A. If a reaction will occur, give the product(s) otherwise write no reaction. Also indicate if the reaction is fast or slow. Explain your answer. 3. Glycogen + I;/KI
A. If a reaction will occur, give the product(s) otherwise write no reaction. Also indicate if the reaction is fast or slow. Explain your answer. 3. Glycogen + I;/KI
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter20: Carbohydrates
Section: Chapter Questions
Problem 20.58P
Related questions
Question
100%
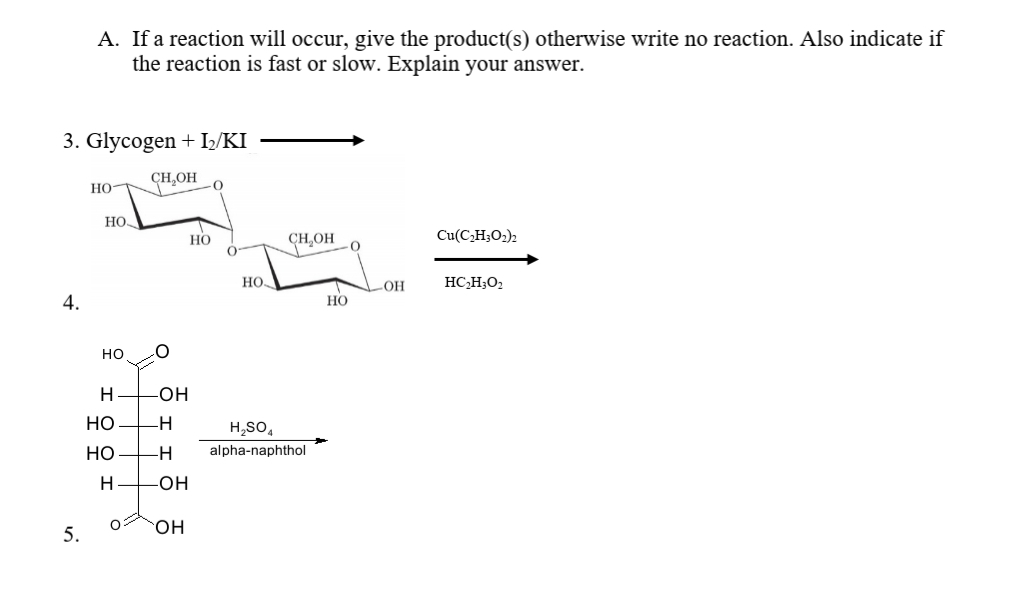
Transcribed Image Text:A. If a reaction will occur, give the product(s) otherwise write no reaction. Also indicate if
the reaction is fast or slow. Explain your answer.
3. Glycogen + I/KI
CH,OH
Но-
но-
Но
CHOH
Cu(C,H;O2)2
но.
OH
HC,H;O2
4.
НО
но
H
-O-
но
-H
H,SO,
но
-H-
alpha-naphthol
H
-HO-
он
5.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning
