consider the following reaction: 4 FeS2 + 11 O2 → 2 Fe2O3 + 8 SO2 When 26.62g of FeS2 reacts with 5....
A: Given data: Mass of FeS2 = 26.62 g Mass of O2 = 5.44 g The molar mass of FeS2 = 119.98 g/mol The mo...
Q: Draw the organic products of the following reactions: +NH, + NAOH(a9) → ? Draw the molecule on the c...
A: In the given reaction the ammonium ion reacts with the sodium hydroxide. The ammonium ion is the con...
Q: Question 10 Which of the two directly affect the intensity of the IR signal? i. Whether the signal i...
A:
Q: for 5
A: ANSWER : Given data are : Actual amount of caffeine in coffee sample = 40 mg and coffee had a caffei...
Q: Complete the following reaction scheme. A H H,0, H,O* HBr, H,02 Br, H,0 conc. H,SO, H;C- CH3 G H,, P...
A: Since you have posted question with multiple sub-parts, we are entitled to solve the first 3 only. T...
Q: Table 2-2: Data and Calculations for Metal + HCI (aq) Write all units next to the numerical values. ...
A: Given data:
Q: How many calories of heat are required to lower the temperature of 550 grams of water from 64.4℉ to ...
A: Mass of water = 550 grams Initial temperature = 64.4 °F Final temperature = 53.6 °F Specific heat...
Q: Question 13 An ē at its excited state is stable unti it return to its ground state. If an ē jumped m...
A: When an electron absorb energy then it excite to the higher energy level and return back to lower en...
Synthesize the following molecules starting with benzene.
need detailed steps.dont copy
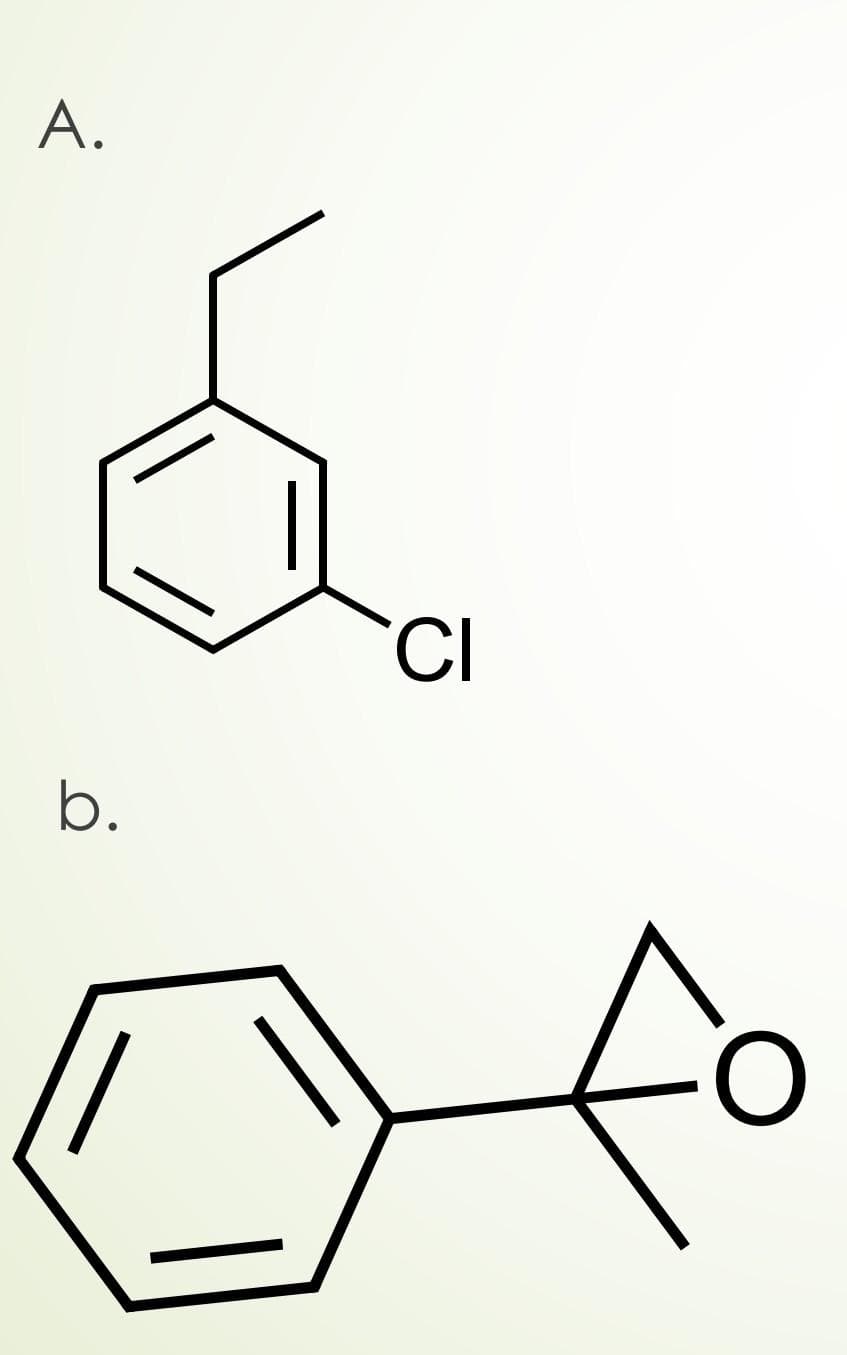
Step by step
Solved in 3 steps with 4 images


