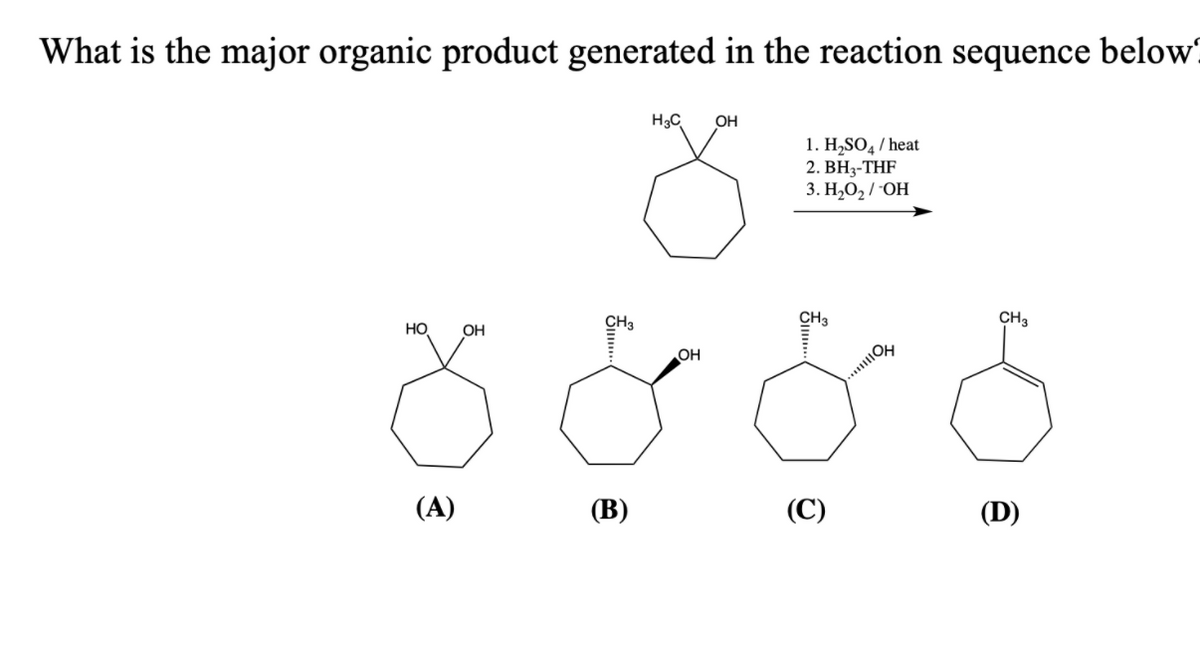
What is the major organic product generated in the reaction sequence below H3C OH 1. H,SO̟ / heat 2. BH3-THF 3. Н.О,/ ОН CH3 CH3 CH3 HO OH OH (A) (B) (C) (D)
Q: draw all four products of this reaction. Assume that the reaction is acidified to pH 2 after…
A:
Q: Which of the following reactions can be used to prepare 3-methyl-3-hexanol? Note: n-Pr stands for…
A:
Q: ОН ARSO3H + H20 Dihydropyran is synthesized by treating tetrahydrofurfuryl alcohol with an…
A:
Q: OSO₂Ar Arrow-pushing Instructions 00 H H H H H Solvolysis of this bicyclic compound in acetic acid…
A: The products are formed via Sigma bond NGP mechanism. The detailed mechanism is given below in step…
Q: NC-CH=CH-CN N. CH CH2 CH 3 76 ROOC -C=C-COOR
A: The organic compounds in which carbon atoms are bonded to each other by a double bond or by a triple…
Q: Organic Chemistry Rearrangement reactions. Name of reaction, Intermediate and educt. Thank you in…
A: Concept behind this question 1. The targen molecule could be synthesized from cyclopentaone through…
Q: give the organic chemical reaction mechanism of the synthesis of acetaldehyde.
A:
Q: HBr NaCN HCI/H2O SOCI2, A C D. E H2O2
A:
Q: 4. Give synthesis O H3CN- H3CH₂C -NH₂
A:
Q: Give the major product for the reaction below. A H20 HCI cold, dilute B KMNO4 KMNO4, OH, heat E HBr…
A: The reactions are summarized as: The acidic hydration of alkenes takes place according to…
Q: U1. Show the reaction steps and final products in the following reaction H2 H,C-C OH H2 1. H,C-OH…
A:
Q: 1. As you can see in Fig. 1(C), there is an interaction between A475 on SARS-CoV-2 and S19 on ACE-2.…
A:
Q: HO2C n-BuLi, CO2 H H Me.,, THE Ме., -78 °C Me Me Me Me
A:
Q: Osaw the Yeaction mechanisms for the reduction NABHU g benzshenone using Reduction (o.138g) NABH4…
A:
Q: The fastest method of synthesis of isopropyl methyl ether (below) is: CH3 CH3 CH-0 H3C CH. H;C…
A: The solution is given below -
Q: What would be the product of the following reaction sequence? i) SOa, 11) C Hg, AICI3 i11) Zn(Hg),…
A: Note: As per our guidelines, we are supposed to solve only one question. Please repost the other…
Q: 2. A). Give the structure for the compounds A through E. (There is a mistake in the structure of the…
A:
Q: (е) 1. ВНз, THF 2. HO, H2O2, H2O
A: The reaction is an exam0le of hydroboration oxidation of alkynes resulting in the formation of…
Q: NaH ? LOH HO PBr3 D в A (d) Propose the structure of the organic compound D formed by reaction with…
A:
Q: 23. Congo Red is a synthetic dye whose use was later abandoned because of its carcinogenic…
A: (23) answer: Correct option (b) From Retrosynthesis analysis we can say, Nelson can synthesize…
Q: 4. Write the following sequence of reactions: HNO3 conc. A H2SO4 conc. CH;CI В AICI3 Cact, t°C 3…
A:
Q: i he diagram below shows two synthetic pathways by which the compound W might be prepared. C3H,OH…
A: Compound W is 2-Hydroxypropanoic acid. This Compound has one chiral Center That's why it shows…
Q: k* КОН, МеОН 0 °C, 1 h RT, 1 h 0 °C, 12 h MeCOCI (3 equiv) K* МеОн (2.4 М) 0 °C, 30 min Room Temp.,…
A: A condensation reaction is a combination of two or more molecules to give a single molecule with the…
Q: 6. Complete the following reactions: 1. NaNH3 (CH,),CHCH,CH,C#CH 2. D,0 a. Lindlar's Catalyst H2 1.…
A: The answer is given as follows
Q: Name the major organic product which results when benzenesulfonic acid reacts with Cl2 in AICI3,…
A: Benzene sulphonic acid is meta directing group and its gives chlorination reaction on meta…
Q: 1 Draw the structure(s) of the major organic product(s) of the following reaction. 1. Dry Et,O…
A: Here we have to write the structure of major organic product formed in the following nucleophilic…
Q: ycits – Where'd they go?! o your synthetic toolbelt to fill in reagents (a) - (g) in the spaces…
A: Given : We have to write the reagents and products for the following transformation.
Q: Question 3
A: Organic compounds that contain a carbon atom directly bonded with metals such as magnesium, lithium…
Q: Identify the starting material in attached reaction.
A: This is reaction is an example of an ozonolysis reaction. In this reaction, the double bond in the…
Q: Write the reaction CH3 CHz Ċ - OH to methyl propylamine 2.) CH3 CH2 C - CI CH3 NH CH3 3-) CH3 CH =…
A: GIVEN:-
Q: nost" und nder the one acetaldehyd e "most" under the one which t" under the one which would be le…
A:
Q: 6. Complete the following reaction scheme (G) H2 Br, Br2 H3C- CH3 EC-CH3 H, CH;-CE (k) Pd/C H,…
A:
Q: For the following reaction, select the major product. 1) CH31 (excess) 2) Ag20, H20 3) heat i. ii.…
A: In this question, we want to select the correct Organic major product. You can see details Solution…
Q: 1. Give the major organic product (s) for the following reactions or sequence of reactions. 1)…
A:
Q: Select the final product from the following reaction sequence: 1. HNO3, H„SO4 2. H2, Ni 3. HNO,, HCI…
A: Benzene undergo nitration in first step and in subsequent steps diazonium salt is formed.
Q: The SN2 reaction ur l-chloro-3-methylbutane with hydroxide OH is relatively slow, but it can be…
A:
Q: 5. 6. Br MeOH i) mechanism: ii) most important determining factor(s): iii & iv) detailed, stepwise…
A: Here we have to write the mechanism of the following given reactions and cause of the mechanism of…
Q: Provide a synthesis for the following reaction. If more than one step is required be sure to number…
A:
Q: Terpin, prepared commercially by the acid-catalyzed hydration of limonene, is used medicinally as an…
A: The required mechanism is given below -
Q: H2 1. Вн/THF A В Lindlar 2. Он, Н-О2, Н,0
A:
Q: 4. Give the product of the reaction below. OCH2CH3 Upload Choose a File
A: This is SN2 reaction and in this reaction inversion product is formed.
Q: - Provide the structure of the major organic product for the reaction sequence shown below. 1.…
A: It is a 3 step reaction as shown in the following step.
Q: Provide the major organic product of the reactions shown below. Show the from each step shown below.…
A: ->Na2Cr2O7 is oxidizing agent which can oxidize primary alcohol to carboxylic acid.…
Q: product formed when the compound shown below undergoes a reaction with 1 equivalent of AcCI. ACCI =…
A: Using concept of nucleophilic substitution by N- center.
Q: Tunicates are marine animals that are called "sea squirts" because when they are taken out of water,…
A:
Q: i. LIAIH4 i. deprotection .CO2ME J CH2=CHCOCH3 protection ii. H30* ii. dehydration K H The synthesis…
A: Here we have to write the major product formed and reagents involved in protection , deprotection…
Q: CI H2N (-HCI) ??? NH2 2.
A: the reasonable mechanism for the reaction is
Q: noose the best reagent(s) for carrying out the conversion of the reaction below CH3CH-CHCH2COOH -->…
A: For the given reaction, oxidation of alkene occurs.
what is the major products?
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

- Which compound is not a possible product in the reaction below ( F2 is in excess F2and UV light the catalyst).CHA + F2 -->O CFAO CHFaO CHF-O CH2F2O CHaFArrange the structure on the image with regard to the reactivity towards hydrolysis. 1 being the least and 3 being the most(a) Illustrate the following name reactions giving suitable example in each case :(i) Clemmensen reduction (ii) Hell-Volhard-Zelinsky reaction(b) How are the following conversions carried out?(i) Ethylcyanide to ethanoic acid (ii) Butan-l-ol to butanoic acid(iii) Benzoic acid to m-bromobenzoic acid
- I am working on a practice assignment for my organic II course and am having difficulty with a question that asks to identify the reaction sequence used to synthesize isopropylcyclopentane. I would really appreciate the help!Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Propose structural formulas for compounds (2) and (3).Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Name the types of functional groups in estrone
- Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.How many chiral centers are present in estrone?Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Show how your proposals for compounds (2) and (3) can be converted to compound (1). (Note: In the course of developing this synthesis, Tietze discovered that vinylic bromides and iodides are more reactive in Heck reactions than are aryl bromides and iodides.)Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q. In the course of the double Heck reactions, two new chiral centers are created. Assume in compound (3), the precursor to rings C and D of estrone, that the fusion of rings C and D is trans and that the angular methyl group is above the plane of the ring. Given this stereochemistry, predict the stereochemistry of…
- 25 For SN1SN1 solvolysis of t‑butyl chloride, rank the solvents from fastest reaction to slowest reaction.Given the sequence of reactions shown below assume the reactions are stoichiometric fill in the reagents needed for steps A B and C and explain what happens in each step of the reactions, by also nameNitrating methyl benzoate. 1.Describe the mechanism of EAS clearly and in detail 2. What is the effect of subtituent of the outcome of EAS;include 3 classes of subtituents



