Q: 1) 2) OTS Me KO H Base CO₂H BF3.OET2 من Me 45 % 43% I
A: Bases abstract acidic hydrogen from the compounds. Positive charges rearranges in the compound to…
Q: 1) о ? 2) H*/H20 O a. OH Ob. OH OH Od. OH
A:
Q: Br2 NaOH Bra. C 1. LDA (excess) D 1. NaH B H,C CH, hr E 2. CH,Br CI, 2. H,0º
A: when alkane reacts with Br2/hv then it will give Alkyle Bromide as a product, and this reaction is…
Q: (g) (g) AICI3 name of reaction (h) (h) name of reaction (i) NH NH, KOH (i) name of reaction
A: Benzene has higher electron density due to the presence of delocalized pi- electrons. hence they can…
Q: а. Cr (aq) + Cl; (9) — Сr207? (ад) + CI (аq) V b. Ni2+ (aq) + Ca(s) — Са* (аg) + Ni(s) V
A:
Q: 1) Hg(OAc)2, MeOH 2) NABH4 ?
A:
Q: 43. H Br "X :S: || 1. HNCNH, 2. NaOH, H₂O
A: Thiourea contains nucleophilic centre S which reacts with electrophilic bromine containing carbon…
Q: In a reaction, Al2(SOa (aq) reacts with K3PO4 (aq), what will be the product(s)? O AlgP, K(SOA2…
A:
Q: AG°F nol·K)] kJ/mol) _8 65.1
A:
Q: 1.) HAI(Bu), or DIBAL-H 2.) H,0", H,0 CEN: H. R' H. 3 3 1.) HAI('Bu)2 or DIBAL-H 2.) H,O*, H,O…
A:
Q: What is true for this reaction? CI4(1) + 4 HCI(g) CH4(g)+ 4 Cl2(g) O AHrxn 0 AHrxn 0; ASrxn 0;…
A: For the reaction ., CCl4(l) + 4HCl(g) ----> CH4(g) + 4Cl2(g) Bond energy ., For C-H bond = 414 KJ…
Q: Complete the reactions given below Br NaOC,H5 / isi b)
A: In presence of base alkyl bromide give elimination reaction ,and form alkene.
Q: (1) Hg(OAc); THF, H;O (1) (2) NaBH, но (2) HO (3) (4) HO Compound (3) b. Compound (1) Compound (4)…
A:
Q: HCI C,H,CI OH CH,O Na,Cr,O, C,H,O Compound A H20 Compound B Compound C
A:
Q: HCI a HO- 人o H2SO4 I H2SO, H20
A: A. Addition of HCl across the double bond in acidic conditions.B. Formation of more stable…
Q: (a) HgCl2 • 2 MeMgCI (b) GaMes Me,NH heating (c) BH3 сно BrPh,P base (d)
A: Here the reactions are reaction of Grignard reagent with metal chloride Forming an adduct of ammonia…
Q: 6. 7. 8. OH Naon CO2 H30+ Воз, ревоз Br2, Sa, tel 4 ΟΥ Fe, Hel, 4 HNO2, H₂SO4 Curl A S03, W₂504 NO.…
A:
Q: Determine el nombre IUPAC para los siguientes compuestos: Br HO NH2 CH3 CI Br OH
A: The IUPAC name of the following compounds are:
Q: d. HCl(aq) + KOH(aq) H2O(1) + KCI(aq) - e. Zn(s) + CuSO«(aq)ZnSO4(aq) + Cu(s) | f. NaH,PO4(aq) +…
A:
Q: (a) HCIO(aq) + H20() = H30*(aq) + CIO(aq) O K-= [H3O*][CIO'/((HCIO][H;0]) O K-= [H3O*][CIO*/[HCIO] O…
A:
Q: Br() "0. 2 Br(aq) + Reactants Products LIOH(s) "0. Litaq) + OH(aq) Reactants Products H20 FeF,(s)…
A: Since you have posted question with multiple subparts as per guidelines we can answer only three per…
Q: 3)b. FePO4*6 H2O d. CUCN f. CuSO4 * 5 H2O h. Sn(SO4)2 j. Mn(CH3CO2)2 1. Hg(NO3)2 * H2O n. CRSO4 *…
A: Chemical name of following compounds
Q: A. COMBINATION or SYNTHESIS REACTION: A + B→AB 1. Fe +_O2 → _Fe;O3 2. Li+_d →_ Lid _C;H2 + _F…
A: Answer A. Combination reactions 1) 4Fe + 3O2 → 2Fe2O3 2) 2Li + Cl2 →2LiCl 3) C2H2 + 2F2 → C2H2F4 4)…
Q: CaCla) +NHy)
A: 3CaCl2(aq) + 2NH3(aq) --> Ca3N2 (aq) + 6HCl(aq)
Q: CH,COH H;SO, CH, KCr,O, CH;COOH H;SO, K.Cr,O, CH CHOH
A: Balance Chemical equation means no of atoms should be equaal in both side reactant and product
Q: (a) Iz(s) + 5 Cu²* (aq) + 6 H2O(1) (b) Hg²*(aq) + 21 (aq) (c) H2SO3(aq) + 2 Mn(s) + 4 H*(aq) → S(s)…
A: I2(s) + 5Cu+2 (aq) + 6H2O → 2IO3-(aq) + 5 Cu (s) + 12 H+ E cell of the reaction = Ecathode…
Q: What are the products of the following reactions?
A: a)
Q: HNO, (1 eq.) H2SO, Br 0 °C NO2 HNO, (1 eq.) H2SO, 0 °C он
A:
Q: OH OH OCH₂ ii + H 1. DIBAL-H 2. H₂O* cat. H*A* excess NaOCH3 Br Br KCN HCN
A:
Q: Br OH а. NaCOOH/ DMSO b. 1. КCN, 2. НзО", сalor С. 1. NaOH; 2. НаCrО4 d. 1. 2 Lil éter; 2. 3. H2CrO4…
A: For 84 the reaction sequence is as follows Option d
Q: O Aind the Concentiation A HaO in a O,250M Ihadirofuaric Oad (HF) soutione the Ka kr HFIS 3.50 104…
A: Since, HF is a weak acid. Weak acid is the one which undergoes partial dissociation. The…
Q: A. B. C. D. 0= Ph OEt E. 1) LDA, -78 °C 2) O X H + Ph 3) H+ NaOH, H₂O, A NaOH, H₂O, A 1) Me,NH 2) 3)…
A:
Q: 1. CI , AICI, 2. Zn(Hg), HCI OH of II IV V O a. I O b. II O c. III o d. IV O e. V
A: Benzene is an aromatic compound and it has three pi bond which are in resonance with each other. Due…
Q: Aै लोकणर पजोन विमीत)ष्ठे भैव्टे B्ीe वष्डे मी ऐए+ mdtiefuocbe्च छक्यो्n 6.torवै a) do tution त…
A: Correct answer is :- Suspension (option- c)
Q: NaOEt 1. Br NaOH Br 2. NaOtBu 3. MeOH 4. Br
A: For the given reactant the major product are formed in the next steps
Q: a. Pb(NO3)2 (aq) + Al2(SO4)s (aq) → b. HBr (aq) + Ca(OH)2 (aq)→ c. H2SO4 (aq) + Hg (1) → d. HNO3…
A: Balanced reaction: A reaction is said to be balanced if the number of atoms of the each element and…
Q: HO ∞ H Br KMnO4 NaOH 0 °C 1. KMnO4, NaOH, A 2. dil. H₂SO4, H₂O 1. KMnO4, NaOH, A 2. dil. H₂SO4, H₂O…
A: Here we have to predict the major products formed in the following given reactions.
Q: PbSO4 O Pb(NO3)2 (ag)+ Na2SO4 (ag)→PBSO4(s) + 2NaNO3(ag) O Pb(NO3)2(aq) + BaSO4 (s)→PBSO(s) +…
A: The precipitate of compounds form when two aqueous solution of one compound is mixed with aqueous…
Q: m. Ag(NH,), + -OH ac- H,0 CI.C- H- n. Ag(NH), + -он Br—CH,CH, —С-н H,0 O. Ág(NH,), H. НО- + H,0
A: Interpretation- To complete the given following reaction which is given as above -…
Q: Use the values of ∆Hfº in Appendix 4 to calculate ∆Hº for the following reactions.
A: ∆H0 = ∆H0product - ∆H0reactant
Q: NHa CN > HCN NH3 A B Oc OA B. OD
A: Conjugate base is produce when acid is deprotonated. Conjugate acid product when base is protonated.
Q: 12 N2 (g) + !/2 0: (g) → NO (g) /2 N2 (g) + O2 (g) → NO2 (g) 2 NO: (g) →N;0, (g) AH = 90.4 kJ/mol AH…
A:
Q: (2a) 2 HCI (aq) + Mg (s)- MgCl2 (aq) + H2 (g) 1) (2c) HC2H3O2 (aq) + NaHCO3 (s) → NaC2H3O2 (aq) +…
A:
Q: 18 ya.) HCO3 Tog) + OH:(a). -Co3?7og) + HzO () b) Noz" Cas) +. HCID4los) HNOZ Cag) + Clay Cag)
A: 18) Given reactions, (a) HCO3-(aq) + OH-(aq) → CO32-(aq) + H2O(l)(b) NO2-(aq) + HClO4(aq) →…
Q: Br Me A CN NH₂ 1. Br2, FeBr3 2. BrMgCH₂CH₂CH3, THF; aq. HCI 3. NaBH, MeOH 4. NaH, Mel, DMF Br B OMe…
A:
Q: Br NaOEt .CI (еxcess) ETOH NC CI
A: Aromatic nucleophile substitution: The aromatic nucleophilic substitution reactions occurs…
Q: ONLY OH HO NH2
A:
Q: i) H. .CO2H 150°C ? + H20 90% H `CO,H Maleic acid ii) (CH;CO),O + (CH3)½C-OH ÇOOCH3 i11) + OH NAOH ?…
A:
Q: 1. H₂CRO4 2.NH, CH3 1. Swern 2. 2 mol EtOH OH 1. H₂CRO4 2. CH₂CH₂CH₂Br. PPh3, KOA 1. KMnO4 2.NH, CH3
A: Given are organic reactions
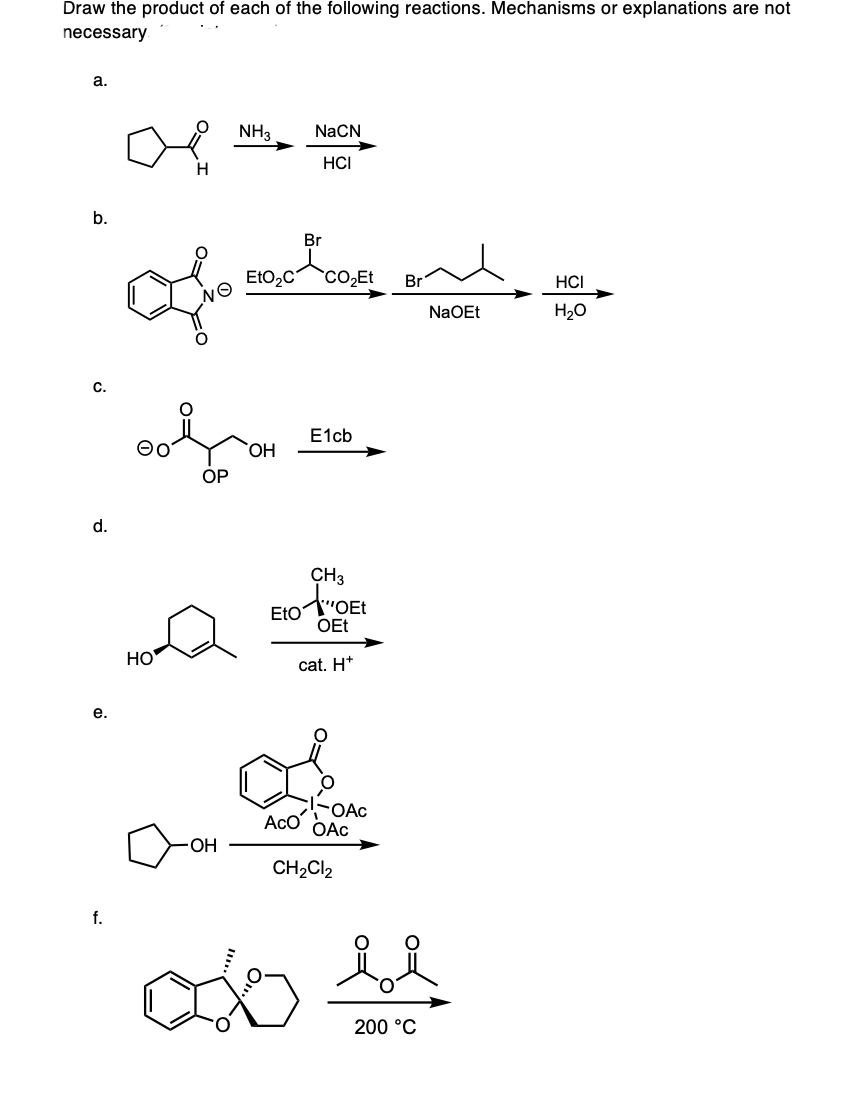
Step by step
Solved in 2 steps with 1 images

- What type of mechanism is exhibited in the reaction? SN1, SN2, E2, E1?Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- Rate = k [CH3CH2Br][CH3COO-] Which mechanism would best fit the data?Show the mechanism and formation of all products for your alkene for each selected reaction (3 separate complete mechanisms). As part of your submission you must: -An explanation of each step of your reaction (as if you were teaching someone) - Identification of the major product - Explanation for why you selected the major product Please make sure to explain in detail for each of the point listed above
- Propose a mechanism for the observed product based on what you know about SN1 and SN2-type mechanisms.Please help with the following ochem mechanisms.... 1. Provide the stepwise mechanisms for the following reactions (see attached picture)Identify which substitution mechanism best fits the following statement: The reaction proceeds through a concerted mechanism. A) SN1 B) SN2
- SHOW THE MECHANISM AND STEPS: Benzene---> Propiophenone(C9H10O)----> Methanolcan someone help me with this? Make a comparison table between the reaction mechanism SN1 and SN2, take into account thereaction order, nature of the substrates, solvents used, nature of the nucleophile, stereochemistry,kinetics, and other characteristics that you consider relevant.For each reaction indicate and explain the predominant mechanism: SN1, SN2, E1 or E2
- Chemistry please provide the flow of electrons aswell!! thank you! using the starting material (on the left) to determine the sythetic route which will be the most reaosnable and effective to theres none, you have to start witth the begin products to get to finish needed by using the minmium reagents and reactions needed to get to the final productFor which reaction mechanisms—SN1, SN2, E1, or E2—of thefollowing statement true? A statement may be true for one or moremechanisms. Tertiary (3°) alkyl halides react faster than 2° or 1° alkyl halides.Show the mechanism of the following reactions and show their products.1. CH3CH2OH+ HBr --> ?2. CH3CH = CH2 + HCl --> ?3. C6H6 + HNO3 --> ?4. CH3CH2OH + H2SO4 (concentrated) --> ?5. CH3O- + CH3Br --> ?

