Chapter8: Sampling, Standardization, And Calibration
Section: Chapter Questions
Problem 8.19QAP
Related questions
Question
Please help with 1,2,3,4 thank you

Transcribed Image Text:A Rate Law•
Date
Name.
pes qo
A. Determination of Reaction Times
Volume of Na,S,O, (L)C.
.10
Molar concentration of Na,S,O, ♡20
Ambient temperature
22
°C
30
; Molar concentration of H,O,
Total volume of kinetic trials (mL)_I0.00ML
Molar concentration of KI
Kinetic Trial
1*
2
3
4
5
6
1. Time for color
change, At (sec)
75
36
23
17
48
B. Calculations for Determining the Rate Law
1. Moles of S,0,2-
consumed (mol)
2. A(mol I,") produced
A(mol I)
3.
(mol/s)
At
A(mol I,)
4. log
At
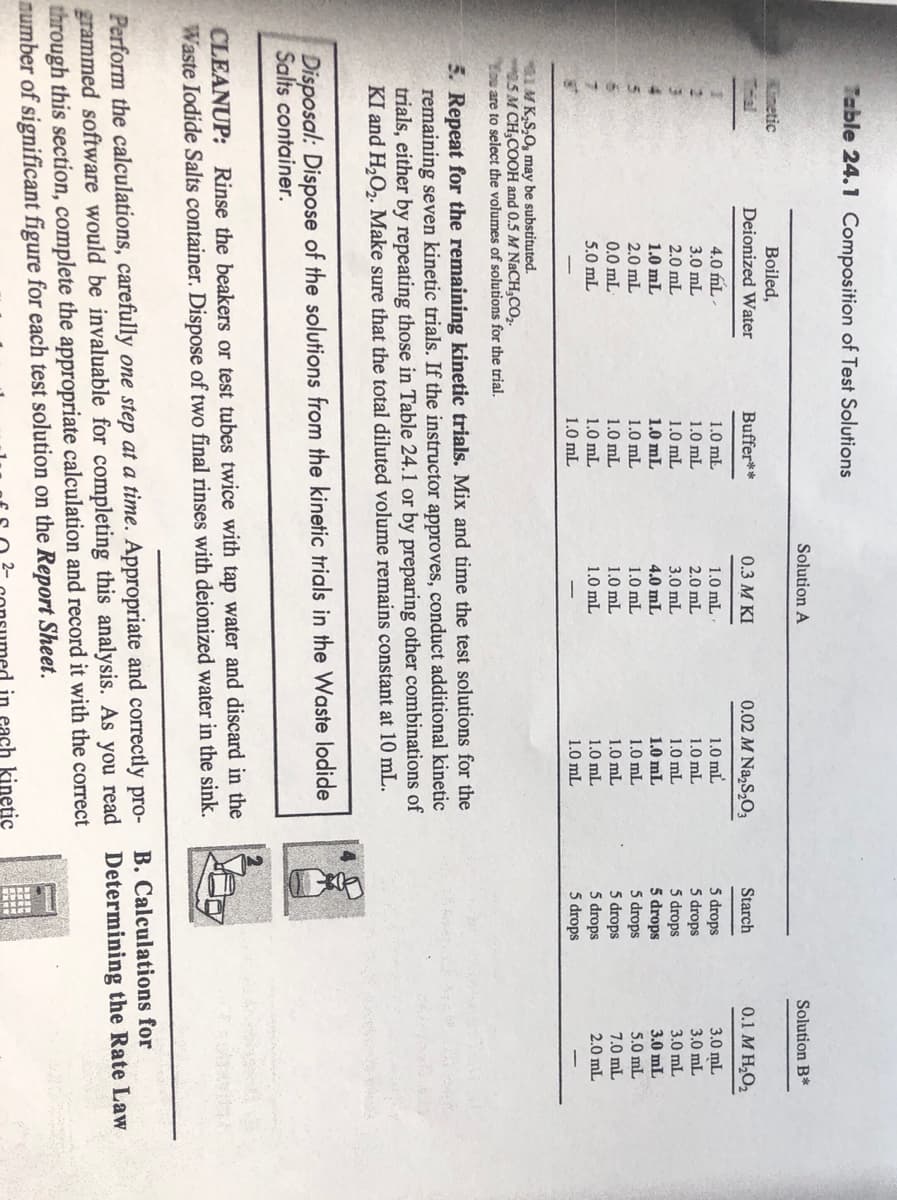
Transcribed Image Text:Table 24.1 Composition of Test Solutions
Solution A
Solution B*
TII
Knetic
Thal
Boiled,
Deionized Water
Buffer**
0.3 M KI
0.02 M Na,S,O3
Starch
0.1 M H2O2
4.0 mL -
5 drops
5 drops
5 drops
5 drops
5 drops
5 drops
5 drops
5 drops
1.0 mL
1.0 mL
1.0 mL
3.0 mL
3.0 mL
1.0 mL
2.0 mL
1.0 mL
3.0 mL
2.0 mL
1.0 mL
3.0 mL
1.0 mL
3.0 mL
1.0 mL
1.0 mL
4.0 mL
1.0 mL
3.0 mL
2.0 mL
1.0 mL
1.0 mL
1.0 mL
5.0 mL
7.0 mL
2.0 mL
1.0 mL
0.0 mL
5.0 mL
1.0 mL
1.0 mL
1.0 mL
1.0 mL
1.0 mL
1.0 mL
1.0 mL
1M K,S,0, may be substituted.
5M CH,COOH and 0.5 M NACH,CO,.
You are to select the volumes of solutions for the trial.
5. Repeat for the remaining kinetic trials. Mix and time the test solutions for the
remaining seven kinetic trials. If the instructor approves, conduct additional kinetic
trials, either by repeating those in Table 24.1 or by preparing other combinations of
KI and H,O,. Make sure that the total diluted volume remains constant at 10 mL.
Disposal: Dispose of the solutions from the kinetic trials in the Waste lodide
Salts container.
CLEANUP: Rinse the beakers or test tubes twice with tap water and discard in the
Waste Iodide Salts container. Dispose of two final rinses with deionized water in the sink.
Perform the calculations, carefully one step at a time. Appropriate and correctly pro-
grammed software would be invaluable for completing this analysis. As you read Determining the Rate Law
through this section, complete the appropriate calculation and record it with the correct
number of significant figure for each test solution on the Report Sheet.
B. Calculations for
in each kinetic
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Recommended textbooks for you



Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning



Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning