access Important values if needed for this question. A buffer solution is made that is 0.497 M in H2S and 0.497 M in KHS. If K for H2S is 1.00 x 107, what is the pH of the buffer solution? pH = Write the net lonic equation for the reaction that occurs when 0.137 mol HBr is added to 1.00 L of the buffer solution. (Use the lowest possible coefficients. Omlt states of matter. Use H,0 Instead of H')
access Important values if needed for this question. A buffer solution is made that is 0.497 M in H2S and 0.497 M in KHS. If K for H2S is 1.00 x 107, what is the pH of the buffer solution? pH = Write the net lonic equation for the reaction that occurs when 0.137 mol HBr is added to 1.00 L of the buffer solution. (Use the lowest possible coefficients. Omlt states of matter. Use H,0 Instead of H')
Chapter15: Acid-base Equilibria
Section: Chapter Questions
Problem 8ALQ: You have a solution of the weak acid HA and add some of the salt NaA to it. What are the major...
Related questions
Question
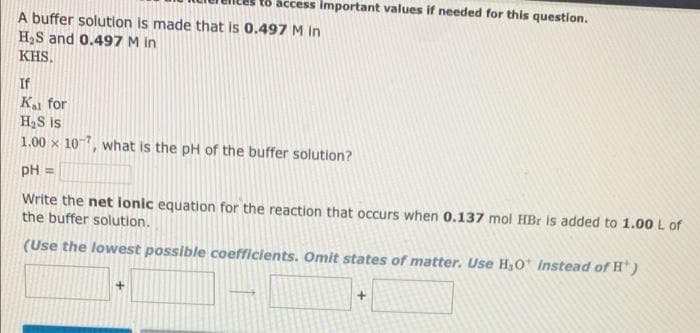
Transcribed Image Text:t8 access Important values if needed for this question.
A buffer solution is made that is 0.497 M in
H2S and 0.497 M in
KHS.
If
K for
HS is
1.00 x 107, what is the pH of the buffer solution?
pH =
Write the net ionic equation for the reaction that occurs when 0.137 mol HBr is added to 1.00 L of
the buffer solution.
(Use the lowest possible coefficients. Omit states of matter. Use H,0 Instead of H' )
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning