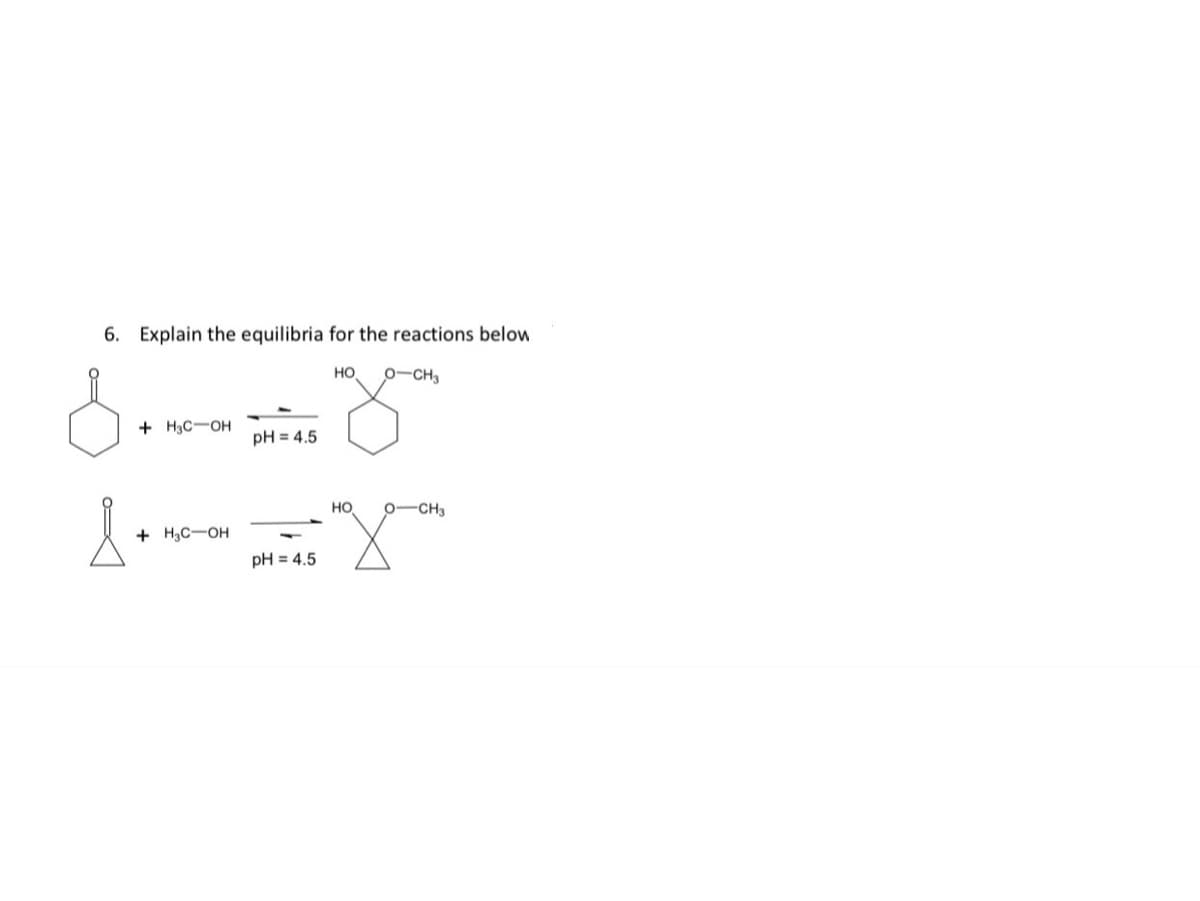
Explain the equilibria for the reactions below но
Q: Consider the multistep reaction below. What is the balanced chemical equation of the overall…
A: A chemical reaction can be fast or slow depending upon the presence or absence of catalysis. The…
Q: explain exergonic and endergonic reactions and provide examples of each?
A: Chemical reaction: It is the process in which one or more substance rearranges themselves in a…
Q: Why is it necessary to specify the temperaturewhen making a table listing ∆G values?
A: ∆G is defined the change of Gibbs (free) energy for a system; ∆G° is the Gibbs energy change for a…
Q: he following exothermic reaction is at 0.00 °C and 1.00 atm SeO4 (g) ⇌ Se(g) + O2(g) ,…
A: Chemical equilibrium is the state in which both reactants and products are present in concentrations…
Q: Identify the coefficients of the correct balanced equation for the reaction, CaCl2 + AGNO3 D AgCl +…
A: The given reaction is, CaCl2(aq)+AgNO3(aq)→ AgCl(s) +Ca(NO3)2(aq).
Q: What is the AG for the following reactions: ATP + H20 → ADP + P. AG= ADP + P → ATP + H20 AG =
A: Hi! Thank you for the questions. As you have posted multiple questions, I will be answering the…
Q: Write an unbalanced equation, using the correct formulas for all reactants and products.
A: An unbalanced equation is a reaction in which the number of atoms in the reactant is not equaled to…
Q: Give a reaction equation for the qualitative and quantitative determination of I2.
A: Iodine: Iodine (I) is a lustrous element; atomic number is 53. It is the heaviest of the stable…
Q: Define activation energy (EA)
A: The chemical process involves the reaction of substrate and enzyme for the production of product…
Q: What are the Determinants of Reaction Rates?
A: Study of chemical reactions is known as reaction kinetics. Chemical reaction refers to the reaction…
Q: Identify the oxidized reactant, the reduced reactant, the oxidizing agent, and the reducing agent in…
A: The electron accepting molecule or ion is known as oxidizing agents. As the electrons are accepted…
Q: Define the light reaction ?
A: Step 1 Photosynthesis is the procedure of the formation of organic compounds from CO2 or carbon…
Q: Identify any combination reactions.
A: Chemical combination means when two reactants combines together to form a single product. Chemical…
Q: Draw the experimental setups for the preparation of benzoin and write the reaction mechanism.
A: Introduction Benzoin condensation involves the formation of benzoin from two benzaldehyde molecules…
Q: Does a catalyst change the potential energies of the products and reactants?
A: A catalyst is a material that accelerates the rate of a reaction by lowering the activation energy…
Q: Which reactions tend to be characterized by an increase in entropy: condensation or hydrolysis…
A: Hydrolysis reactions use water to breakdown polymers into monomers. It break bonds and release…
Q: Define the following terms: a. velocity b. kinetics c. half-life d. first-order reactions e.…
A: Chemical reactions, a process in which one or more substances, the reactants are converted to one or…
Q: What is the difference between exothermic and exergonic reactions?
A: The chemical reactions are those reactions that deals with the conversion of reactants to the…
Q: Given log þ. 2. b. Balance the following reactions: S S(s) + NO(g) in acidic medium (aq) 8 (aq)…
A: Answeing 4b as you have asked. In acidic medium, protons will be there. The protons along with the…
Q: For questions 25-27, please use the diagram above. What reaction is being displayed ahove?
A: INTRODUCTION The TCA cycle is an amphibolic pathway. Anaplerotic reactions replenish TCA cycle…
Q: ifferentiate exergonic and endergonic reactions from exothermic and endothermic reactions.
A: In the absence of an enzyme rate of reaction is 30 times slower than the rate of reaction in…
Q: For the reaction below: CH3CH2I DMF
A: It is an williamson ether synthesis
Q: Consider a reaction with ΔH = 15 kJ and ΔS = 50 J · K−1. Is the reaction spontaneous (a) at 10°C,…
A: Gibbs free energy refers to the chemical energy associated with the reaction that is used to do…
Q: Calculate the heat energy released when 25.7 g of liquid mercury at 25.00 °C is converted to solid…
A: Mercury is the metal that is present in its liquid form.
Q: 11.74 The table below lists the density of O2 at various temperatures and at 1 atm. The normal…
A: Oxygen molecule (O2) Oxygen molecule (O2) is a very essential molecule of nature, it make life…
Q: Examine this chemical equation below. The reactant(s) (aka substrates), are: 6 CO2 + 6 H2O -->…
A: The given reaction is a reaction of photosynthesis. In an enzyme-catalyzed reaction, the term…
Q: Y WX WX W In the diagram, which represents a catalyst? O W O WX
A: Enzymes are catalysts which catalyze the biological reactions. Enzymes are protein molecule having a…
Q: 1. Calculate the overall AG°' (reported up to two decimal places) for the net reaction. kJ/mol 2.…
A: A system's Gibbs free energy, denoted by the letter G, is a measurement of the amount of energy that…
Q: Which has greater entropy, liquid water at 0°C or ice at 0°C?
A: The measurement of the thermal energy of the particular structure with temperature when there is no…
Q: Answer true or false to the following statements about energy diagrams and reactions. Q. An…
A: Exergonic reaction is a type of chemical reaction which releases free energy in the process of a…
Q: Compare between Kreb's reversible reactions add structures and equations# 14:0
A: Krebs cycle is also known as the citric acid cycle or tricarboxylic acid cycle (TCA). It is one of…
Q: Consider the following endothermic reaction at equilibrium: H2(g) + Zn2+(aq) ⇌ Zn(s) +…
A: The equilibrium law is also known as Le-Chateliers's principle. This law states that when an…
Q: PSII: primary electron Sun P680 oxidized to ассeptor PSI: Sun
A: Photosynthesis is a biochemical process that takes place in green plants and some lower organisms…
Q: Draw Electron distribution diagram.
A: The atomic model proposed by Bohr depicts that atom consists of a central nucleus and the electrons…
Q: e the entropy change AS universe for the following reaction, at 25 °C.
A:
Q: What can be concluded regarding a spontaneous reaction that becomes nonspontaneous when the…
A: Spontaneous reactions are favourable when they result in a decrease in the enthalpy and an increase…
Q: Briefly describe the Beer-Lambert Law.
A: Spectroscopy is a technique that is used to test the quality. It is mainly used to monitor a…
Q: Calculate the standard Gibbs energy of formation of H,O(g) at 25 °C.
A: The reaction of the formation of water is: 2H2(g)+O2(g)⟶2H2O(l) Gibb's Free Energy calculation…
Q: Classify the following reactions by what atoms do. Note: There may be more than one classification…
A: 1st reaction is a type of acid bas, gas forming reaction.. 2nd reaction is a type of redox…
Q: + H2 → Ag + HCl none of the above Identify any decomposition reactions. (Select all that apply.)…
A: The method or effect of breaking down a single chemical object into two or more fragments is known…
Q: Why are most spontaneous reactions exothermic?
A: A spontaneous process is the time-evolution of a system in which it releases free energy and it…
Q: Define the following terms: a. catalytic dyad b. transition state theory c. hydroxide ion catalysis…
A: BASIC INFORMATION ENZYMES They are the catalyst. They help in accelerating the chemical reaction.…
Q: Predict the trend of entropy in the following scenario: Entropy of vinegar v. salt A Entropy is…
A: Entropy is a thermodynamic quantity representing the degree of disorder or randomness in the system…
Q: Minor product(s): Мео
A: Elimination reaction
Q: The amount of heat in kJ required to convert 0.7 gal of liquid water to steam equal… ?
A: 0.7 gallon of water 1 gallon = 3.7854 kg water 0.7 gallon = 2.65 kg water
Step by step
Solved in 2 steps with 2 images

- A reaction at 23°C has ∆G = 1 kJ mol–¹. Why might this reaction become spontaneous at 37°C?1) what is the equilibrium constant for the reaction A + CT AT+ C? Show calculations. 2) the imageWhen the temperature rises from 100°C to 145°C, the rate of a certain reaction increases by 3.7. 1) Calculate the reaction's activation energy in J/mol 2) Determine the ratio between the rates at 100°C and 45°C?
- 1. Show the complete mechanism for the following reaction 2.) What other method or reaction also reduces benzophenone to diphenylmethane generating few pollutants to keep the environment green? Write the complete reaction.Tell whether the entropy of the following reactions will be negative, positive or zero. b) NaNO3 + H2O -> Na^+ + NO3^- c) 4H + O2 <-> 2 H2O d) 4HO2 <-> 2H2O + O2 a) Solid Carbon Dioxide <-> Gas Carbon DioxideIdentify any combination reactions. (Select all that apply.) C4H10 + O2 → CO2 + H2O S8 + O2 → SO3 NaNO3 → NaNO2 + O2 HgO → Hg + O2 Ga + H2SO4 → Ga2(SO4)3 + H2 AgCl2 + H2 → Ag + HCl none of the above Identify any decomposition reactions. (Select all that apply.) C4H10 + O2 → CO2 + H2O S8 + O2 → SO3 NaNO3 → NaNO2 + O2 HgO → Hg + O2 Ga + H2SO4 → Ga2(SO4)3 + H2 AgCl2 + H2 → Ag + HCl none of the above
- 4c give the product if the reaction will occur, write no reaction if noneWhen K > 1, which of the following statements is/are definitely TRUE? I. Products are favored II. ΔG for the reaction is positive III. kfwd > krev IV. the reaction is exothermicCalculate ΔG° (answer in kJ/mol) for each of the following reactions from the equilibrium constant at the temperature given. (d)CoO(s)+CO(g)⇌Co(s)+CO2(g) T=550°C Kp=4.90×102 (e)CH3NH2(aq)+H2O(l)⟶CH3NH3+(aq)+OH−(aq) T=25°C Kp=4.4×10−4 (f)PbI2(s)⟶Pb2+(aq)+2I−(aq) T=25°C Kp=8.7×10
- 1. What do you think is the role of papaya in cooking the chicken?2. What conclusion can you derive on the effect of catalyst based on the role of papaya in cooking the chicken?What is the half-reactions and the Ecell to this equation: 2Ag (s)+H2S (g)+ 1⁄2O2 (g) →Ag2S+H2O (l)12. Find the ΔH for the reaction below, given the following reactions and subsequent ΔH values: b. FeO(s) + CO(g) ⟶⟶ Fe(s) + CO2(g) ΔH = __________ 3Fe2O3(s) +CO(g) ⟶⟶ 2Fe3O4 +CO2 (g) ΔH = -68.26 kJ Eq 1 Fe2O3(s) + 3CO(g) ⟶⟶ 2Fe(s) + 3 CO2(g) ΔH = -23.44 kJ Eq 2 Fe3O4(s) + CO(g) ⟶⟶ 3FeO(s) + CO2(g) ΔH = +21.79 kJ Eq 3



