Q: The reaction of 2-ethyl-1-pentene with Br2, with H2 + Pd/C, or with R2BH/THF followed by aqueous HO-…
A: In bromination reaction, bromine atom is introduced to a compound. Hydrogenation can be done using…
Q: ) Purpose the efficient synthesis for the following transformations: B) Give the product(s) for each…
A: The synthesis of the given compounds are given below-
Q: NH2 NH. HO-P-O Br, in H,O OH ОН ОН Вa(ОН). е.
A:
Q: ce CH3-C=CH 2 Hce CH3-C-CH3 ce
A: In the first step the terminal carbon atom of the alkyne will get protonated by one HCl molecule.…
Q: Explain the mechanism of the following reactions and give the name of product? CH,CH(OH)C(CH,), –…
A:
Q: A) Show the mechanism of the conversion of 2-methyl-1-propene (shown below) into PAN…
A: A) The mechanism can be shown as follows: Step 1: The initial reaction is between 2-methyl-1-propene…
Q: The product of the following reaction was isolated in 99% yield. What is it?
A: The reaction scheme is generally framed by considering the factors like atom availability and…
Q: a) Show the mechanism of the conversion of 1-propene (shown below) to CO2 and peroxy acetyl nitrate…
A: 2-methyl-1-propene When react with O3, produces CO2 and ketone in the atmosphere and when ketonic…
Q: HO2C n-BuLi, CO2 H H Me.,, THE Ме., -78 °C Me Me Me Me
A:
Q: 3. Provide the mechanism for the following rxn: ethanol heat Br a. CH3 CH3 NAOCH3, CH3OH Br b. DH H.…
A: Mechanism of Elimination reaction:
Q: 4. Write the product and give mechanism. *CO Et (ii) -CH3 RCN/.. \PMe3 Rh Me,P
A:
Q: Primary and secondary alcohols can be converted to alkyl chlorides via treatment with thionyl…
A: In this step of mechanism there is an attack of nucleophilic chloride ion (Cl-) on the electrophilic…
Q: 5. (a) Carry out the following conversions with suitable reagents, reaction conditions and provide…
A:
Q: NaSCH3 DMSO Mechanism Determine the anticipated ELIIMINATION mechanism for the reaction below. Using…
A: a) Here, substrate is secondary alkyl bromide and nucleophile is strongest that is NaSCH3 and…
Q: 7A. Show the sequence of reagents needed to accomplish the synthesis shown below. H,C- H,C-CEC-H CH,
A: Conversion of alkyne to alkene can be achieved by partial hydrogenation. It can be reduced to…
Q: (8) Draw the principal organic product cxpected from the following reactions; indicate…
A:
Q: (iii) Explain the products in the following addition reactions with mechanism. CH i) Hg(OAC)2, H,0…
A: Here the reaction on right side is addition of BH3 to a double bond followed by basic hydrolysis to…
Q: 3. Write the product and the mechanism for reaction: Kynol. to H3C H₂C AICI3
A:
Q: 3) Predict the products of the ff reagents upon ozonolysis (reductive-work up): A. В. - CH
A: Ozonolysis of alkene: One of the important reactions of alkene is ozonolysis. In the ozonolysis…
Q: Provide a detailed step-by-step mechanisms for the reaction shown. CH;CH2SH + Br, + NaOH
A:
Q: 3. Propose a mechanism for the formation of the 4 alkenes shown below from the reaction of…
A: hydroxy group abstract hydrogen from sulphuric acid and OH will convert into good leaving group…
Q: 1. Illustrate details mechanism of reactions for bromination of the following alkane. State each of…
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question…
Q: CH2-CH3 1. СО 2. H3O*
A: Given incomplete reaction is : Give the major product of the reaction = ?
Q: for each reaction give the expected substitution product and predict wether the mechanism will be…
A: Given reactions, a) 2-chloro-2-methylbutane + CH3COOH b) isobutyl bromide + NaOMe c)…
Q: ОН to CF3CO,H
A:
Q: please show the detailed mechanism for each step for the reaction below. OTBDPS Me TMANO (anhydrous)…
A: Step 1 : alklyne addition to two coblat atoms. Step 2 : olefin addition to one of the coblat atom.…
Q: 4. Provide reasonable mechanisms for the conversions below. но- H3C, ICHS H,O* H3C CH3 он
A: The given mechanistic pathway demonstrates a ring contraction mechanism by means of…
Q: Predict the major products formed when 2- methyl-1-butene reacts with: H2, Pt/25°C. Show the…
A:
Q: CH,=CH-C-CH3 ОН CH3-CH2-CH2-C-H CH;CH3
A: Base used for formation of enolate (nucleophile).
Q: Propose a mechanism for the following reactions. Use an appropriate organic chemistry software (e.g.…
A:
Q: The reaction below is the addition of HCl to 3-methylenecyclohex-1-ene. CH2 + HCI (i) Draw and name…
A:
Q: Predict the most likely mechanism for the reaction shown below. CI CH₂ CH3 H₂O A
A: The following mechanism are as follow.
Q: 49. Identify structures A and B in the following reaction sequence. D 1) 0₂ 2) DMS A NaOH H₂O B…
A:
Q: a) Propose a mechanism for the reaction shown below and predict the stereochemistry at C2…
A: K-Selectride is a reducing agent containing KBHs-Bu3 which reduces cyclohexenone to cyclohexanone…
Q: Which of the structures below (A - D) is the major product (Y) of the following reaction scheme? 1.…
A:
Q: What is the major organic product generated in the reaction sequence below H3C OH 1. H,SO̟ / heat 2.…
A: The reaction taking place is given as,
Q: 30. Provided a detailed mechanism showing all intermediates and the proper curved-arrows shown how…
A: Cyclohex-3-enol on treatment with aqueous sulfuric acid followed by heat produces…
Q: Give the major product(s) of the following reaction. KMNO4 -HO- H2SO4
A: KMnO4 and H2SO4 both are oxidising agent .they will oxidised primary alchol to carboxylic acid and…
Q: N-CH3 DMF Cu-N F3C CI
A: (1 10-phenanthroline)(trifluoromethyl)copper(I) is an important organometallic reagent. It reacts…
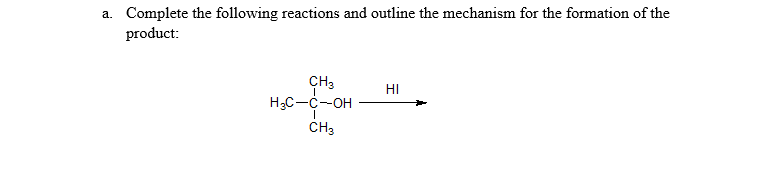
Q: a. Complete the following reactions and outline the mechanism for the formation of the product: CH3…
A: Hello. Since your question has multiple questions, we will solve the first question for you. If you…
Q: Li/ C2H5NH2/-78°C ? 2-Pentin
A: The organic reactions are carried out with the help of various reagents. Each reaction is carried…
Q: 5) Provide the products necessary to complete the synthesis below. Indicate what mechanism was used…
A: The above organic transformation is given below
Q: Give the major product(s) for the following reaction. CH2OH он 0- CH; CH,OH он он он
A: The following is the reaction of the given compound with ethanol in presence of H+
Q: 5) Obtain the suitable product(s) for acid-catalyzed "ether cleavage (A-DY" arud "acid-catalyzed (E…
A: SN type reactions in ethers: Primary alkyl ethers readily undergo SN2 reaction , in…
Q: II. Predict the products of the ff: elimination reactions and show the mechanisms involv HO H2SO4…
A:
Q: b) Ozonolysis of an unknown alkene yields the diketone shown in the scheme below. Suggest the…
A: This is the ozonolysis reaction of Alkene.In this reaction ozonide is an intermediate form and PPh3…
Q: a) Complete the following reactions: (i) H;C-C-Ci + H2O A + B HO H;C. CH3 SOCI, + D + so, (iii) H;O*…
A: Introduction : We have to make the products for the following. As per company norms we can not…
Q: CI H2N (-HCI) ??? NH2 2.
A: the reasonable mechanism for the reaction is
Q: 10. Complete the following reactions: , CH3 + KOH - Bm b) CH3 CH2 CH=CHz + Hz0
A:
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- A chemist reacts 2.4 grams of 2-methyl-2-butanol with 15 mL of 6M sulfuric acid. What is the most likely mechanism for alkene formation in the elimination reaction (please draw it out to demonstrate).] The reaction of 2-bromopropane and sodium ethoxide in ethanol reacts 6.7 times faster than 2-bromo-1-deuteriopropane under the same conditions. Explain what mechanism this data is consistent with, and whyGive a detailed mechanism for the reaction utilizing oxygen-18 labeled (18O) 1-butanol and acetic acid. Label the 18O throughout the mechanism.
- E 21 write mechanisms for the given reactionsFor each of the compounds given, write the mechanism for the reaction between the compound and LDA in THF at low temperatures (-78 deg C).I am working on a practice assignment for my organic II course and am having difficulty with a question that asks to identify the reaction sequence used to synthesize isopropylcyclopentane. I would really appreciate the help!
- (ii) The elimination reaction between 2-bromobutane and NaOCH2CH3 gives two organic products. Draw a mechanism for the reaction which produces the major organic elimination product and provide a rationale as to why that is the major product.(with explanation)provide the structure for the major organic products for the reaction. identify the mechanism as SN1, SN2. Show stochiometry30. The following molecule is susceptible to nucleophilic attack at the carbonyl carbon as well as two other positions. Provide an explanation for this observation (structures would be useful).
- The synthesis of B-ionone (reterisyntheric analysis ) organic 2 Please explain the synthetics steps in details.(ORGANIC CHEMISTRY) (mechanisms) please give a answer for the following mechanism. Provide a detailed stepwise mechanism for the following transformation.write the reaction of the action of methyl-2, bromo-2-propane with hot KOH solution. a) Name the formed product b) Explain why the reaction takes place according to the SNI mechanism

