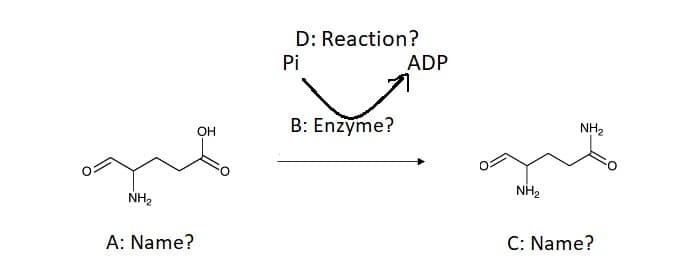
a. Refer to the reaction below and provide A,B,C and D. b. What type of reaction is taking place in reaction D?
Q: Consider the multistep reaction below. What is the balanced chemical equation of the overall…
A: A chemical reaction can be fast or slow depending upon the presence or absence of catalysis. The…
Q: Complete the following tables by providing the reactants/substrates, enzymes and their functions as…
A: Energy is needed to the cell and the energy is obtained via the metabolic pathways. Metabolic…
Q: (d) Derive Michaelis-Menten equation of enzymatic reaction.
A: Michaelis Menten equation shows the relationship between the concentration of the substrate and…
Q: Balance the Alka-Seltzer tablet chemical equation and Identify the acid and base in the reaction.
A: Seltzer tablets neutralize excess stomach acid. These tablets dissolve in water when ingested. When…
Q: Define the following terms:a. allyl groupb. epoxidec. SAM d. PAPSe. phase I reaction
A: In chemical structure, the group of atoms that replace the hydrogen atoms on the parent hydrocarbon…
Q: Identify the redox reactions involved in nucleic acid metabolism. Explain microscopically
A: For the cell growth nutrients is predominantly required. Cells in the medium can generate reducing…
Q: h. Give an outline of one example of an anaplerotic reaction.
A: Introduction Anaplerotic reactions are metabolic mechanisms that restore oxaloacetate after it has…
Q: Give a reaction equation for the qualitative and quantitative determination of I2.
A: Iodine: Iodine (I) is a lustrous element; atomic number is 53. It is the heaviest of the stable…
Q: The equilibrium constant for the reaction Q → R is 25. Solve, (a) If 50 μM of Q is mixed with 50 μM…
A: The constant of equilibrium, K, represents the relationship between products and reactants of the…
Q: Explain whether the statement, "Linking the energetically unfavorable reaction A- B to a second,…
A: Chemical thermodynamics depicts an energy-absorbing, non-spontaneous reaction with a positive…
Q: B: For each of the following reaction types, find a step in a pathway that is an example and for…
A: Proteins that catalyze biochemical reactions inside living organisms are called enzymes. Depending…
Q: b) The above molecule may be further converted into a polymer with similar molecular structure to…
A: The molecule given in the question is named the molecule" 5-hydroxymethyl-2-furaldehyde or…
Q: Explain whether the statement, "If an oxidation occurs in a reaction, it must be accompanied by a…
A: Introduction :- Redox reactions include a change in the oxidation state of the substrate. Loss of…
Q: Identify the oxidized reactant, the reduced reactant, the oxidizing agent, and the reducing agent in…
A: The electron accepting molecule or ion is known as oxidizing agents. As the electrons are accepted…
Q: ОН 9. CH OH 1 H ОН Complete the following reaction by drawing the molecular structure of the main…
A: Sugars are the most essential components that have various roles in plants and animals alike. These…
Q: Describe the processes involved in anabolic and catabolic reactions
A: Metabolism is the sum of anabolic and catabolic reactions. The metabolic rate is used to measure the…
Q: What type of reactions is represented below? CH,OH CH2OH C=0 H-C-OH HO-C-H HO-C-H CH,OH CH2OH O…
A: This is a ketotetrose which is a tetrose having a single ketone group at the 2-position.
Q: what is the equilibrium constant for the reaction A + CT AT+ C? Show calculations. 2) the image
A: The equilibrium constant, K, expresses the interaction between the products and reactants of a…
Q: [References] In the following reaction, the rate of appearance of I, is measured at the times shown.…
A: Introduction The rate of a reaction is given by rate = ΔP/Δt where ΔP is the change in product…
Q: Give at least 5 examples of biological compounds having an alcohol functional group and identify the…
A: Biochemical compounds are those that are present in living organisms and are involved in different…
Q: If the starting conditions were standard conditions (at 298 K) O The reaction would proceed from…
A: ∆G=-RT In(K'eq) A reaction moves in the forward direction (left to right) if the delta G for the…
Q: Given log þ. 2. b. Balance the following reactions: S S(s) + NO(g) in acidic medium (aq) 8 (aq)…
A: Answeing 4b as you have asked. In acidic medium, protons will be there. The protons along with the…
Q: (i) Name the reaction. (ii) Complete the reaction by drawing the structures of the products of this…
A: The fatty acids that occur in nature are acted on by sodium hydroxide to generate sodium salts of…
Q: Write the different classes of enzymes and there corresponding reactions.
A: Introduction: Proteins That Operate As Biological Catalysts Are Known As Enzymes (biocatalysts).…
Q: Consider a transition of X → y. assume that the only difference between X and y is the presence of…
A: Introduction Equilibrium is a condition of equilibrium or a stable situation in which opposing…
Q: Explain the metabolic significance of reactions that function near equilibrium and reactions that…
A: In a metabolic pathway, reactions can be divided into two categories: 1. The ones that are very…
Q: Same diagram, but now identify the flat line for the product molecules. The energy of the product…
A: A reaction mechanism, also known as a reaction route, specifies the molecular processes that occur…
Q: d) Which reactions are predicted to be far-from-equilibrium? Explain your rationale. e) What type of…
A: Glyoxylate cycle is an anabolic pathway that occurred in plants, bacteria, protists, and fungi in…
Q: Curve A (red), is enzyme catalyzed reaction treated with a compound. Curve B (blue), is the same…
A: The given graph represents the Lineweaver-Burk plot. Lineweaver-Burk plot is the plot between 1/V…
Q: 1. In zero order reaction, the reaction rate depends only on enzyme concentration. In first…
A: Thank you for the question, as per the honor code, we are allowed to answer the first question at a…
Q: Y WX WX W In the diagram, which represents a catalyst? O W O WX
A: Enzymes are catalysts which catalyze the biological reactions. Enzymes are protein molecule having a…
Q: -8 K, for HCO3 ` = 2.38 × 10 Calculate Ka for the conjugate acid ( H¿CO;) For the reaction :…
A: Conjugate acid-base pairs are those that differ from each other by a proton or H+ ion. For example,…
Q: How will each change affect the reaction? HNO2(aq)⇌H^+(aq)+NO2^-(aq) a.) Decreasing volume b.)…
A: Introduction According to Le Chatelier's principle, if a reaction at equilibrium is stressed, the…
Q: When K > 1, which of the following statements is/are definitely TRUE? I. Products are favored…
A: CONCEPT For a given reaction A + B <--------> C+ D At equilibrium, the rate of…
Q: DIY Glue State the materials that use to make it. Show the mechanisms of the reaction that occur.
A: The materials necessary for the preparation of glue are as follows: 12 cup of skim (non fat) milk 1…
Q: Which of the following statements about the reaction shown is not necessarily true? LO'Na NaOH H,C.…
A: An ester is a chemical compound which is derived from an acid in which at least one hydroxyl group…
Q: In modern and recent application of a catalyst and an enzyme used in a chemical and biochemical…
A: Enzymes are biocatalyst that increases the speed of reaction by lowering the activation energy.…
Q: Sketch and label the various parts of transition state diagrams for a reaction with and without a…
A: Catalyst can be defined as the substance, which increases the rate of the reaction but does not…
Q: NH HO-P-o Br, in H,O OH он ОН Ba(ОН),
A: ATP→cAMP(cyclic AMP) Adenosine triphosphate reacts with barium in presence of water or with barium…
Q: Write a sample chemical reaction of chymotrypsin in a complete balanced equation label the…
A: Chymotrypsin is a site specific serine protease which cleaves a peptide at the C-terminal of all…
Q: A. Complete the table (Handwritten) Expanded Structure Fischer Projection Haworth Projection…
A: Introduction
Q: Compare and contrast catabolic and anabolic reactions
A: Metabolism is a biochemical process that allows an organism to live, grow, reproduce, heal, and…
Q: For the reaction shown, which statement is true? B Transition state EA Reactants AB AG<0 Products…
A: Reactions occur when there are changes in the reactants that break the bonds and make new bonds to…
Q: Explain the equilibria for the reactions below но
A:
Q: Classify the following reactions by what atoms do. Note: There may be more than one classification…
A: 1st reaction is a type of acid bas, gas forming reaction.. 2nd reaction is a type of redox…
Q: 9. Pb(NO3)2 (aq) + HNO3 (aq) 10. H2SO4 (aq) + NaHCO3 (aq) 11. H2SO4 (aq) + Ba(NO3)2 (aq)
A: “Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: C. b. A+B е. a. d. C+D Progress of the Reaction Free Energy
A: Gibbs free energy of reaction is equal to free energy of the product minus free energy of the…
Q: Peptide bonds are formed via what type of reaction? A. Condensation B. Hydrolysis C.…
A: When the carboxyl group of one molecule combines with the amino group of the other molecule,…
a. Refer to the reaction below and provide A,B,C and D.
b. What type of reaction is taking place in reaction D?
Step by step
Solved in 3 steps with 2 images

- Name an enzyme that acts on each molecule.(a) Amylose (b) Peroxide (c) DNAA molecule that donates electrons becomes _________, and the one that accepts the electrons becomes ________ . a. reduced; oxidized c. oxidized; reduced b. ionic; electrified d. electrified; ionicEnzymes _______. a. enhance reaction rates b. are affected by pH c. act on specific substrates d. all of the above
- Which of the 2 equations represent cellular respiration? 6CO2 + 6H2O + energy --------> C6H12O6 + 6O2 C6H12O6 + 6O2 ---------> 6CO2 + 6H2O + energyWhich enzymes are required to complete the following reactions? 16:1Δ9 →18:1Δ11→18:2Δ5, 11 A. Δ2 Desaturase; elongase B. Elongase; Δ2 desaturase C. Δ5 Desaturase; elongase D. Elongase; Δ5 desaturaseWhat enzyme converts ATP to ADP?
- Do enzymes act better under acidic or alkaline pHs? Explain your answerWhich of the following reactions would decrease the entropy within a cell? A) anabolic reaction B) synthesis of a hemoglobin molecule C) dehydration reaction D) reaction with a positive △G E) All of the aboveWhich molecule produces the most ATP?a. carbohydratesb. FADH2c. triglyceridesd. NADH



